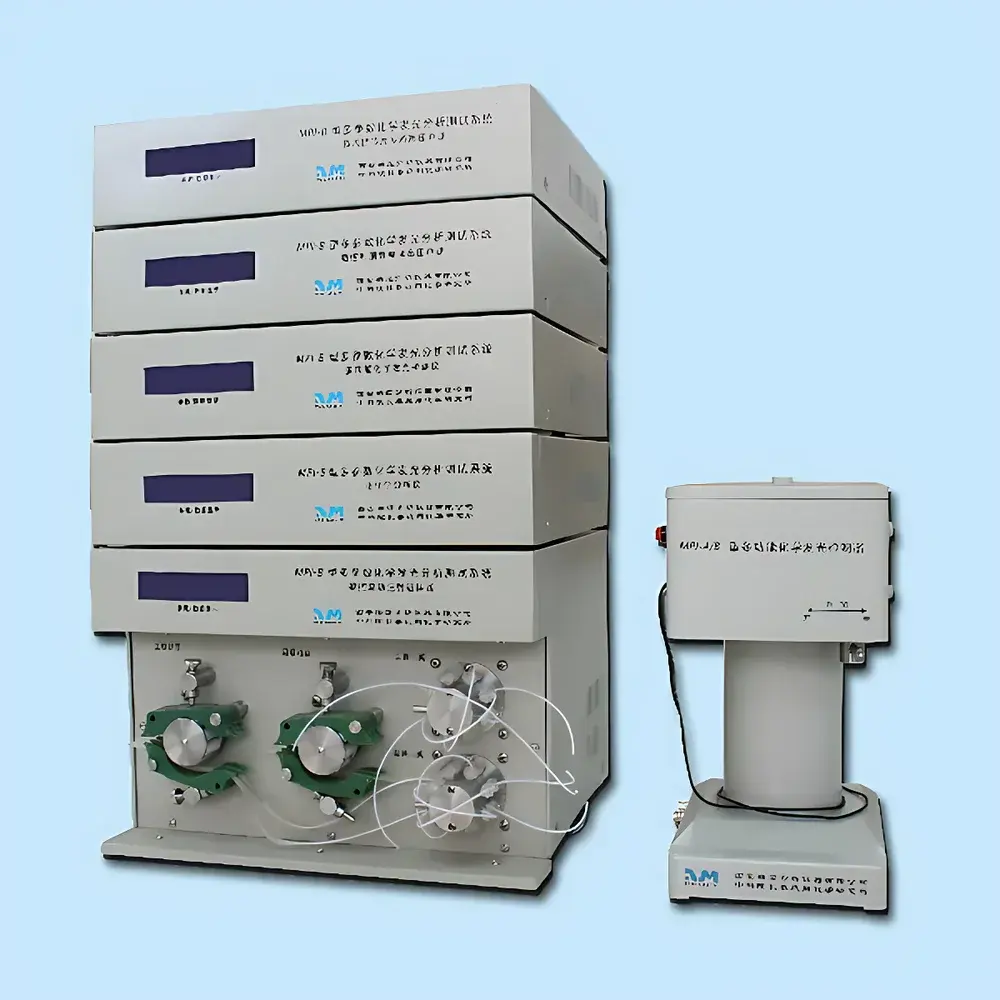
REMEX MPI-B Multi-Parameter Chemiluminescence Analysis System
| Brand | REMEX (Xi’an Remex Analytical Instrument Co., Ltd.) |
|---|---|
| Origin | Shaanxi, China |
| Model | MPI-B |
| Key Components | Integrated Electrochemical Analyzer, Multifunctional Chemiluminescence Detector, CNC Capillary Electrophoresis High-Voltage Power Supply, Microfluidic Chip Multi-Channel HV Power Supply, Flow Injection Sampler, and PMT-Based Luminescence Detection Module |
| Dynamic Range | >5 decades |
| Electrochemical Potential Range | ±10 V |
| Current Range | ±250 mA |
| CE HV Output | 0–20 kV / 0–300 µA |
| Microfluidic HV Output | 0–2000 V per channel (4 or 8 channels) |
| PMT Operating Voltage | −100 to −1000 V |
| Spectral Response | 300–650 nm (peak @ 420 nm) |
| Sampling Rate | 1–200 Hz |
| Integration Time | 0.001–10 s |
| Filter Frequencies | 10/20/50/100 Hz |
| Input Impedance (Electrochemical) | 10 MΩ |
| Bias Current | <50 pA |
| Scan Rate | 0.001–65 V/s |
Overview
The REMEX MPI-B Multi-Parameter Chemiluminescence Analysis System is a modular, research-grade analytical platform engineered for high-sensitivity detection and mechanistic investigation of chemiluminescent (CL), electrochemiluminescent (ECL), and photo-induced luminescent phenomena. Built upon a Windows-based control architecture, the system integrates six synchronized functional modules—electrochemical analysis, multifunctional CL detection, capillary electrophoresis (CE), microfluidic chip electrophoresis, flow injection analysis (FIA), and programmable high-voltage power delivery—enabling concurrent or sequential execution of orthogonal detection modalities. Its core measurement principle relies on photon emission quantification via low-noise photomultiplier tube (PMT) detection, coupled with precise electrochemical stimulus generation and separation-driven analyte enrichment. This architecture supports fundamental studies in CL reaction kinetics, ECL interface thermodynamics, and hybrid separation–detection workflows where CE or microfluidics provides spatial resolution while CL detection delivers sub-picomolar sensitivity. Designed for laboratory environments requiring method development flexibility—not routine clinical throughput—the MPI-B meets structural and functional requirements for GLP-compliant instrument qualification and supports audit trails compatible with FDA 21 CFR Part 11 data integrity frameworks when deployed with validated software configurations.
Key Features
- Modular six-unit architecture enabling simultaneous or time-resolved integration of electrochemistry, chemiluminescence, capillary electrophoresis, microfluidics, and flow injection.
- Electrochemical module delivering ±10 V potential control, ±250 mA current sourcing/sinking, and scan rates from 0.001 to 65 V/s—with <50 pA input bias current and 10 MΩ input impedance for low-noise interfacial measurements.
- Photomultiplier-based chemiluminescence detector with programmable high-voltage supply (−100 to −1000 V), spectral response from 300 to 650 nm (peak sensitivity at 420 nm), and signal-to-noise ratio >1000 A/lm.
- CNC-controlled capillary electrophoresis power supply offering 0–20 kV output with independent timing control for injection and separation phases, supporting both hydrodynamic and electrokinetic sample introduction.
- Microfluidic chip multi-channel HV source providing up to eight independently programmable outputs (0–2000 V/channel, 0–2 mA/channel), each configurable for grounded, floating, or output mode with stepwise sequence programming (up to 10 steps).
- Flow injection sampler equipped with dual five-channel peristaltic pumps (0–99 rpm digital speed control), two 8-port selection valves, and full computer-driven sequencing for FIA, SIA, CFA, and stop-flow protocols.
- Real-time signal processing with selectable filter frequencies (10/20/50/100 Hz), auto-zeroing, gain auto-control, and integration time ranging from 1 ms to 10 s—ensuring adaptability across transient and steady-state luminescence events.
Sample Compatibility & Compliance
The MPI-B accommodates liquid-phase samples compatible with aqueous or mixed organic–aqueous electrolytes, including biological fluids (serum, urine, cell lysates), environmental extracts, pharmaceutical formulations, and synthetic reaction mixtures. It supports direct injection of clarified samples into CE or microfluidic channels and accommodates derivatized or native analytes amenable to oxidation/reduction-triggered CL or ECL (e.g., luminol, ruthenium complexes, acridinium esters). The system’s hardware and firmware architecture conforms to IEC 61010-1 safety standards for laboratory electrical equipment. When operated with version-controlled software and documented configuration files, it supports instrument qualification (IQ/OQ/PQ) per ISO/IEC 17025 and aligns with analytical method validation principles outlined in ICH Q2(R2) and USP . Data acquisition logs—including timestamped parameter sets, HV settings, PMT gain, integration windows, and valve/pump actuation events—are stored in structured binary formats with optional CSV export, facilitating traceability under GLP or GMP-aligned workflows.
Software & Data Management
Control and data acquisition are managed through a native Windows application developed in C++ with Qt framework, featuring a tabbed interface for module-specific configuration, real-time waveform visualization, and post-acquisition spectral deconvolution tools. All hardware modules communicate via USB 2.0 using vendor-defined HID-class protocols with CRC-verified command packets. Raw data files include embedded metadata (instrument ID, operator name, date/time, method name, parameter snapshots) and support HDF5 container format for long-term archival. Software includes built-in calibration utilities for PMT gain normalization, electrochemical current scaling, and CE migration time referencing. Export options include ASCII (tab-delimited), MATLAB .mat, and generic XML schema—enabling interoperability with third-party chemometrics packages (e.g., MATLAB Statistics Toolbox, Python SciPy). Audit trail functionality records user login/logout, method modification history, and data export actions; optional database back-end (SQLite or Microsoft SQL Server) enables role-based access control and electronic signature enforcement per 21 CFR Part 11 Annex 11 requirements.
Applications
- Mechanistic studies of chemiluminescent reaction pathways, including radical intermediates, energy transfer efficiency, and solvent effects on light emission quantum yield.
- Development of ECL biosensors for immunoassays, DNA hybridization detection, and aptamer-based target recognition—leveraging electrode surface modification and redox-cycling amplification strategies.
- Hyphenated CE–CL analysis of complex biological matrices (e.g., catecholamines in cerebrospinal fluid, drug metabolites in plasma), where electrophoretic separation precedes zone-specific CL detection without post-column derivatization.
- Microfluidic ECL immunoassays integrating on-chip antibody immobilization, magnetic bead capture, and localized electrochemical excitation—reducing assay time from hours to minutes.
- Quantitative determination of reactive oxygen species (ROS), nitric oxide derivatives, and enzymatic activity (e.g., NADH oxidase, xanthine oxidase) via CL intensity correlation with analyte concentration.
- Method validation studies supporting regulatory submissions, including limit of detection (LOD) assessment, precision profiling (intra-day/inter-day RSD), and robustness testing across voltage, temperature, and reagent batch variables.
FAQ
Is the MPI-B suitable for regulated clinical diagnostics use?
No—it is designed for research, method development, and preclinical assay optimization. It lacks CE-IVD marking, clinical validation documentation, and locked-down software required for in vitro diagnostic use.
Can the system be integrated with third-party HPLC or CE instruments?
Yes—via TTL-triggered synchronization signals and analog voltage inputs/outputs; however, external instrument control requires custom scripting or OPC UA middleware implementation.
What maintenance is required for the PMT detector?
Annual dark-current verification and factory recalibration are recommended; PMT gain stability is monitored via internal reference LED pulses during each run.
Does the software support automated peak identification and integration?
Yes—using derivative-threshold algorithms and customizable baseline correction models; results can be exported with retention/migration time, peak area, and signal-to-noise ratios.
Are consumables such as capillaries or microfluidic chips supplied by REMEX?
REMEX provides technical specifications and compatibility guidance but does not manufacture or distribute consumables; users procure fused-silica capillaries or PDMS/glass chips from certified vendors meeting ISO 8573-1 purity standards.