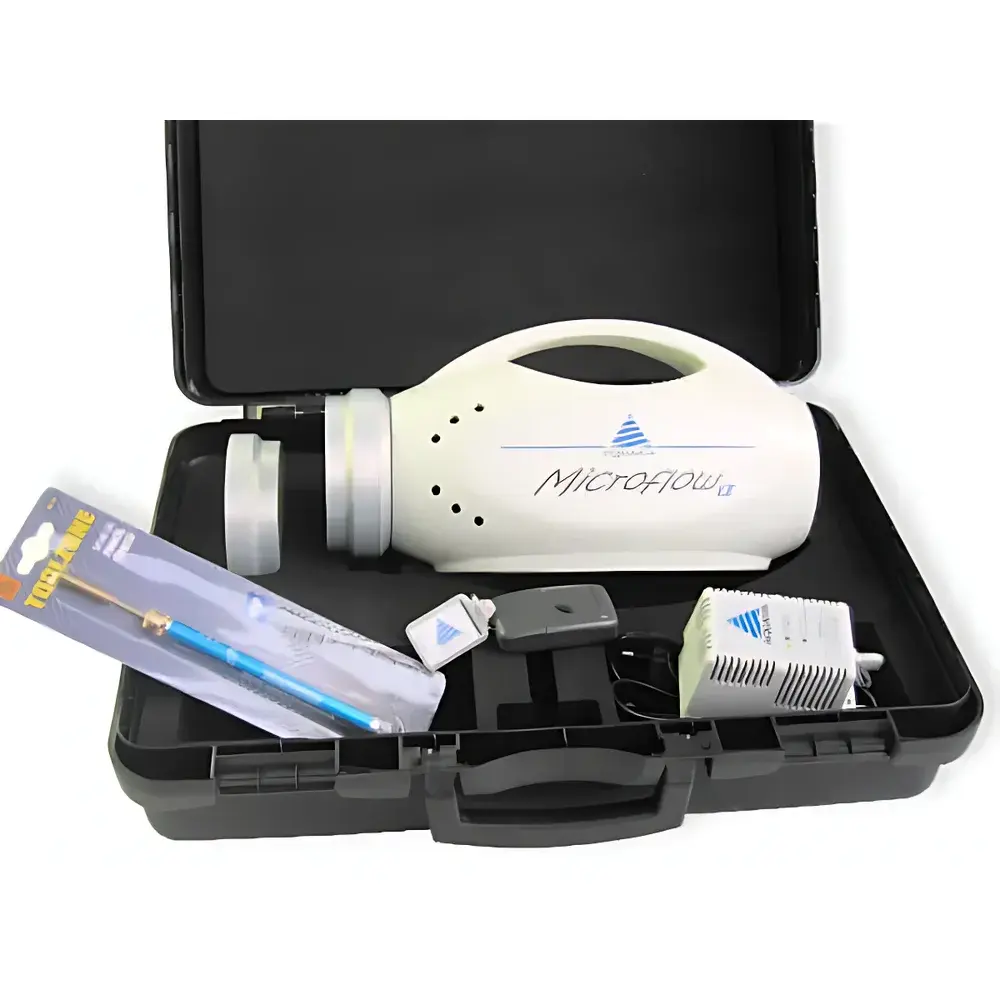
Aquaria MICROFLOW α Portable Air Microbial Sampler
| Brand | Aquaria |
|---|---|
| Origin | Italy |
| Model | MICROFLOW α |
| Sampling Principle | Impaction-Based |
| Flow Rates | 28.3–120 L/min (up to 180 L/min peak) |
| Culture Dish Compatibility | Ø60 ± 2 mm and Ø90 ± 2 mm Petri Dishes |
| Display | LCD with Multilingual Interface (English, Italian, French, German, Spanish) |
| Housing Material | Medical-Grade Polyurethane |
| Sampling Head | Anodized Aluminum, Autoclavable |
| On-Site Sterilization | Integrated UV Sterilization Pen |
| Operating Modes | Programmable, Continuous Programmed, Delayed Start, Remote-Controlled Sampling |
| Weight | <2.5 kg |
| Dimensions | 220 × 140 × 180 mm (W × D × H) |
Overview
The Aquaria MICROFLOW α is a portable, impaction-based air microbial sampler engineered for quantitative collection of viable airborne microorganisms—including bacteria, fungi, yeasts, and spores—onto standard agar culture media. Its operational principle relies on inertial impaction: aerosol-laden air is drawn at controlled flow rates through precision-machined nozzles, accelerating particles toward the surface of a rotating or static Petri dish. Particles with sufficient aerodynamic diameter (>0.6 µm) impact the nutrient agar surface with high deposition efficiency and minimal desiccation, preserving viability for subsequent colony-forming unit (CFU) enumeration and identification. Designed and manufactured in Italy, the MICROFLOW α meets ISO 14698-1:2003 requirements for biocontamination control in cleanrooms and controlled environments, and aligns with Chinese national standard GB/T 18204.1–2013 for air microbial sampling by impaction. It serves as a primary tool for environmental monitoring in regulated pharmaceutical, biotechnology, hospital, and public health settings where trace-level microbial load assessment is critical.
Key Features
- Medical-grade polyurethane housing: Chemically inert, non-shedding, and resistant to mechanical stress and repeated disinfection cycles.
- Autoclavable anodized aluminum sampling head: Withstands steam sterilization at 121°C for 20 minutes; compatible with ISO 14644-1 and EU GMP Annex 1 cleaning validation protocols.
- Dual-dish compatibility: Supports both Ø60 ± 2 mm and Ø90 ± 2 mm Petri dishes—enabling flexible assay design, including comparative sampling or multi-target media deployment.
- Integrated UV sterilization pen: Delivers localized 254 nm germicidal irradiation for rapid decontamination of sampling surfaces between runs, minimizing cross-contamination risk in field or mobile lab use.
- Intuitive LCD interface with five-language support (English, Italian, French, German, Spanish): Ensures operational consistency across multinational teams and regulatory audits.
- Compact ergonomic form factor (220 × 140 × 180 mm; <2.5 kg): Optimized for handheld operation, cart-mounted deployment, or integration into mobile environmental monitoring carts.
Sample Compatibility & Compliance
The MICROFLOW α accommodates standard microbiological growth media—including Tryptic Soy Agar (TSA), Sabouraud Dextrose Agar (SDA), and malt extract agar—without modification. Its impaction geometry ensures >95% collection efficiency for particles ≥1.0 µm at 100 L/min, per ISO 8573-7:2010 test methodology. Device calibration is traceable to NIST-certified flow meters, and routine verification follows ISO/IEC 17025-compliant procedures. The instrument supports GLP and GMP documentation workflows: all sampling events—including start/stop timestamps, flow rate, duration, and user ID—are logged internally and exportable via USB for audit-ready reporting. It complies with FDA 21 CFR Part 11 when paired with validated data management software, enabling electronic signatures and secure audit trails.
Software & Data Management
The MICROFLOW α operates autonomously but interfaces with Aquaria’s optional MICROFLOW Connect desktop application (Windows/macOS) for advanced configuration, real-time telemetry, and batch report generation. The software enables creation of multi-step sampling schedules, remote firmware updates, and export of CSV/Excel-compatible logs with metadata fields (location ID, operator, environmental conditions). All stored data are encrypted at rest; user access levels (admin/operator/viewer) enforce role-based permissions aligned with ALCOA+ data integrity principles. Audit trail records include timestamped entries for parameter changes, calibration actions, and sterilization events—fully compliant with EMA Annex 11 and WHO TRS 992 Annex 5 requirements.
Applications
- Aerosol science research: Characterizing size-resolved microbial emission profiles from HVAC systems, medical devices, or industrial processes.
- Pharmaceutical manufacturing: Environmental monitoring in Grade A/B cleanrooms per EU GMP Annex 1 and USP , including routine surveillance and investigation of out-of-limit (OOL) events.
- Public health surveillance: Field deployment by CDC-equivalent agencies for outbreak tracing, indoor air quality assessments in schools, transport hubs, and elderly care facilities.
- Hospital infection control: Monitoring surgical suites, isolation rooms, and pharmacy compounding areas to validate air handling performance and HEPA filtration integrity.
- Biotechnology R&D labs: Assessing biosafety cabinet efficacy, containment validation for BSL-2 workspaces, and microbial challenge testing of air purification technologies.
FAQ
What is the recommended calibration frequency for the MICROFLOW α?
Annual calibration is advised per ISO/IEC 17025 guidelines; interim verification using a calibrated rotameter or thermal mass flow meter should be performed before each critical sampling campaign.
Can the MICROFLOW α be used in laminar flow hoods without disrupting airflow patterns?
Yes—the device’s low-profile design and optional suction hose extension minimize turbulence; placement should follow ISO 14644-3:2019 positioning recommendations for point sampling in unidirectional airflow zones.
Is the UV sterilization pen validated against common healthcare pathogens?
The 254 nm UV-C source delivers ≥40 mJ/cm² dose within 15 seconds, exceeding log₄ reductions for Staphylococcus aureus, Bacillus subtilis spores, and Aspergillus niger per ISO 15714:2019.
Does the instrument support integration with LIMS platforms?
Yes—CSV exports contain structured metadata fields compatible with major LIMS vendors (e.g., LabWare, Thermo Fisher SampleManager); API integration is available under NDA for enterprise deployments.
What maintenance is required beyond routine sterilization?
No consumable parts require replacement; annual inspection of nozzle alignment, flow sensor drift, and battery health is recommended using Aquaria’s certified service centers in Europe and APAC regions.