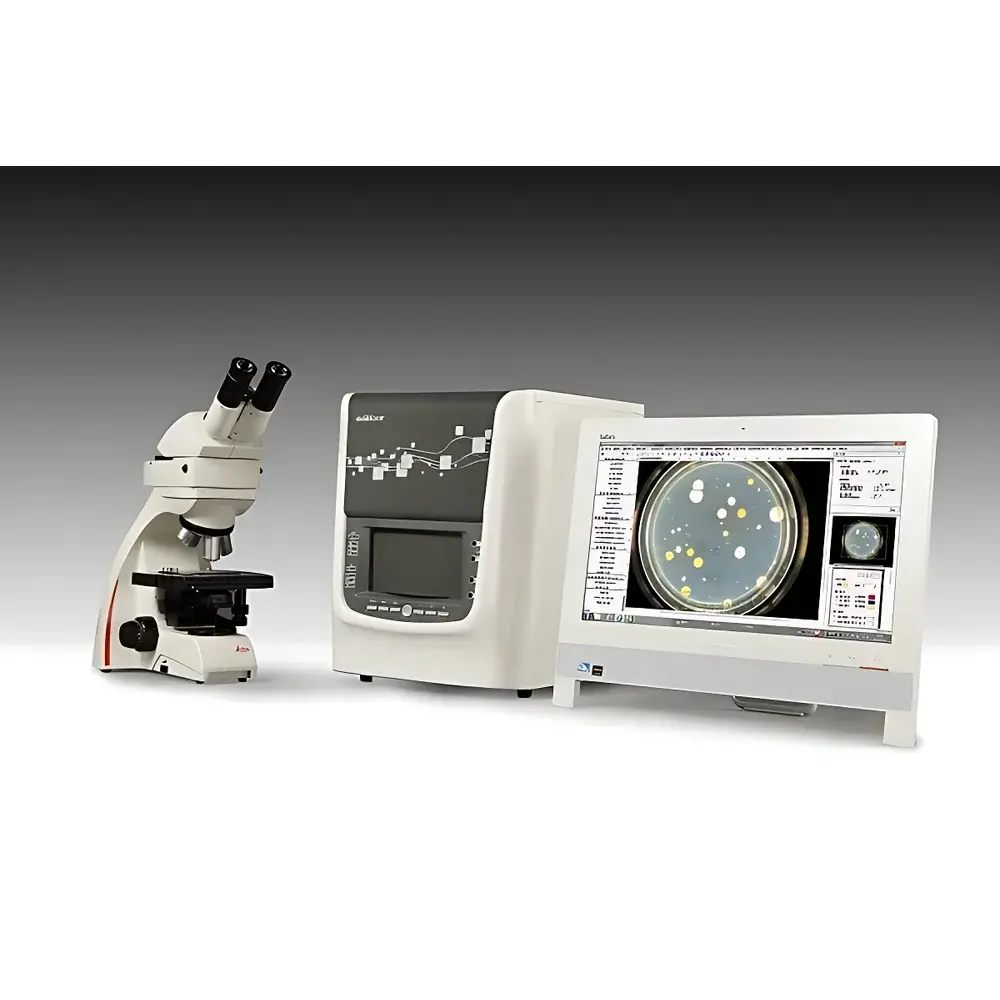
Xunsu MF5 Multifunctional Colony Counter & Microscopic Image Analysis System
| Brand | Xunsu |
|---|---|
| Origin | Zhejiang, China |
| Manufacturer Type | OEM Manufacturer |
| Instrument Type | Fully Automated Colony Counter |
| Counting Speed | <1 second for up to 500 colonies |
| Microscope | Leica DM750 Trinocular Microscope |
| Microscope Camera | Sony 2/3" Global Shutter CMOS, 5 MP, 35 fps |
| Colony Imaging Sensor | 4/3" Color CMOS, 16 MP |
| Lens | 16 mm High-Resolution Fixed-Focal Lens |
| Illumination | Adjustable 3500–8500 K Tri-LED White Light, 254 nm UV Lamp, Top 360° Shadowless Light, Bottom Suspended Dark-Field Light |
| Compliance | Meets requirements of Chinese Pharmacopoeia (2015 Edition), FDA 21 CFR Part 11 (audit trail, electronic signature, data integrity), GLP/GMP-aligned architecture |
Overview
The Xunsu MF5 Multifunctional Colony Counter & Microscopic Image Analysis System is an integrated, high-precision platform engineered for quantitative microbiology and cellular morphometric analysis in regulated laboratory environments. It combines three core imaging subsystems—colony digital imaging,抑菌圈 (zone-of-inhibition) quantification, and high-resolution microscopic cell imaging—within a single, unified hardware and software architecture. The system operates on dual optical pathways: a macro-scale colony imaging module based on high-fidelity visible-light capture with dynamic illumination control, and a micro-scale imaging module anchored by a Leica DM750 trinocular microscope coupled to a Sony global shutter CMOS sensor. Its analytical foundation rests on advanced image segmentation algorithms—including fourth-generation level-set active contour models constrained by RGB color space, morphology, and topological adaptability—enabling robust separation of microbial colonies from heterogeneous backgrounds (e.g., agar gradients, membrane filters, 3M Petrifilm™), even under challenging conditions such as high clumping density, low contrast, or overlapping growth zones. Designed for compliance-critical applications in pharmaceutical QC, food safety testing, clinical microbiology, and academic research, the MF5 implements a four-tier user permission model and full audit-trail logging aligned with FDA 21 CFR Part 11, ISO/IEC 17025, and Chinese GMP guidelines.
Key Features
- Tri-modal illumination system: Adjustable 3500–8500 K tri-LED white light for true-color colony representation; 254 nm UV lamp for chamber decontamination and mutagenesis studies; top 360° shadowless illumination for surface texture enhancement; bottom suspended dark-field illumination for precise edge delineation of fungal hyphae and actinomycete colonies.
- High-fidelity macro-imaging: 4/3″ 16 MP CMOS sensor paired with a 16 mm large-format fixed-focus lens (150 lp/mm resolution) and fully enclosed dark-box enclosure to eliminate ambient stray light interference.
- Microscopic imaging module: Leica DM750 trinocular microscope with C-mount adapter and Sony 2/3″ global shutter CMOS camera (5 MP, 35 fps), enabling motion-free capture of live, motile cells without image lag or smearing.
- Advanced segmentation engine: Proprietary level-set active contour algorithms—including “Fast Active Contour”, “RGB-Constrained Color Level Set”, “Multi-phase Level Set”, and “Morphology-Constrained Grid-Aware Segmentation”—optimized for complex colony types (e.g., mucoid, filamentous, pigmented, or mixed-species cultures) and specialized substrates (e.g., 3M test plates, filter membranes).
- Regulatory-grade data governance: Four-role access architecture (System Administrator, Data Manager, Operator, Reviewer); automated timestamped electronic signatures; immutable database logging of all image files, dilution factors, algorithm parameters, manual corrections, and statistical outputs; watermarked final reports with tamper-evident digital seals.
Sample Compatibility & Compliance
The MF5 supports diverse sample formats including pour plates, spread plates, membrane filtration units, spiral plating (FDA-compliant counting modes), multi-well microtiter plates (for OPKA/SBA assays), 3M Petrifilm™ products (Total Aerobic Count, Staphylococcus aureus, Coliform/E. coli), and grid-patterned filters. Its image processing pipeline accommodates variable background textures—such as uneven agar opacity, condensation droplets, or residual pen markings—via dedicated background restoration tools (e.g., ink-removal and stain-elimination modules). Regulatory alignment includes full support for Chinese Pharmacopoeia 2015 Edition methods for antibiotic potency determination (two-dose and three-dose assay protocols), microbial limit tests, and β-lactamase detection using sulbactam-sensitive disk diffusion. System validation documentation satisfies ISO 13485, USP /, and EU Annex 11 requirements for computerized system validation in GxP environments.
Software & Data Management
The MF5 runs a unified software suite featuring modular, context-aware analysis workflows: Colony Analysis, Zone-of-Inhibition Measurement (Szone™), Antibiotic Potency Calculation, β-Lactamase Screening, and Microscopic Particle Quantification. All modules share a common database backend with encrypted storage, role-based view permissions, and export capabilities to PDF, Excel, and CSV. Audit trail records include operator ID, timestamp, original image hash, applied algorithm settings, correction history, and reviewer approval status—fully traceable via the “Intelligent Test Flow Reconstruction” function. Data integrity safeguards include automatic watermarking of approved reports, cryptographic hashing of raw images, and write-once archival mode for finalized datasets. Optional integration with LIMS via HL7 or RESTful API enables seamless workflow handoff in centralized QA/QC laboratories.
Applications
The MF5 serves mission-critical functions across multiple domains: pharmaceutical stability and sterility testing; food and beverage pathogen enumeration (e.g., E. coli, S. aureus, L. monocytogenes); environmental monitoring in cleanrooms and water systems; antimicrobial susceptibility profiling (Kirby-Bauer, broth microdilution follow-up); immunological assays (OPKA, SBA); fungal quantification and hyphal growth kinetics; clonogenic survival assays in oncology research; and rapid screening of enzyme-producing strains (e.g., amylase, protease, lipase) via hydrolysis zone analysis. Its ability to perform simultaneous multi-region counting, color-based classification of up to 24 chromatic variants, and standardized morphological descriptors (e.g., diameter, circularity, texture gradient, RGB-based hue quantification) makes it uniquely suited for comparative microbiome studies and strain characterization workflows.
FAQ
Does the MF5 comply with FDA 21 CFR Part 11 requirements for electronic records and signatures?
Yes—the system implements full Part 11 compliance through mandatory user authentication, audit-trail logging of all data modifications, electronic signatures with time-stamped identity verification, and tamper-proof report generation with embedded watermarks and digital seals.
Can the MF5 accurately count colonies on 3M Petrifilm™ or black-grid membrane filters?
Yes—its proprietary “Morphology-Constrained Level Set” segmentation algorithm explicitly distinguishes colony pixels from grid lines or membrane artifacts, enabling one-click enumeration without manual masking or background subtraction.
What microscopy capabilities does the MF5 provide beyond basic imaging?
It supports real-time dynamic imaging at 35 fps, sub-pixel morphometric measurement (diameter, area, perimeter, angle, arc length), RGB-adjusted contrast enhancement, six-filter noise reduction, and customizable annotation layers for publication-ready figure preparation.
How does the system ensure data integrity during multi-user operation?
Through a four-tier permission architecture, immutable database logging, automatic electronic signatures, and “test environment reconstruction” that allows reviewers to replay the exact imaging and analysis conditions used by the operator—ensuring full traceability and reproducibility.
Is the MF5 validated for pharmacopoeial antibiotic potency testing?
Yes—it implements all calculation logic and statistical validation criteria specified in the Chinese Pharmacopoeia 2015 Edition for two-dose and three-dose assays, including parallel-line analysis, validity checks, and relative error reporting (<0.01% repeatability, <0.1% uniformity).