ELGA PURELAB Chorus 1/2/3 Ultra-Pure Water System
| Brand | ELGA (Veolia Water Technologies) |
|---|---|
| Origin | United Kingdom |
| Manufacturer | ELGA LabWater, Veolia |
| Product Type | Modular Laboratory Water Purification System |
| Model | CHORUS 1 / CHORUS 2 / CHORUS 3 |
| Pure Water Grade | Type I+ / Type I / Type II / Type III (RO) |
| Resistivity | ≥18.2 MΩ·cm at 25 °C |
| Heavy Metals & Soluble Silica | <0.1 ppb |
| Total Organic Carbon (TOC) | 1–3 ppb |
| Microbial Content | <0.001 EU/mL |
| Particulates (>0.1 µm) | <0.05 particles/mL |
| Flow Rate (CHORUS 3 RO) | Up to 120 L/h |
| Compliance | ISO 9001:2008, ISO 14001:2004, IEC/EN 61010-1, UL 61010-1, CE, WEEE, RoHS |
Overview
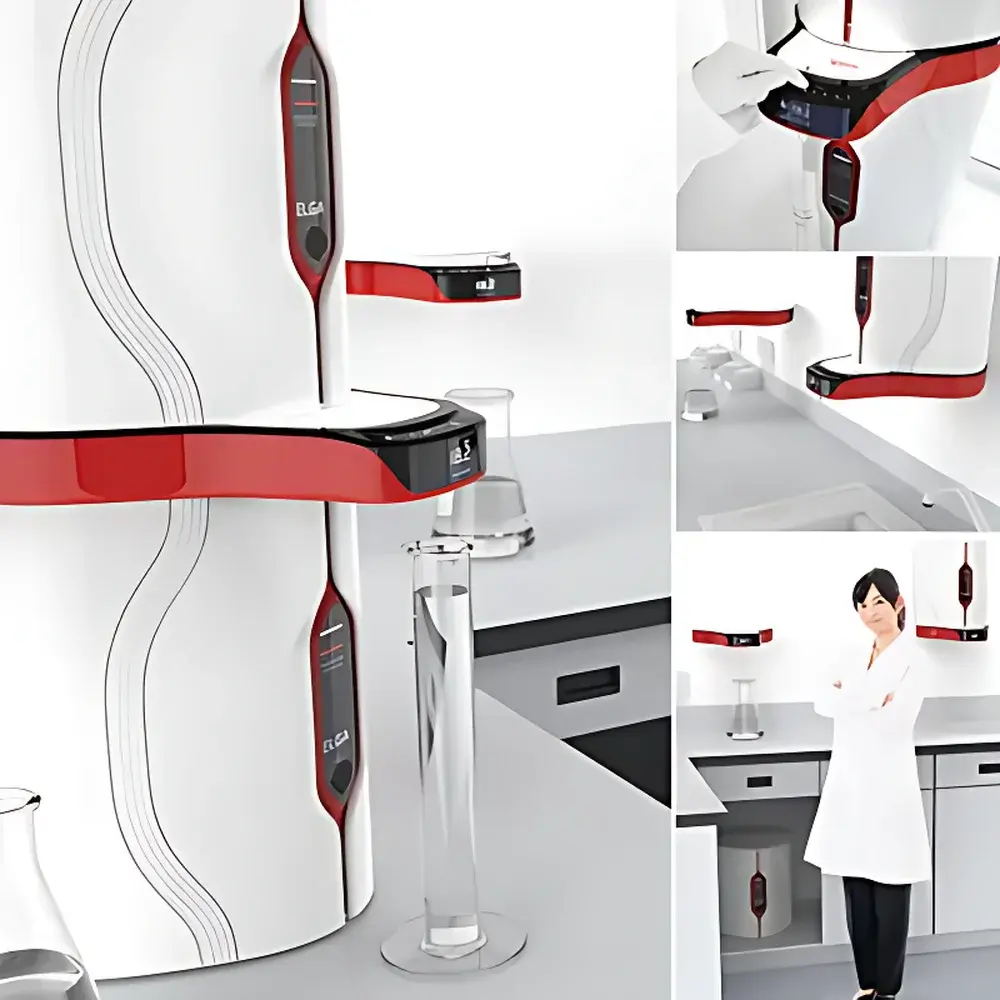
The ELGA PURELAB Chorus 1/2/3 is a modular, application-driven ultra-pure water purification platform engineered for precision-critical laboratory environments. Designed and manufactured in the UK by ELGA LabWater — a Veolia Water Technologies company — the system employs multi-stage purification architecture grounded in reverse osmosis (RO), electrodeionization (EDI), dual-wavelength UV oxidation (185/254 nm), and continuous recirculation with integrated PureSure™ deionization technology. Unlike conventional single-path systems, Chorus implements real-time, on-line monitoring of resistivity, TOC, temperature, flow, and pressure at point-of-use — enabling traceable, auditable water quality data aligned with GLP, GMP, and ISO/IEC 17025 requirements. The platform’s modularity allows laboratories to deploy only the purification stages necessary for their specific grade requirements: Chorus 1 delivers Type I+ water (≥18.2 MΩ·cm, TOC ≤3 ppb, endotoxin <0.001 EU/mL) for ultra-trace analytical applications; Chorus 2 supplies robust Type II water optimized for high-volume cell culture and routine analytical workflows; and Chorus 3 provides cost-efficient Type III (RO-grade) water — validated to 1–50 µS/cm conductivity — for feedwater, glassware rinsing, and preparatory tasks.
Key Features
- Modular architecture supporting scalable deployment of Type I+, I, II, or III water — configured per application demand, not over-specification
- PureSure™ dual-column, dual-sensor deionization system with automatic column switching — ensures uninterrupted operation and eliminates downtime during cartridge replacement
- Continuous on-line TOC monitoring (1–3 ppb range) with UV/persulfate oxidation and NDIR detection — compliant with ASTM D5542 and USP
- Full-system automated sanitization cycle using hot water (80–85 °C) and ozone-free thermal disinfection — validated to eliminate biofilm formation across all wetted surfaces (including reservoirs, pumps, and distribution loops)
- Halo™ remote dispensing ecosystem: supports up to four independent dispensers (Halo, Halo Advanced, Halo Flex) with programmable volume delivery (50 mL–60 L), drip-to-2 L/min flow control, footswitch compatibility, and handheld flexibility
- Integrated 15–100 L stainless-steel reservoirs with CO₂-absorbing and sterile vent filters — designed to maintain stored water purity without degradation over time
- Comprehensive digital interface with audit trail, user access levels, alarm logging, and exportable CSV reports — supports 21 CFR Part 11 compliance when paired with ELGA’s validated software package
Sample Compatibility & Compliance
The PURELAB Chorus series is validated for use across life science, pharmaceutical, clinical diagnostics, and materials research laboratories where water quality directly impacts assay integrity, instrument longevity, and regulatory submission validity. All models meet or exceed ASTM D1193 Type I, II, and III specifications. Chorus 1 complies with ISO 3696 Class 1, CLSI EP28-A3c, and USP for purified water used in parenteral preparation and molecular biology. Its endotoxin specification (<0.001 EU/mL) satisfies FDA guidance for cell therapy manufacturing. The system’s construction adheres to ISO 14644-1 Class 5 cleanroom-compatible materials (electropolished 316L SS, PTFE, and sanitary diaphragm valves), and its electrical safety conforms to IEC/EN 61010-1 and UL 61010-1 standards. Full factory validation documentation — including IQ/OQ protocols, calibration certificates, and material traceability — is provided with each unit.
Software & Data Management
Chorus integrates with ELGA’s AquaManager™ software suite, enabling centralized fleet monitoring, predictive maintenance alerts, consumables lifecycle tracking, and electronic logbook generation. The embedded controller records timestamped measurements for resistivity, TOC, UV absorbance, flow rate, and sanitizer cycle parameters — all stored with immutable timestamps and user authentication. Audit trails are exportable in PDF or CSV format and support ALCOA+ principles (Attributable, Legible, Contemporaneous, Original, Accurate, Complete, Consistent, Enduring, Available). When deployed in regulated environments, optional 21 CFR Part 11-compliant electronic signatures, role-based access controls, and electronic record retention policies can be activated via firmware configuration — eliminating manual paper logs while satisfying FDA, EMA, and MHRA inspection expectations.
Applications
- Chorus 1: ICP-MS, HPLC-MS, GFAAS, single-cell RNA sequencing, CRISPR-Cas9 editing, low-background radiometric assays, and sterile mammalian cell culture
- Chorus 2: Routine HPLC, IC, AAS, ELISA/RIA, electrophysiology, tissue culture media preparation, and buffer formulation requiring low ionic contamination
- Chorus 3: Feedwater for ultrapure systems, autoclave and steam generator supply, general lab cleaning, hydroponics, environmental chamber humidification, and reagent dilution
- All configurations support integration into central utility plants, cleanroom water loops, and multi-user facility networks — with configurable loop circulation rates and pressure stabilization
FAQ
What distinguishes Chorus 1 from standard Type I water systems?
Chorus 1 incorporates PureSure™ dual-column monitoring and real-time TOC verification — ensuring sustained removal of boron, silica, and organic leachables below detection limits, critical for sub-ppt elemental analysis.
Can Chorus systems be validated for GMP compliance?
Yes — IQ/OQ documentation, calibration records, and traceable sensor certifications are supplied. Optional PQ support and 21 CFR Part 11-ready software enable full GMP alignment.
How does the Halo dispensing system improve lab safety and space utilization?
Halo decouples dispensing hardware from the purifier — allowing wall-mount, under-bench, or overhead installation while maintaining full water quality monitoring at point-of-use.
Is microbial control maintained during extended storage?
Yes — the thermally sanitized reservoir system, combined with sterile venting and recirculation through 0.1 µm final filters, prevents colony-forming unit (CFU) proliferation for up to 72 hours post-purification.
What maintenance intervals are required for Chorus units?
Pre-filters require quarterly replacement; RO membranes every 2–3 years; and PureSure™ cartridges every 6–12 months depending on feedwater quality — all tracked automatically via the onboard system.