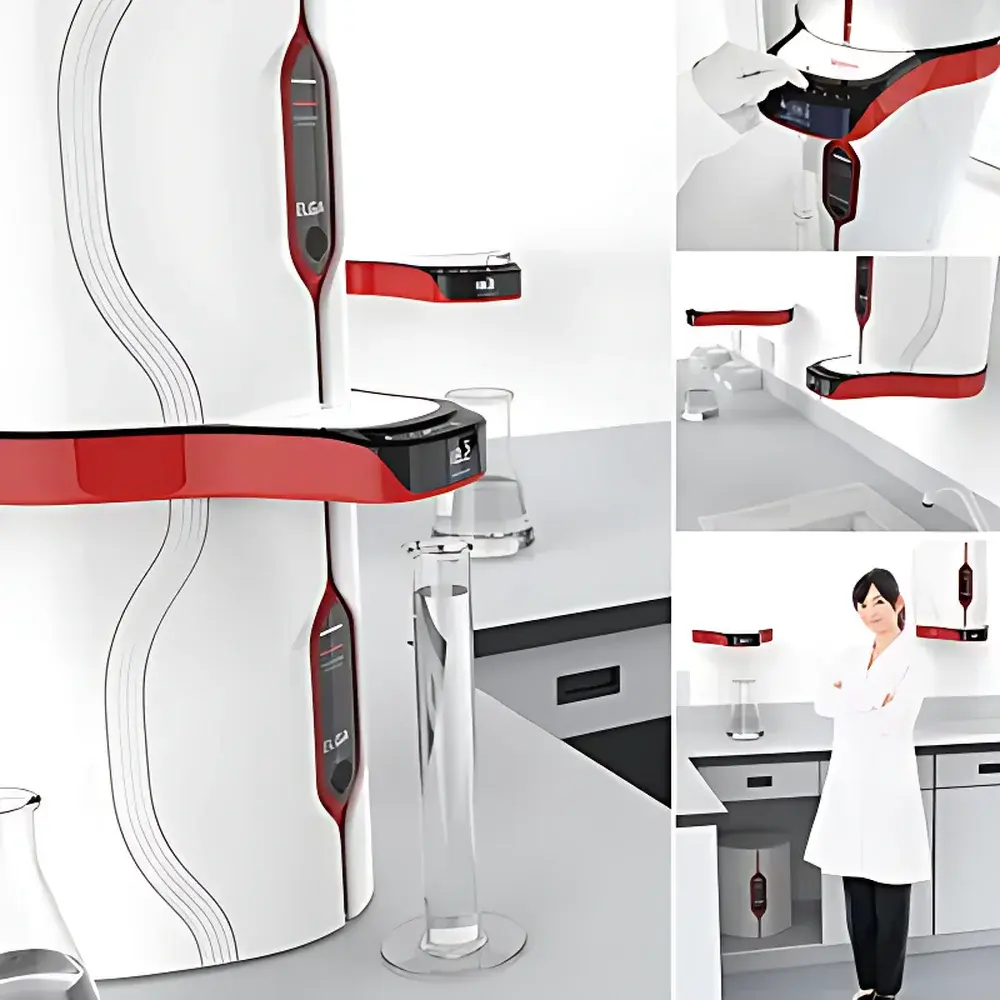
ELGA PURELAB® Chorus Series Ultra-Pure Water System
| Brand | ELGA |
|---|---|
| Origin | United Kingdom |
| Model | Chorus 1 / Chorus 2 / Chorus 3 |
| Pure Water Grade | Type I+/I (Chorus 1), Type II (Chorus 2), Type III (Chorus 3) |
| Resistivity | 18.2 MΩ·cm @ 25 °C |
| Heavy Metals & Soluble Silica | <0.1 ppb |
| Total Organic Carbon (TOC) | 1–3 ppb |
| Microbial Count | <0.001 EU/mL |
| Particulates (>0.1 µm) | <0.05 µm |
Overview
The ELGA PURELAB® Chorus Series is a modular, application-driven ultra-pure water system engineered for laboratories requiring rigorous control over water purity across multiple quality tiers—Type I+/I (ultra-pure), Type II (high-purity), and Type III (general-purpose purified water). Unlike monolithic purification platforms, the Chorus architecture employs a scalable, configuration-based design grounded in validated separation science: reverse osmosis (RO) as primary pretreatment, followed by electrodeionization (EDI), dual-stage ion exchange with PureSure® monitoring, and optional UV photo-oxidation and 0.1 µm final filtration. Each model integrates real-time electrochemical and optical sensing to continuously verify resistivity, TOC, endotoxin, and particulate compliance—ensuring water quality remains within defined specifications at the point of use, not just at the generation unit. The system adheres to ISO 3696:1987 (Grade 1), ASTM D1193-06 (Type I), and CLSI EP22-A guidelines for laboratory water quality verification, supporting regulatory readiness for GLP, GMP, and ISO/IEC 17025 environments.
Key Features
- Modular platform supporting three distinct water grades: Chorus 1 (Type I+/I, 18.2 MΩ·cm, TOC ≤3 ppb), Chorus 2 (Type II, ≤1 µS/cm conductivity), and Chorus 3 (Type III, RO-only, 1–50 µS/cm)
- PureSure® dual-column ion exchange with real-time conductivity monitoring—enables seamless column switching without interruption to water flow or quality degradation
- Integrated online TOC monitor (UV/persulfate oxidation + NDIR detection) providing continuous, trace-level organic carbon quantification at point-of-use
- Full-system automated sanitization cycle using hot water (≥80 °C) or ozone—validated per ASTM F1980 for biofilm prevention across all wetted surfaces including reservoirs, distribution loops, and delivery manifolds
- Halo® flexible dispensing architecture: supports up to four independent dispensers (Halo, Halo Advanced, Halo Flex) with programmable volume delivery (50 mL–60 L), variable flow (0.1–2 L/min), footswitch activation, and handheld operation
- Compact, space-optimized footprint with optional under-bench, wall-mounted, or benchtop installation; integrated or standalone tank configurations (15, 30, 60, or 100 L) featuring CO₂-absorbing and sterile-grade vent filters
Sample Compatibility & Compliance
The PURELAB Chorus Series delivers water compliant with internationally recognized standards for analytical, biological, and clinical applications. Chorus 1 output meets or exceeds requirements for ICP-MS, HPLC, IC, GFAAS, and mammalian cell culture per ISO 15189 Annex B and USP . Chorus 2 satisfies ASTM D1193 Type II specifications for buffer preparation, electrophoresis, and general analytical chemistry. Chorus 3 fulfills ISO 3696 Grade 3 and CLSI GP12-A3 criteria for glassware rinsing, autoclave feed, and reagent dilution. All models support FDA 21 CFR Part 11-compliant audit trails when paired with ELGA’s DataTrace® software—recording operator actions, calibration events, alarm history, and system diagnostics with electronic signature capability.
Software & Data Management
DataTrace® is the embedded supervisory software enabling full lifecycle traceability of water quality parameters and system performance. It logs timestamped resistivity, TOC, temperature, pressure, UV intensity, and sanitizer cycle completion status with configurable data export (CSV, PDF, XML). Alarm thresholds are user-definable per parameter, triggering visual/audible alerts and email/SNMP notifications upon deviation. Audit trail integrity is maintained through role-based access control (RBAC), immutable event logging, and cryptographic hash verification of historical records—meeting ALCOA+ principles for data reliability. Optional integration with LIMS via OPC UA or RESTful API enables automated sample metadata association and QC workflow synchronization.
Applications
- Chorus 1: Trace elemental analysis (ICP-MS, GFAAS), molecular biology (qPCR, sequencing, transfection), monoclonal antibody production, stem cell expansion, and pharmaceutical stability testing
- Chorus 2: HPLC mobile phase preparation, ELISA/RIA assay buffers, electrophysiology saline solutions, AAS standard dilutions, and histology reagent formulation
- Chorus 3: Autoclave chamber fill, steam generator feed, hydroponic nutrient mixing, environmental chamber humidification, and general lab cleaning protocols
- Cross-tier compatibility enables hybrid workflows—e.g., Chorus 3 feeds Chorus 1’s RO pre-treatment stage; Chorus 2 supplies rinse water to Chorus 1’s recirculation loop—maximizing resource efficiency while preserving grade integrity
FAQ
How does PureSure® ensure uninterrupted ultra-pure water supply during column exhaustion?
PureSure® employs two parallel ion exchange columns with independent conductivity sensors. When the primary column approaches saturation, the system automatically switches flow to the secondary column while initiating regeneration—eliminating downtime and preventing resistivity drift beyond 18.2 MΩ·cm.
Can TOC monitoring be validated per USP ?
Yes—the integrated TOC analyzer uses certified 185/254 nm UV oxidation and calibrated NDIR detection, with daily system suitability tests (SST) and documented linearity verification across 1–1000 ppb range per USP requirements.
Is microbial control maintained during extended storage in the tank?
The reservoir incorporates continuous low-dose UV-C irradiation (254 nm), periodic thermal sanitization cycles (≥80 °C), and sterile vent filtration—validated to maintain <0.001 EU/mL endotoxin levels for ≥72 hours post-generation.
What documentation is provided for regulatory audits?
Each system ships with Factory Acceptance Test (FAT) reports, IQ/OQ protocols aligned with ISO 13485 and GAMP 5, and a complete validation support package including risk assessments (FMEA), calibration certificates, and material compatibility data for all wetted components.
How is cross-contamination prevented between different water grades?
Physical isolation of fluid paths, dedicated pumps and sensors per grade, and strict pressure differentials prevent backflow; interlocks disable dispensing if upstream purity thresholds are breached—verified during Installation Qualification.