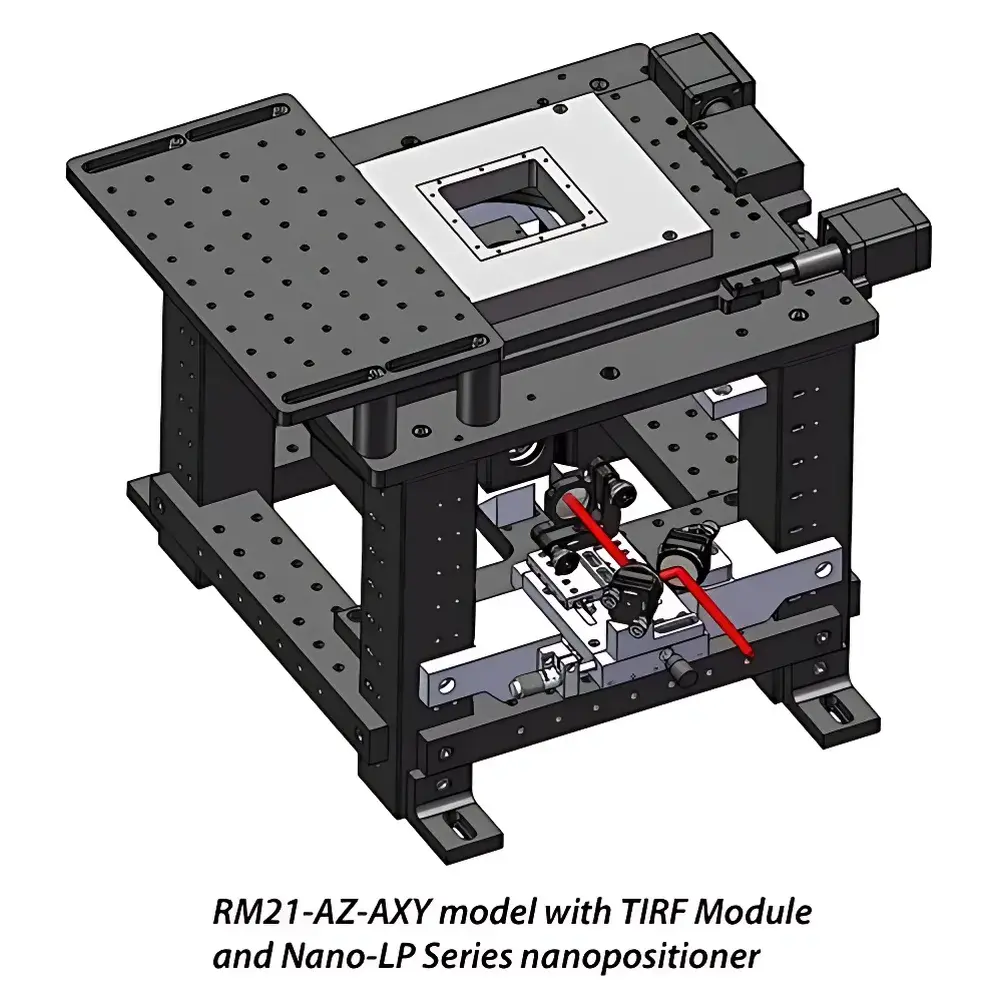
Mad City Labs TIRF Module for RM21™ Platform
| Brand | Mad City Labs |
|---|---|
| Origin | USA |
| Model | TIRF Module |
| Microscope Type | Inverted Fluorescence Microscope |
| Excitation Source | Metal Halide Lamp |
| Eyepiece | Standard Eyepiece |
| Objective | Standard Objective |
| Fluorescence Configuration | Epi-Fluorescence |
| Compatible Light Sources | Universal (Any Illumination Source) |
| Observation Head | Inverted |
| Illumination Modes | Epi-Fluorescence, Total Internal Reflection Fluorescence (TIRF), Highly Inclined and Laminated Optical (HILO) |
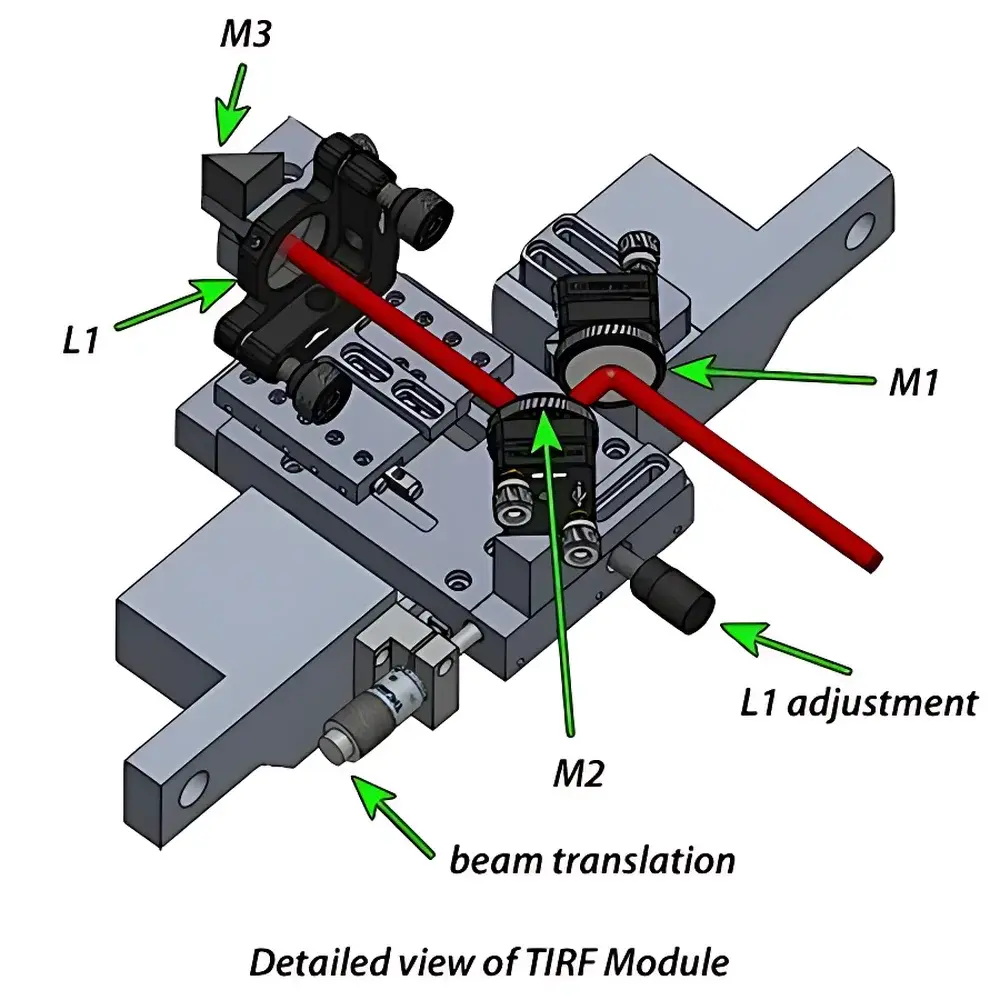
| Beam Steering Precision | Micrometer-Driven Mirror Translation (M2), Axial Lens Adjustment (L1), Fixed-Angle Steering Mirror (M1) |
| Output Coupling | TIRF Lock™ Interface for Z-Axis Feedback Stabilization |
| Compatibility | RM21™ and MicroMirror™ TIRF Platforms |
| Mounting | Modular, Tool-Free Alignment Design |
| Optical Path Control | Full Continuum from 0° (Epi) to >Critical Angle (TIR) |
Overview
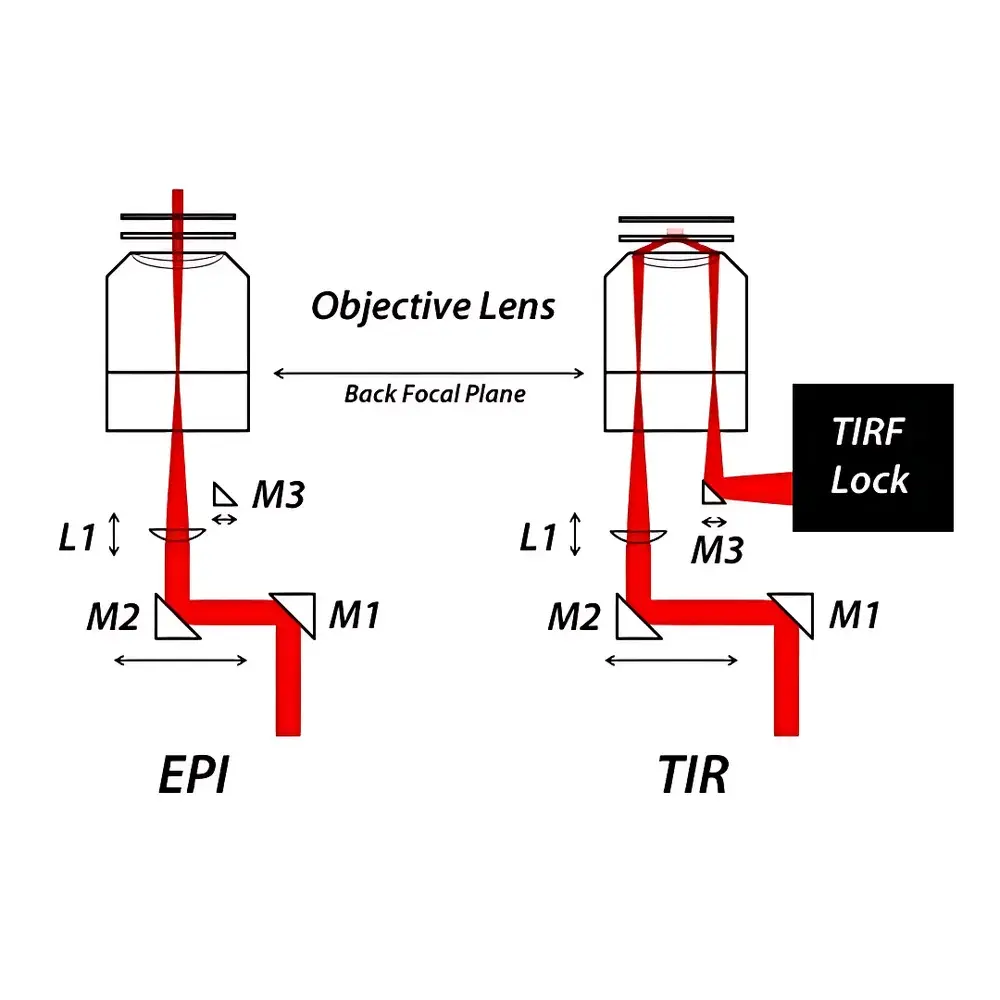
The Mad City Labs TIRF Module is a precision-engineered optical add-on designed to convert the RM21™ inverted microscope platform into a multimodal fluorescence imaging system capable of seamless transition between epi-fluorescence, total internal reflection fluorescence (TIRF), and highly inclined and laminated optical (HILO) illumination. Based on the physical principle of evanescent field excitation—where incident light undergoes total internal reflection at the glass–water interface, generating an exponentially decaying evanescent wave penetrating ~60–200 nm into the sample—the module enables selective excitation of fluorophores in the immediate vicinity of the coverslip. This provides exceptional axial resolution, dramatically reduced background signal, and superior signal-to-noise ratio (SNR) for single-molecule localization, live-cell membrane dynamics, and sub-diffraction-limited vesicle trafficking studies. Unlike fixed-angle TIRF solutions, this module implements continuous, real-time angular control over the excitation beam’s incidence at the objective’s back focal plane (BFP), allowing empirical optimization of illumination geometry per experimental condition.
Key Features
- Full-spectrum illumination angle control—from standard epi-illumination (0° relative to optical axis) through HILO (near-critical angle) to full TIRF (>critical angle)—via micrometer-actuated beam steering optics.
- Three-stage optical alignment architecture: M1 (fixed angular mirror), M2 (translatable mirror with micrometer-driven lateral displacement), and L1 (axially adjustable focusing lens) enabling precise positioning of the excitation spot on the objective BFP.
- Modular, tool-free integration with RM21™ and MicroMirror™ platforms; no permanent modification to host microscope required.
- Dedicated output port (M3 mirror) for direct coupling to the TIRF Lock™ feedback module, which monitors TIR beam displacement and dynamically corrects Z-position via closed-loop nanometer-scale stage control.
- Universal light source compatibility—designed to accept collimated input from metal halide lamps, LEDs, or laser combiners without optical recalibration.
- Optimized mechanical stability: kinematic mirror mounts, low-drift thermal design, and vibration-damped housing ensure long-term alignment retention during time-lapse and multi-hour acquisition protocols.
Sample Compatibility & Compliance
The TIRF Module supports standard No. 1.5H coverglass (170 µm thickness) mounted specimens—including adherent mammalian cells, reconstituted lipid bilayers, supported membranes, and immobilized protein complexes. Its angular flexibility accommodates variations in refractive index (e.g., aqueous buffers, glycerol-based mounting media) by permitting manual or software-assisted critical angle recalibration. The system conforms to ISO 19012-1:2020 (microscope optical performance standards) and supports GLP-compliant documentation when integrated with TIRF Lock™’s audit-trail-enabled controller firmware. While not FDA-cleared as a diagnostic device, the module meets essential requirements for research-grade instrumentation under IEC 61000-6-3 (EMC emission standards) and IEC 61000-6-2 (immunity testing).
Software & Data Management
Operation is fully compatible with Mad City Labs’ MicroManager-compatible drivers and third-party acquisition platforms including Nikon NIS-Elements, Zeiss ZEN, and open-source Python-based control (via PySerial and custom API wrappers). All angular adjustments, mirror positions, and L1 focus offsets are programmable and loggable with timestamped metadata. When paired with TIRF Lock™, the system records Z-correction history, beam displacement metrics, and SNR trends per frame—enabling retrospective quality assessment and compliance-ready reporting. Data export supports TIFF (with embedded EXIF tags), HDF5 (for multi-dimensional time-series), and CSV (for quantitative intensity and drift analysis), all compliant with FAIR data principles.
Applications
- Single-molecule tracking of receptor diffusion and clustering at plasma membranes.
- Real-time visualization of clathrin-coated pit assembly/disassembly kinetics.
- High-contrast imaging of synaptic vesicle docking and fusion events in primary neurons.
- Quantitative analysis of actin polymerization dynamics at leading edges using LifeAct-GFP.
- HILO-enabled volumetric sectioning in thick spheroids or organoid slices where conventional TIRF penetration depth is insufficient.
- Correlative TIRF–PALM experiments requiring identical sample positioning across modalities.
FAQ
Is the TIRF Module compatible with objectives other than those supplied with the RM21™ platform?
Yes—provided the objective has a back focal plane accessible via the designated C-mount port and a numerical aperture ≥1.45 (e.g., Nikon CFI Apo TIRF 60x/1.49, Olympus UPLSAPO 100xO). Custom adapter rings are available upon request.
Can the module be used for quantitative FRET or FLIM measurements?
While not optimized for time-resolved detection, the module preserves excitation polarization and beam uniformity necessary for ratiometric FRET. FLIM requires external TCSPC hardware and is supported via synchronized TTL triggering.
Does the system support automated multi-position TIRF acquisition?
Yes—through integration with Mad City Labs’ NanoDrive™ XYZ stages and MicroManager’s multi-dimensional acquisition engine, enabling unattended grid-based TIRF imaging across 96-well plates or tissue sections.
What maintenance is required for long-term angular calibration stability?
No routine recalibration is needed. The kinematic mounts and hardened aluminum housing maintain alignment within ±0.05° over 12 months under laboratory conditions (20–25°C, <60% RH). Annual verification using a calibrated autocollimator is recommended for GLP environments.