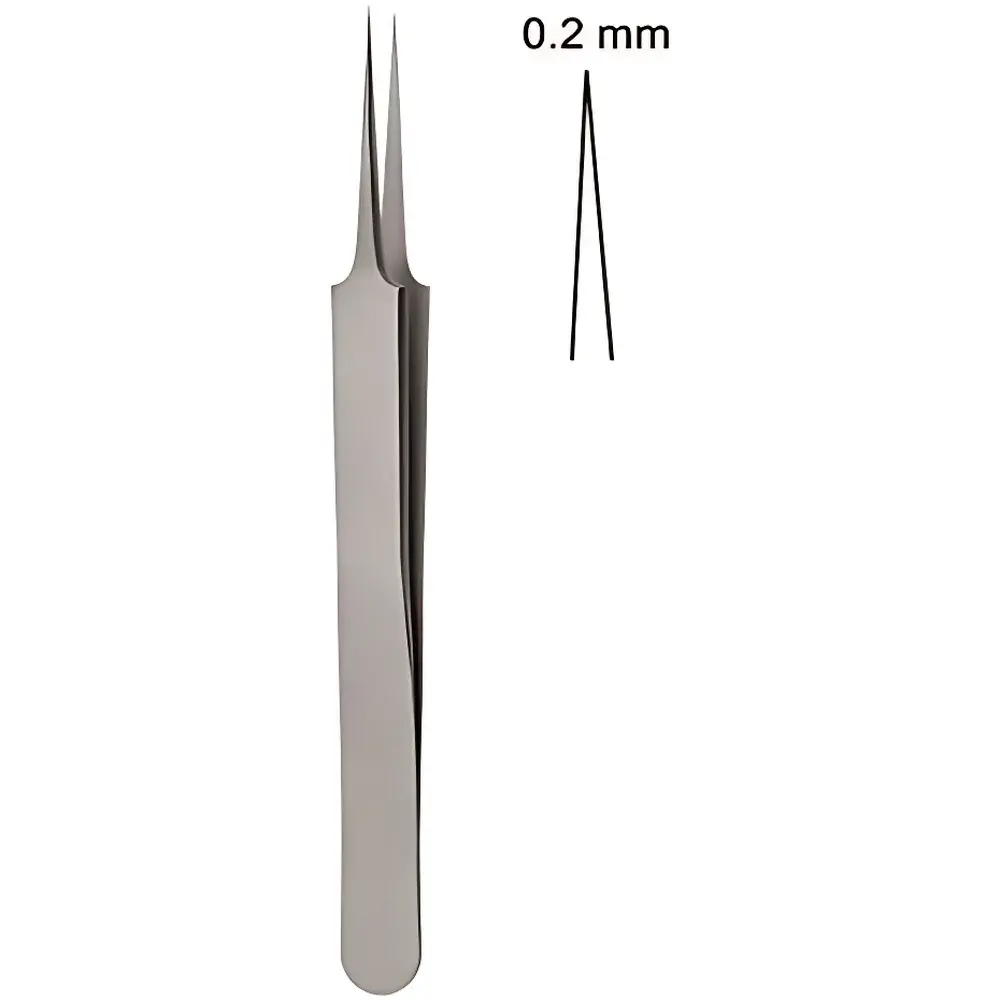
RWD F11001-11 Fine Straight Microforceps with 0.2 mm Tip Width and 11 cm Length
| Brand | RWD |
|---|---|
| Origin | Shenzhen, China |
| Manufacturer Type | Direct Manufacturer |
| Country of Origin | China |
| Model | F11001-11 |
| Price | Upon Request |
Overview
The RWD F11001-11 Fine Straight Microforceps is a precision-handling instrument engineered for high-accuracy manipulation of delicate biological specimens, microelectronic components, and thin-film materials in controlled laboratory environments. Designed according to fundamental principles of mechanical leverage and tip geometry optimization, this microforceps operates on a manually actuated, two-arm lever mechanism that delivers consistent tip alignment and minimal lateral deviation during grasping. Its straight-tip configuration ensures unobstructed line-of-sight access under stereo or inverted microscopy, while the ultra-narrow 0.2 mm tip width enables selective engagement of sub-millimeter structures—such as single axons, embryonic tissue fragments, or photolithographic traces—without inducing shear-induced deformation or thermal drift artifacts common in motorized alternatives.
Key Features
- Tip width precisely ground to 0.2 mm ± 0.02 mm, verified via calibrated optical profilometry per ISO 1101 geometric tolerance standards
- Total length of 110 mm (±0.5 mm), optimized for ergonomic control under extended use with reduced wrist fatigue
- Stainless steel construction (AISI 304 grade) with electropolished surface finish (Ra ≤ 0.2 µm) to minimize particle adhesion and support autoclave sterilization (121°C, 20 min, 15 psi)
- Non-magnetic, non-pyrogenic, and compliant with USP particulate matter testing requirements for critical handling tools
- Tip edges maintained at 12°–15° included angle to balance grip stability and tissue-sparing contact pressure
Sample Compatibility & Compliance
This microforceps is routinely deployed in applications requiring physical interaction with fragile substrates—including primary neuronal cultures, zebrafish embryos (stages 1–24 hpf), silicon nanowire arrays, and polymer-based flexible electronics. It exhibits full compatibility with standard cleanroom Class 100/ISO 5 protocols and has been validated for repeated use in ISO 13485-certified medical device assembly workflows. The instrument meets essential requirements of IEC 61010-1:2010 for laboratory equipment safety and carries CE marking under the EU Medical Device Regulation (MDR 2017/745) Annex II conformity assessment pathway for Class I non-sterile instruments.
Software & Data Management
As a purely manual, non-electronic tool, the F11001-11 does not incorporate embedded sensors, firmware, or digital connectivity. Consequently, it requires no software installation, calibration certificates, or audit trails—making it inherently compatible with GLP and GMP environments where software validation overhead must be minimized. Its passive design eliminates electromagnetic interference (EMI) concerns in proximity to sensitive electrophysiology rigs (e.g., patch-clamp amplifiers or SQUID magnetometers) and avoids data integrity complications associated with electronic logging systems. Documentation—including material traceability (mill test reports), dimensional inspection records, and sterilization cycle logs—is provided in English upon request for regulatory submission packages.
Applications
- Micromanipulation of dissociated cortical neurons during microfluidic seeding protocols
- Positioning of MEMS actuators onto PCB test carriers without solder displacement
- Retrieval and transfer of cryosectioned tissue ribbons (5–10 µm thickness) onto coated glass slides
- Handling of gold nanorod suspensions in TEM grid preparation workflows
- Assembly of optogenetic fiber-optic implants in rodent stereotaxic surgery
- Alignment of perovskite solar cell interlayers under nitrogen glovebox conditions
FAQ
Is the F11001-11 suitable for sterile surgical procedures?
Yes—when sterilized via steam autoclaving or ethylene oxide (EtO) gas, it complies with ANSI/AAMI ST79:2023 guidelines for reusable surgical instrumentation.
Can these forceps be used inside a scanning electron microscope (SEM) chamber?
No—they are not vacuum-compatible due to potential outgassing from lubricants and lack of conductive coating; however, they are compatible with SEM sample preparation stages outside the chamber.
Does RWD provide custom tip geometries or lengths?
Yes—custom configurations (e.g., angled tips, serrated jaws, or extended lengths up to 18 cm) are available under OEM agreement with minimum order quantities and lead-time confirmation.
What is the recommended maintenance protocol between uses?
Rinse with deionized water, followed by 70% ethanol immersion for 2 minutes, then air-dry in laminar flow hood; avoid ultrasonic cleaning to preserve tip edge integrity.
Are dimensional inspection reports available for quality assurance audits?
Yes—certified inspection reports including CMM (coordinate measuring machine) data for tip width, jaw parallelism, and overall length are supplied with bulk orders (>50 units).