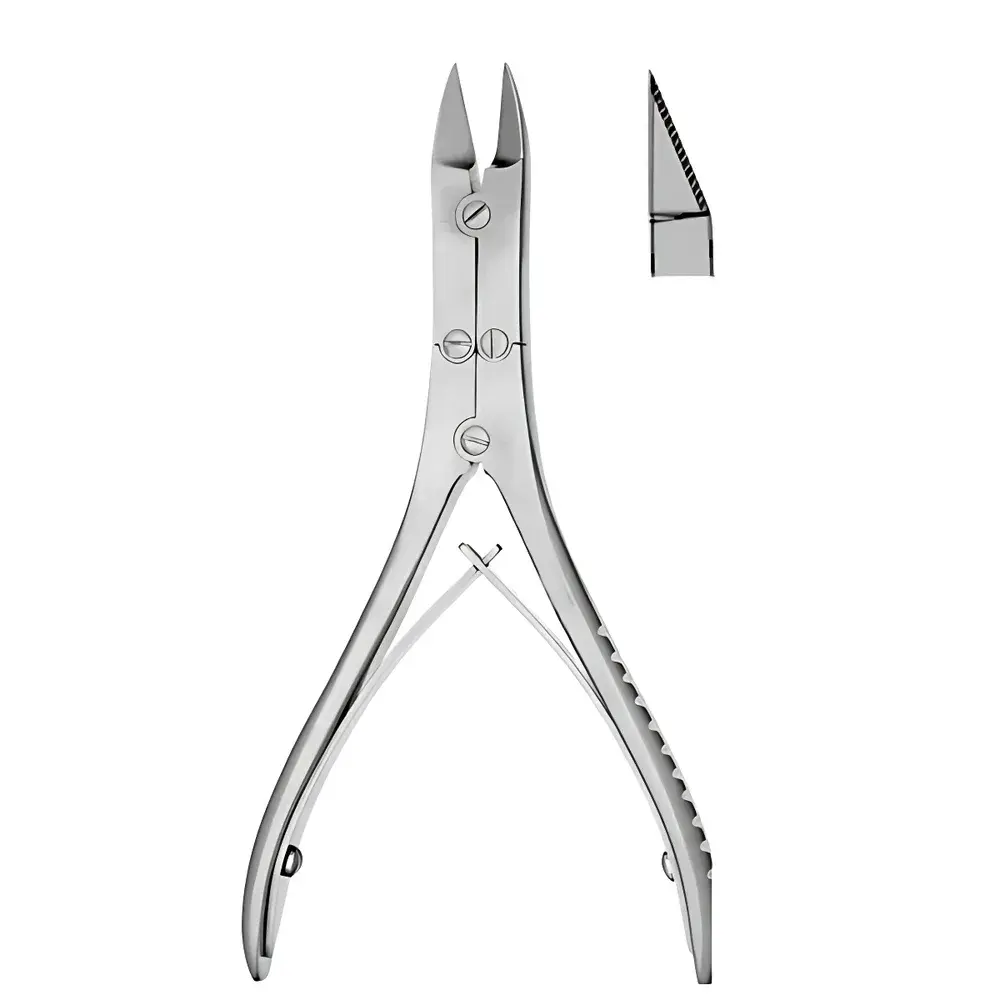
BOHLER S22003-14 Double-Jointed Bone Rongeur
| Brand | RWD |
|---|---|
| Origin | Shenzhen, China |
| Manufacturer Type | Direct Manufacturer |
| Country of Origin | China |
| Model | S22003-14 |
| Price | USD 110 (FOB Shenzhen) |
Overview
The BOHLER S22003-14 Double-Jointed Bone Rongeur is a precision surgical instrument engineered for controlled bone and calcified tissue removal in preclinical research and veterinary procedures. Designed with a true double-joint mechanical linkage, this rongeur delivers enhanced leverage and improved tactile feedback compared to single-hinge alternatives—critical for delicate osteotomies, craniotomy edge trimming, and spinal laminectomy preparation in rodent, rabbit, and small canine models. Its 14.5 cm overall length balances maneuverability with ergonomic control, while the flat-blade configuration (non-toothed, straight-cutting edges) ensures clean, non-crushing excision of cortical bone fragments without microfracture propagation. The instrument operates on classical lever-arm biomechanics, where force amplification is achieved through dual pivot points, enabling high cutting efficiency at low hand pressure—a key requirement for reproducible surgical standardization in GLP-compliant animal studies.
Key Features
- Double-joint articulation system for optimized mechanical advantage and reduced operator fatigue during prolonged use
- Flat, non-serrated cutting blades (0.8 mm blade thickness) fabricated from ISO 7153-1 compliant stainless steel (AISI 420 equivalent), hardened to 54–56 HRC for sustained edge retention
- Ergonomic, satin-finished stainless steel handles with laser-etched model number (S22003-14) and RWD manufacturer mark for traceability
- Autoclavable up to 134°C (273°F) for 18 minutes per EN 285; validated for ≥500 sterilization cycles without structural degradation
- Complies with ISO 7153-2:2016 (Surgical instruments — Materials — Part 2: Stainless steels) and ISO 80369-3:2016 (Small-bore connectors for liquids and gases in healthcare applications — Part 3: Connectors for enteral applications)
Sample Compatibility & Compliance
This rongeur is intended for ex vivo and in vivo use in accredited academic, contract research organization (CRO), and veterinary clinical settings. It is routinely employed in IACUC- and AAALAC-approved protocols involving murine calvarial defect models, rat spinal cord injury laminectomies, and porcine cranial burr hole preparations. The device carries no CE marking for human clinical use and is supplied strictly as a Research Use Only (RUO) instrument under FDA 21 CFR §809.10. All materials meet USP Class VI biocompatibility requirements for short-term tissue contact (≤24 h). Device labeling conforms to ISO 15223-1:2021 for symbols used in medical device labeling.
Software & Data Management
As a manually operated mechanical instrument, the S22003-14 requires no embedded electronics, firmware, or software integration. However, it supports full digital traceability within laboratory information management systems (LIMS) and electronic lab notebooks (ELN) via its permanent laser-engraved serial identifier. Institutions adhering to 21 CFR Part 11 may document usage logs—including sterilization batch records, maintenance history, and procedure-specific calibration verification (e.g., blade gap measurement using optical comparator)—within validated audit-trail-enabled platforms. RWD provides a Certificate of Conformance (CoC) with each unit, including material certification (MTR), dimensional inspection report, and pass/fail results for hardness and corrosion resistance testing per ASTM A967.
Applications
- Preclinical neurosurgery: Precise removal of parietal bone flaps in mouse/rat stereotaxic surgery
- Orthopedic implant studies: Trimming of cortical bone margins around intramedullary nail insertion sites
- Dental and maxillofacial research: Resection of alveolar ridge segments in periodontal defect models
- Tissue engineering scaffold implantation: Contouring of bony beds prior to hydrogel or ceramic scaffold placement
- Veterinary surgical training: Standardized simulation of orthopedic instrumentation techniques under supervision
FAQ
Is the S22003-14 suitable for human surgical applications?
No. This instrument is designated Research Use Only (RUO) and is not certified for human clinical use under FDA, MDR, or PMDA regulatory pathways.
What is the recommended maintenance protocol between uses?
After each procedure, rinse thoroughly with deionized water, ultrasonically clean for 10 minutes in enzymatic solution (pH 7.0–8.5), inspect blade alignment under 10× magnification, and lubricate pivot joints with medical-grade mineral oil prior to autoclaving.
Does RWD offer re-sharpening services for this rongeur?
Yes. RWD provides factory-certified reconditioning—including blade resharpening, joint realignment, and hardness verification—at authorized service centers in Shanghai and Singapore. Turnaround time is 10 business days from receipt.
Can this instrument be integrated into automated surgical robotics platforms?
Not natively. The S22003-14 lacks actuation interfaces or position feedback sensors. Integration would require third-party robotic end-effector adaptation and independent validation per ISO 13482 and IEC 62304.
What documentation accompanies each unit?
Each shipment includes a Certificate of Conformance, Material Test Report (MTR), dimensional inspection report, and IFU (Instructions for Use) in English and EU-compliant multilingual format (EN/DE/FR/ES).