FluorCam Desktop Multispectral Fluorescence Imaging System by PSI
| Brand | PSI (Photosynthesis Systems Instruments) |
|---|---|
| Origin | Czech Republic |
| Model | FluorCam Desktop Multispectral Fluorescence Imaging System |
| Imaging Area | 13 × 13 cm |
| CCD Frame Rate | Up to 50 fps |
| OJIP Kinetics Acquisition | Yes (optional module) |
| QA Reoxidation Kinetics | Yes (STF pulse ≤100 µs, up to 120,000 µmol·m⁻²·s⁻¹) |
| Protocol Flexibility | Fully programmable, multi-step fluorescence protocols with time-stamped automated cycles |
| Dual-Color Actinic Light Sources | Standard red + white |
| Optional Modules | TetraCam RGB imaging (20 × 25 cm), hyperspectral imaging unit, infrared thermography unit, GFP imaging capability |
Overview
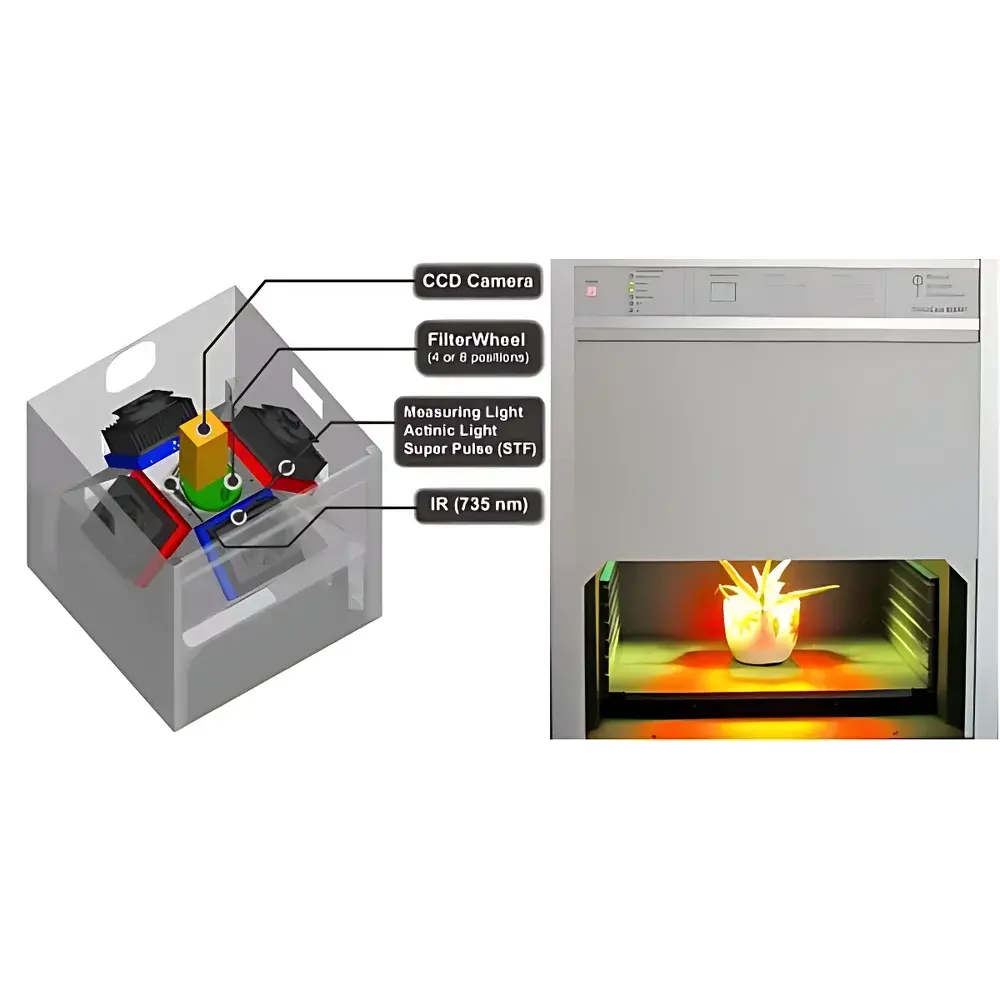
The FluorCam Desktop Multispectral Fluorescence Imaging System is a research-grade, benchtop-integrated platform engineered for quantitative, spatially resolved analysis of chlorophyll a fluorescence and multispectral emission in living plant tissues. Developed by Photosynthesis Systems Instruments (PSI), Czech Republic — the pioneer and globally recognized leader in fluorescence imaging since the first commercial implementation of PAM (Pulse-Amplitude Modulation) fluorescence imaging in 1996 — this system combines high-sensitivity CCD detection with precisely controlled, spectrally defined LED excitation arrays within a light-tight dark-adaptation chamber. Its core measurement principle is based on non-invasive, real-time monitoring of photosynthetic electron transport efficiency via kinetic fluorescence induction curves (e.g., Kautsky effect), OJIP transient analysis, and QA⁻ reoxidation dynamics following single-turnover saturating flashes. The system delivers pixel-level quantification of photochemical yield (ΦPO = Fv/Fm), performance index (PIABS, PICS), energy dissipation parameters (ΦDO, ΨO), and reaction center density (RC/CS), enabling rigorous physiological interpretation grounded in biophysical models of Photosystem II function.
Key Features
- Integrated dark-adaptation chamber with 7-position adjustable sample stage, ensuring consistent dark acclimation prior to measurement
- High-quantum-efficiency, cooled CCD sensor with temporal resolution up to 50 frames per second, optimized for low-light fluorescence capture
- Four fixed, uniform LED illumination panels (standard configuration: red + white actinic light; optional red + blue dual-band) delivering stable, homogeneous irradiance across the full 13 × 13 cm imaging field
- Capable of acquiring OJIP transient kinetics with microsecond-scale temporal sampling (via optional high-speed acquisition module), yielding >20 biophysically derived parameters including Mo, Sm, Fix Area, and PIABS
- Supports single-turnover flash (STF) protocols for QA⁻ reoxidation kinetics analysis, with flash duration ≤100 µs and peak irradiance up to 120,000 µmol photons·m⁻²·s⁻¹
- Fully programmable protocol engine allowing user-defined sequences: Fv/Fm, NPQ (two independent quenching protocols), LC light-response curves, NDVI mapping, PAR absorption imaging, and optional GFP or multispectral fluorescence modes
- Automated time-series operation with scheduled protocol switching (e.g., diurnal Fv/Fm during day, NPQ at night), full timestamping, and auto-saving in standardized HDF5 or CSV formats
Sample Compatibility & Compliance
The FluorCam Desktop system accommodates a broad range of biological specimens without destructive preparation: intact leaves, fruits, seedlings, whole rosettes, clonal mosses/lichens, and algal suspensions or biofilms on solid supports. It complies with standard laboratory biosafety and electrical safety requirements (IEC 61010-1). Data acquisition and processing workflows support GLP/GMP-aligned documentation practices, including audit-trail-enabled software logging, user-access controls, and exportable metadata compliant with MIAPPE (Minimum Information about a Plant Phenotyping Experiment) standards. While not FDA 21 CFR Part 11-certified out-of-the-box, the FluorCam software architecture permits integration into validated environments through configurable electronic signatures and secure data archiving protocols.
Software & Data Management
FluorCam software (v8.x or later) provides a unified interface for hardware control, real-time visualization, parameter calculation, and batch analysis. All raw intensity time series are stored with embedded experimental metadata (light intensities, timing, temperature, user ID). Built-in algorithms compute standardized fluorescence parameters according to established conventions (Strasser et al., 2004; Schansker et al., 2005) and allow region-of-interest (ROI) masking for heterogeneous samples. Export options include TIFF stacks (16-bit), MATLAB .mat files, and tabular CSV with hierarchical column labeling. The software supports third-party integration via COM/ActiveX interfaces and Python API bindings for custom pipeline development in Jupyter or R environments.
Applications
This system serves as a foundational tool in plant phenomics, stress physiology, and functional genomics. It is routinely deployed in studies of abiotic stress responses (drought, salinity, heavy metal toxicity, UV-B exposure), biotic interaction screening (pathogen susceptibility, symbiont efficacy), breeding program trait validation (photosynthetic efficiency, photoprotective capacity), and high-throughput screening of mutant libraries. Its multispectral capability enables concurrent assessment of chlorophyll content (NDVI), anthocyanin distribution, and GFP-tagged reporter expression — facilitating correlative analysis across structural, biochemical, and functional phenotypic dimensions. Published applications span peer-reviewed journals including Plant Physiology, Journal of Experimental Botany, and Remote Sensing of Environment.
FAQ
What is the minimum detectable fluorescence change (dynamic range)?
The system achieves a signal-to-noise ratio >1,000:1 under standard dark-adapted conditions, enabling robust detection of sub-1% fluorescence yield differences across pixels.
Can the system be used for field-deployable measurements?
While designed as a desktop platform, its modular construction and battery-compatible power supply enable portable deployment in greenhouse or mobile lab settings; however, ambient light exclusion remains essential for quantitative fluorescence acquisition.
Is calibration traceable to NIST or other metrological standards?
Absolute radiometric calibration is performed using certified reference LEDs and calibrated photodiodes; spectral responsivity is characterized per ISO 17025-accredited procedures by PSI’s internal metrology lab.
How does the system handle sample movement or uneven surfaces during imaging?
The 7-level height-adjustable stage ensures optimal focus plane alignment; optional motorized Z-stage and autofocus routines (available in advanced configurations) further mitigate positional variability.
Are raw fluorescence kinetics data accessible for custom modeling?
Yes — all time-resolved intensity traces (F(t)) are exported in open, lossless formats (HDF5, ASCII) with precise timestamping and stimulus synchronization markers, suitable for kinetic modeling in COPASI, Berkeley Madonna, or custom MATLAB scripts.