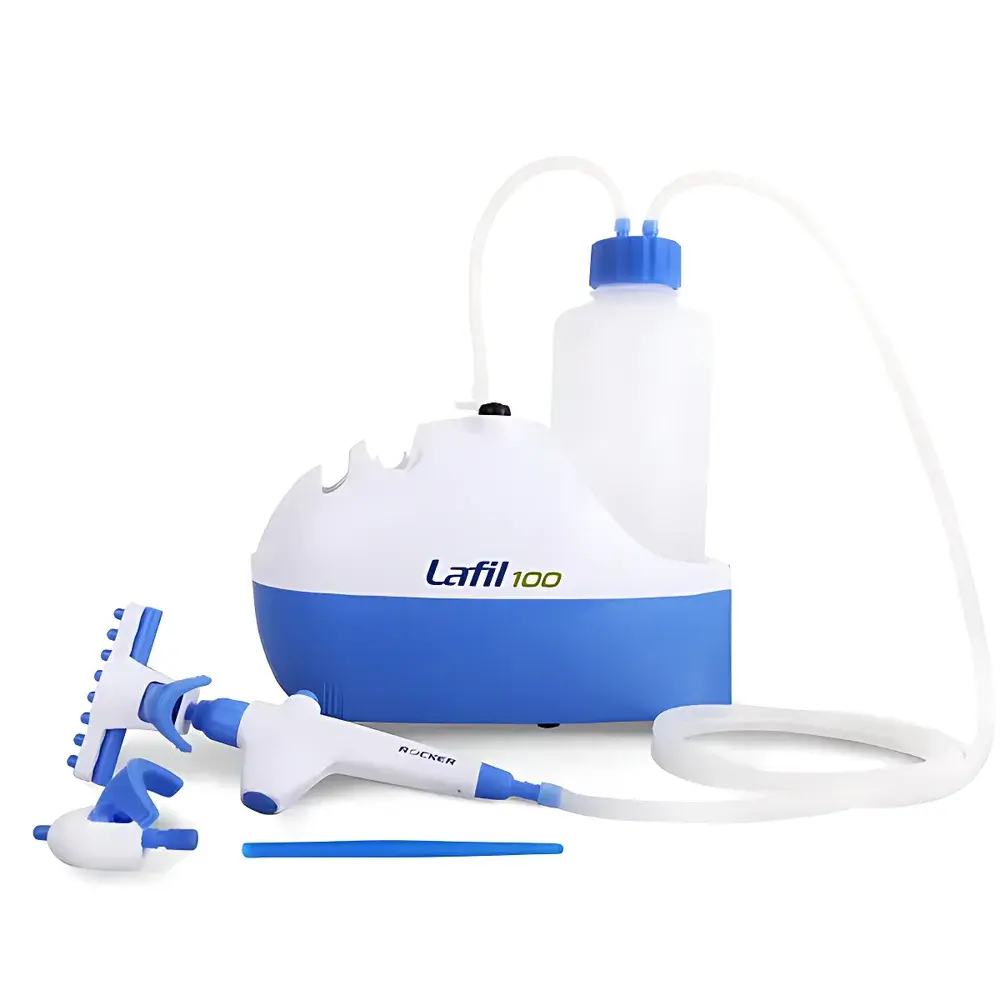
ROCKER Lafil100 Vacuum Liquid Aspiration System
| Brand | ROCKER |
|---|---|
| Origin | Taiwan |
| Model | Lafil100 |
| Ultimate Vacuum | 347 mbar |
| Maximum Flow Rate | 1000 mL/min |
| Dimensions | 265 × 130 × 210 mm |
| Motor | Brushless DC |
| Input Voltage | AC 100–240 V, 50/60 Hz (converted to DC 12 V) |
| Weight | 1.3 kg |
| Housing Material | ABS |
| Handheld Controller & Tip Ejectors | POM |
| Serum Bottle Cap & 1000 mL Waste Bottle | PP |
| Sterilization | Autoclavable accessories (121 °C, 20 min) |
Overview
The ROCKER Lafil100 Vacuum Liquid Aspiration System is an integrated, benchtop-grade vacuum pump platform engineered for precise, contamination-controlled liquid handling in cell culture, molecular biology, and sterile laboratory workflows. Unlike conventional vacuum manifolds or standalone pumps requiring external controllers, the Lafil100 integrates a brushless DC vacuum pump, ergonomic handheld controller, and modular aspiration accessories into a single compact unit measuring only 265 × 130 × 210 mm — enabling direct placement inside laminar flow hoods and biosafety cabinets without compromising workspace integrity. Its operating principle relies on controlled negative pressure generation (up to −347 mbar absolute vacuum) to drive aspiration through disposable or reusable filtration assemblies, supporting both continuous vacuum filtration and discrete sample withdrawal tasks with high reproducibility and minimal aerosol generation.
Key Features
- Compact footprint (265 × 130 × 210 mm) optimized for integration into Class II biosafety cabinets and laminar airflow workstations
- Dual-function capability: seamless transition between vacuum-assisted liquid aspiration (e.g., spent media from culture dishes or microplates) and sterile filtration of buffers, sera, or conditioned media
- Ergonomic handheld controller with tactile Lock button for sustained aspiration during extended procedures — reduces operator fatigue and improves workflow consistency
- Single-hand tip ejection mechanism using dual-configurable POM tip ejectors: single-claw (for standard pipette tips) and octo-claw (for multi-tip racks), both autoclavable at 121 °C for 20 minutes
- Integrated centrifuge tube support stand compatible with 15 mL and 50 mL conical tubes, enabling direct gravity-assisted or vacuum-enhanced filtration using optional PES membrane funnels
- All fluid-path components — including serum bottle caps, waste bottles (1000 mL PP, optional 500 mL glass), and tip ejectors — constructed from chemically resistant, non-leaching polymers (POM, PP, ABS) compliant with ISO 10993 biocompatibility guidelines
- Brushless DC motor ensures quiet operation (<52 dB(A)), extended service life, and consistent vacuum output across variable load conditions
Sample Compatibility & Compliance
The Lafil100 accommodates a broad range of sample formats and container types, including Petri dishes, multiwell plates (6–96-well), Erlenmeyer flasks (up to 1 L), and centrifuge tubes (15–50 mL). It interfaces seamlessly with both disposable filtration systems (e.g., Autofil™ PES membrane funnels, 0.22 µm pore size) and reusable stainless-steel or polypropylene filter holders. All accessories meet USP Class VI and ISO 10993-5 cytotoxicity standards. The system complies with EU CE safety directives (2014/35/EU Low Voltage Directive and 2014/30/EU EMC Directive), and its electrical design conforms to IEC 61010-1 for laboratory equipment safety. While not intrinsically certified for GMP environments, the Lafil100 supports GLP-aligned documentation practices when used with audit-trail-capable lab notebooks and validated sterilization cycles.
Software & Data Management
The Lafil100 operates as a hardware-only platform with no embedded firmware, display, or digital interface — intentionally designed to eliminate software validation requirements in regulated environments. Vacuum output is mechanically stabilized via precision-machined diaphragm pump architecture, ensuring repeatable performance independent of firmware updates or calibration drift. Users maintain full traceability through manual log entries linked to batch numbers of sterilized accessories (e.g., lot-coded PES filters, autoclave cycle records). For laboratories implementing 21 CFR Part 11 compliance, the Lafil100 integrates naturally into electronic lab notebook (ELN) workflows where aspiration events, filter lots, and sterilization logs are recorded externally with user authentication and timestamping.
Applications
- Aspiration of supernatants post-centrifugation in cell culture workflows
- Sterile filtration of growth media, PBS, trypsin-EDTA, and other aqueous reagents using 0.22 µm PES membranes
- Vacuum-assisted washing of adherent cells in T-flasks or multiwell plates
- Removal of culture medium from transwell inserts or organoid culture plates
- Pre-filtration of crude lysates prior to chromatography or centrifugal concentration
- Small-volume buffer exchange in low-throughput protein purification protocols
FAQ
Can the Lafil100 be used inside a Class II biosafety cabinet without disrupting laminar airflow?
Yes — its compact dimensions and low acoustic emission (<52 dB(A)) allow stable operation within standard biosafety cabinets while maintaining ISO Class 5 air quality at the work surface.
Are all wetted parts autoclavable?
All accessories — including POM tip ejectors, PP serum bottle caps, and 1000 mL PP waste bottles — are rated for steam sterilization at 121 °C for 20 minutes. The main unit housing (ABS) is not autoclavable and must remain outside sterilization cycles.
What vacuum level is required for effective aspiration of viscous cell culture media?
A stable vacuum of −300 to −347 mbar provides optimal flow rates (up to 1000 mL/min) for standard DMEM/FBS formulations; higher viscosity samples (e.g., Matrigel™-supplemented media) may require reduced aspiration speed or pre-dilution.
Is the Lafil100 compatible with third-party filtration devices?
Yes — it accepts any standard GL45-threaded filtration flask or vacuum manifold with compatible hose barb connections (6.4 mm ID silicone tubing included).
Does the system include regulatory documentation for IQ/OQ validation?
ROCKER provides Factory Acceptance Test (FAT) reports and material declarations (RoHS, REACH) upon request; site-specific Installation and Operational Qualification protocols must be developed by the end-user per ISO/IEC 17025 or ASTM E2500 guidelines.