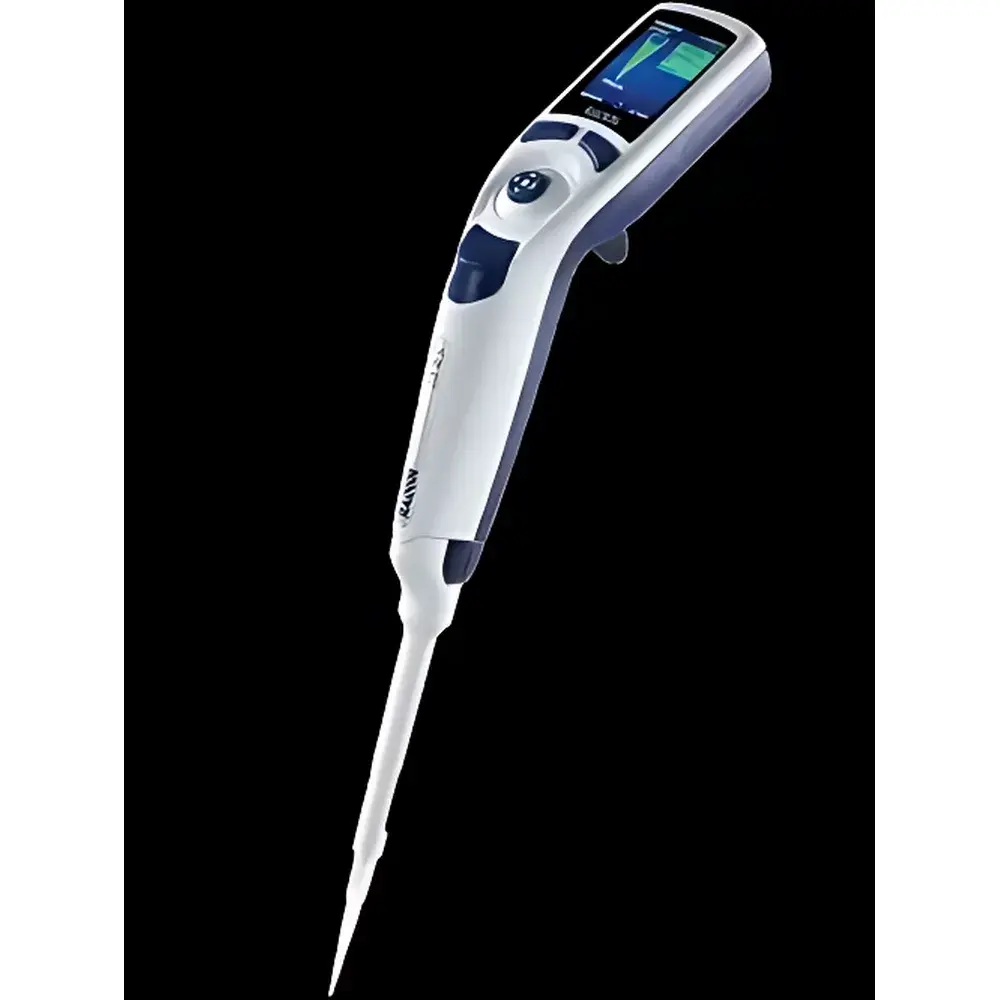
Rainin E4 XLS+ Single-Channel Electronic Pipette (Conical Tip)
| Brand | Rainin |
|---|---|
| Origin | Switzerland |
| Model | SE4-10XLS+ |
| Volume Range | 0.5–10 µL |
| Type | Single-Channel Electronic Pipette |
| Drive Mechanism | Stepper Motor (4,000-step resolution) |
| Display | Full-color graphical LCD |
| Control Interface | Joystick + rotary navigation dial |
| Calibration | Auto-zeroing at end of each pipetting cycle |
| GLP/GMP Compliance | Password-protected settings, tamper-proof service logs, audit trail-capable |
| Power | Rechargeable lithium-ion battery (≥1 day runtime, >1,000 charge cycles) |
| Ergonomics | Ambidextrous design, LTS (Low-Torque Seal) tip ejection system (ejection force ≤0.6 kg), integrated finger hooks, glove-compatible operation |
| Software Integration | Preloaded PureSpeed™ mode for biomolecule purification (proteins, oligonucleotides) |
| Onboard Memory | Multiple user-defined protocols stored locally |
Overview
The Rainin E4 XLS+ Single-Channel Electronic Pipette (Conical Tip) is an ISO 8655-compliant, microprocessor-controlled liquid handling instrument engineered for precision, repeatability, and regulatory traceability in regulated laboratory environments. Operating on the principle of positive displacement with closed-loop stepper motor actuation, the E4 XLS+ delivers volumetric accuracy across its full range (0.5–10 µL for the SE4-10XLS+ model) through real-time positional feedback and automatic zero-point recalibration after every dispense event. Its conical-tip architecture ensures compatibility with universal low-retention and filtered pipette tips—critical for viscous, volatile, or low-volume biological samples including DNA libraries, enzymatic reaction mixtures, and clinical assay reagents. Designed and manufactured in Switzerland, the device meets stringent mechanical stability requirements under IEC 61326-1 (EMC) and IEC 61010-1 (safety), supporting long-term performance consistency in QC, R&D, and GMP production labs.
Key Features
- High-Fidelity Motorized Actuation: 4,000-step high-resolution stepper motor enables sub-microliter positioning control, minimizing mechanical hysteresis and ensuring consistent piston travel velocity and distance per user-defined protocol.
- GLP/GMP-Ready Architecture: Password-protected configuration modes, encrypted service logs, and immutable cycle counters satisfy FDA 21 CFR Part 11 and EU Annex 11 requirements for electronic record integrity and operator accountability.
- Ergonomic Universal Design: Symmetrical body geometry, tactile finger hooks, and a 0.6 kg LTS tip ejection force reduce musculoskeletal strain during extended use—validated per ISO 13485 human factors guidelines for medical device usability.
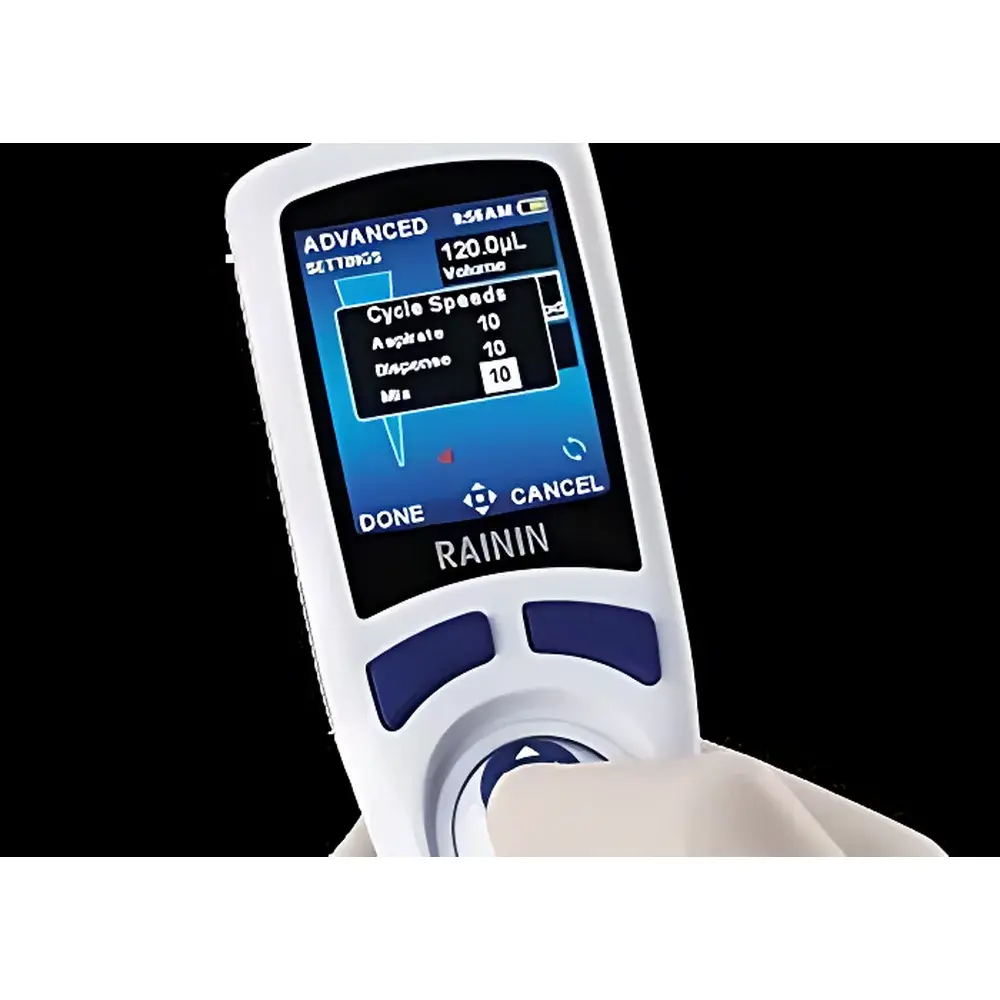
- Intuitive Graphical Interface: 2.8-inch color LCD with joystick-driven navigation supports rapid parameter adjustment, multi-level menu traversal, and visual confirmation of volume, mode, and battery status without requiring secondary software.
- PureSpeed™ Integrated Workflow: Pre-installed firmware module optimizes aspiration/dispense profiles for rapid, gentle elution of affinity-purified proteins and synthetic oligonucleotides—reducing shear stress and improving recovery yield in spin-column and magnetic bead-based workflows.
Sample Compatibility & Compliance
The E4 XLS+ accommodates standard conical-tip pipette consumables (including Rainin LTS, FilterMax, and third-party ISO-certified tips) across all volume ranges. Its sealed internal mechanism prevents aerosol ingress, maintaining calibration integrity when handling RNase-free, cytotoxic, or PCR-inhibitory samples. Device firmware complies with ISO/IEC 17025 documentation standards for metrological traceability; calibration certificates include uncertainty budgets derived from gravimetric verification per ISO 8655-6. All units ship with a factory-issued Certificate of Conformance (CoC) referencing EN ISO 9001:2015 quality management systems and Swiss manufacturing origin traceability.
Software & Data Management
No external PC or proprietary software is required for routine operation—the E4 XLS+ stores up to 12 user-defined protocols directly in non-volatile memory. Each protocol retains independent settings for volume, speed profile, mixing steps, reverse pipetting, and tip touch-off parameters. Audit trails—including date/time stamps, operator ID (via password login), calibration events, and error codes—are retained onboard and exportable via USB-C to CSV format for integration into LIMS or ELN platforms. Firmware updates are performed via secure signed packages verified through embedded cryptographic keys, ensuring integrity against unauthorized modification.
Applications
- Quantitative preparation of qPCR master mixes and NGS library normalization standards
- Serial dilution of monoclonal antibody candidates under GMP analytical method validation (ICH Q2(R2))
- High-throughput cell culture media supplementation with growth factors at picomolar concentrations
- Automated sample prep for LC-MS/MS bioanalysis requiring strict carryover control and inter-run reproducibility
- GLP-compliant environmental toxin screening where pipette usage logs must accompany final analytical reports
FAQ
Does the E4 XLS+ support ASTM E2758-compliant calibration verification?
Yes—the device includes built-in gravimetric test mode compatible with Class E2 analytical balances and certified reference weights, generating ISO 8655-6–aligned deviation reports.
Can multiple users maintain separate protocol libraries on one unit?
No—protocols are shared across all operator sessions, but individual access rights (e.g., edit vs. execute-only) can be enforced via hierarchical password levels.
Is LTS tip compatibility mandatory for optimal performance?
While universal tips function, Rainin LTS tips are required to achieve the specified 0.6 kg ejection force and ensure repeatable seal formation across >10,000 cycles.
How often does the E4 XLS+ require factory recalibration?
Annual verification is recommended; however, the auto-zeroing feature and onboard diagnostics extend calibration interval validity per ISO/IEC 17025 clause 7.7.2.
Does PureSpeed™ mode alter the fundamental ISO 8655 accuracy specifications?
No—PureSpeed™ modifies only timing and acceleration profiles; volumetric accuracy remains within ±0.8% CV at 10 µL, as validated per ISO 8655-2.