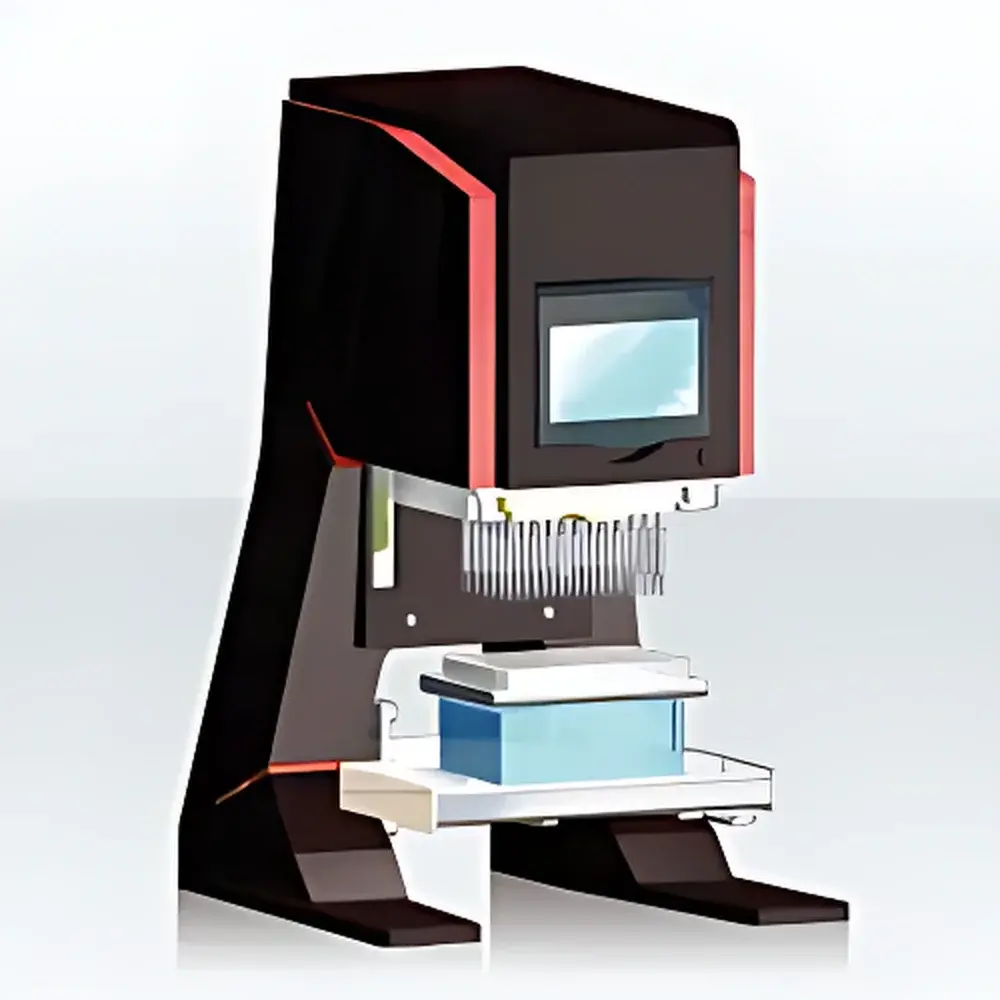
Analytik Jena SELMA 96 Portable Liquid Handling Workstation
| Brand | Analytik Jena |
|---|---|
| Origin | Germany |
| Manufacturer | Analytik Jena GmbH |
| Country of Origin | Imported |
| Model | SELMA 96 |
| Pricing | Upon Request |
Overview
The Analytik Jena SELMA 96 is a compact, benchtop liquid handling workstation engineered for precision, reproducibility, and operational flexibility in regulated and non-regulated laboratory environments. Based on positive-displacement and air-displacement pipetting principles—calibrated per ISO 8655 and validated against ASTM E1154 standards—the SELMA 96 delivers traceable volumetric accuracy across its full range of disposable tip configurations. Designed specifically for low-to-medium throughput workflows, it bridges the gap between manual multichannel pipettes and high-end robotic platforms. Its modular architecture supports both 96-well and 384-well microplate formats without mechanical reconfiguration, enabling seamless transitions between assay development, QC testing, and exploratory screening. The system operates independently of external computers or network infrastructure, making it ideal for biosafety cabinets (BSL-2/BSL-3), cleanrooms, field-deployable labs, and GLP-compliant quality control suites where instrument footprint, contamination control, and procedural autonomy are critical.
Key Features
- Compact footprint (307 × 480–520 × 325 mm) and lightweight design (15–20 kg) for placement inside biosafety cabinets or on crowded lab benches
- Dual-format compatibility: native support for 96-well and 384-well plates with no hardware modification required
- Four interchangeable 96-channel tip heads (0.5–25 µL, 0.5–60 µL, 0.5–250 µL, 0.5–1000 µL) and two 384-channel tip heads (0.5–25 µL, 0.5–60 µL), all compliant with ISO 8655-2 volumetric tolerance classes
- Integrated 3.5-inch color touchscreen with intuitive, fully localized interface—supports rapid protocol creation, editing, and recall without external software
- TipTray™ technology ensures consistent tip ejection force, hermetic sealing during aspiration, and reduced risk of aerosol generation
- Automated functions include column-wise and full-plate dispensing, multi-step dilution series, plate reformatting (96 ↔ 384), wall-touch dispensing, magnetic bead-based nucleic acid extraction, and programmable mixing cycles
- Onboard memory stores dozens of user-defined protocols with timestamped versioning and operator ID tagging—enabling audit-ready workflow continuity
Sample Compatibility & Compliance
The SELMA 96 handles aqueous buffers, viscous solutions (up to 50 cP), PCR master mixes, cell suspensions, serum-containing media, and low-volume organic solvent dilutions—provided within specified viscosity and vapor pressure limits for each tip head. It meets IEC 61000-6-2/6-3 electromagnetic compatibility requirements and complies with EU Machinery Directive 2006/42/EC and RoHS 2011/65/EU. For regulated applications, its deterministic execution model, non-volatile protocol storage, and absence of cloud dependency support adherence to FDA 21 CFR Part 11 (when paired with controlled access policies and electronic signature procedures), ISO/IEC 17025:2017 clause 7.7 (method validation), and CLSI EP15-A3 guidelines for precision assessment. Routine verification follows USP Analytical Instrument Qualification protocols.
Software & Data Management
Operation is managed exclusively via the embedded touchscreen UI—no PC, drivers, or third-party software required. Protocols are constructed using a hierarchical step editor supporting conditional logic (e.g., “if volume > 50 µL, activate pre-wet”), loop iterations, and inter-step delays. All executed runs generate immutable log files containing date/time stamps, tip head serial numbers, dispense volumes, error codes (e.g., “tip ejection failure”, “aspiration timeout”), and operator identifiers. Export is supported via USB 2.0 to CSV or PDF for integration into LIMS or ELN systems. Audit trails comply with ALCOA+ principles when deployed under documented SOPs governing data integrity, backup frequency, and retention periods.
Applications
- High-fidelity standard curve preparation for ELISA, qPCR, and immunoassays
- Cell-based assays including viability testing, cytotoxicity screening (e.g., IC50 determination), and transfection setup
- Nucleic acid workflows: plasmid miniprep, viral RNA extraction (including BSL-3-compatible setups), and NGS library normalization
- Small-molecule compound management: dose-response matrix generation, solubility profiling, and LC-MS sample dilution
- Agricultural residue analysis: QuEChERS extract dilution and internal standard spiking prior to GC-MS/MS injection
- Antimicrobial resistance profiling: broth microdilution MIC plate setup with antibiotic gradient generation
- Protein crystallization trials requiring nanoliter-scale reagent dispensing with wall-contact precision
FAQ
Does the SELMA 96 require routine calibration by a certified service technician?
No—users perform daily verification using gravimetric checks per ISO 8655-6; annual calibration is recommended but not mandatory unless mandated by internal QA policy or regulatory jurisdiction.
Can the SELMA 96 be integrated into a larger automated workflow?
Yes—it features TTL-level I/O ports for external trigger synchronization (e.g., plate presence detection, robotic arm handshake) and can serve as a dedicated module within hybrid automation cells.
Is method transfer between SELMA 96 and SELMA 384 straightforward?
Yes—protocol templates are format-agnostic; only tip head selection and well-coordinate mapping require adjustment; no reprogramming of core liquid handling logic is needed.
What maintenance is required beyond tip head replacement?
Monthly cleaning of the tip ejector mechanism and annual inspection of the pneumatic manifold seals are recommended; no lubrication or optical alignment is required.
Does the system support audit trail export compatible with 21 CFR Part 11?
Yes—when operated under controlled access conditions (password-protected user roles, electronic signatures enabled via external authentication), exported logs meet Part 11 record retention and attribution requirements.