Cytiva
Filter
Showing 61–86 of 86 results
| Brand | Cytiva |
|---|---|
| Origin | Switzerland |
| Instrument Model | Sefia S-2000 |
| Type | Fully Automated |
| Regulatory Designation | Import (CE-marked for IVDR/MDR-compliant use in cell therapy manufacturing) |
| Single-Use Kit Compatibility | CT-300.1, CT-400.1 + PB-100.1, CT-200.1, CT-800.1, CT-350.1S |
| Software Integration | Chronicle™ v3.2+ with eSOP/eBMR and 21 CFR Part 11 / EU Annex 11 compliance |
| Brand | Cytiva |
|---|---|
| Origin | Switzerland |
| Type | Fully Automated |
| Model | Sefia™ Automated Cell Expansion System |
| Compliance | CE-marked (2006/42/EC), IEC 61010, IEC 61326, ICES-001 Class A, China RoHS SJ/T11364-2014 |
| Software Compliance | Chronicle™ Viewer validated per GAMP™5 and FDA 21 CFR Part 11 |
| Operating Environment | 15–25°C, 20–80% RH (non-condensing), 840–1060 mbar, indoor use only, max. altitude 2000 m |
| Consumables | Single-use FEP or silicone-based disposable kits (e.g., Cat. No. 29716713, 29739346) |
| Integration | Compatible with Chronicle™ Viewer for audit-trail-enabled data management and real-time process monitoring |
| Brand | Cytiva |
|---|---|
| Origin | Switzerland |
| Manufacturer Type | Authorized Distributor |
| Origin Category | Imported |
| Model | Sepax C-Pro Cell Processor |
| Type | Fully Automated |
| Compliance | CE (2006/42/EC), IEC 61010, IEC 61326, IEC 62304 |
| Software Integration | Chronicle eSOP, GAMP 5-aligned, FDA 21 CFR Part 11-ready audit trail capability |
| Brand | Cytiva |
|---|---|
| Origin | Sweden |
| Model | STF-IRc |
| Tube OD Capacity | Up to 22.2 mm |
| Automation Level | Fully Automated |
| Sterility Assurance | Validated Aseptic Connection without Biosafety Cabinet |
| Application Domain | Bioprocessing Fluid Transfer |
| Brand | Cytiva |
|---|---|
| Origin | Sweden |
| Model | TE22 |
| Instrument Type | Wet Protein Electrophoretic Transfer System |
| Gel Capacity | Up to 4 × 9 cm × 10 cm gels per run |
| Cooling System | Integrated aluminum heat exchanger with recirculating buffer cooling |
| Temperature Rise | ≤5°C during standard transfer |
| Buffer Volume Requirement | 1 L per full-load run (4 gels) |
| Cassette Design | Open-mesh gel clamp for uniform pressure, gel/membrane alignment, and prevention of gel wrinkling or drying |
| Compliance | Designed for compatibility with standard SDS-PAGE and Western blotting workflows per ISO/IEC 17025-aligned laboratory practices |
| Brand | Cytiva |
|---|---|
| Origin | Sweden |
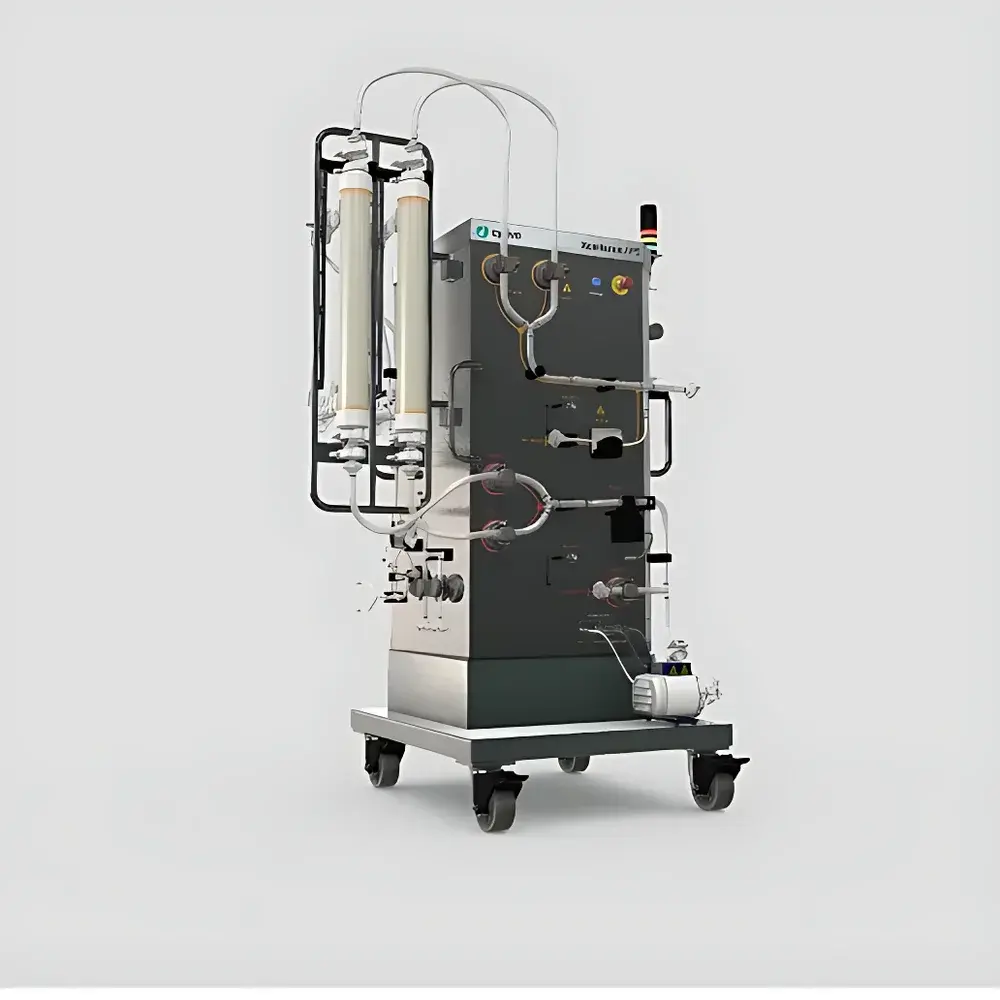
| Model | UniFlux Tangential Flow Filtration System |
| Flow Rate Options | 10, 60, 120 L/min (400 L/min available on custom basis) |
| Vessel Capacity Range | 5–600 L (stainless steel or single-use mixing systems supported) |
| Automation Platform | UNICORN Software (21 CFR Part 11 compliant) |
| Application Scope | Microfiltration (e.g., cell harvest) and Ultrafiltration (e.g., protein concentration & diafiltration) |
| Brand | Cytiva |
|---|---|
| Origin | Sweden |
| Model | UniFlux™ |
| System Variants | UniFlux™ 10, 30, 120, 400 |
| Membrane Area Range | 0.11–52 m² |
| Feed Volume Capacity | 1–10,000+ L |
| Construction Materials | 316L Stainless Steel, USP Class VI Polymers |
| Control Software | UNICORN™ v7.x or later |
| Regulatory Compliance | FDA 21 CFR Part 11, GMP/GLP-Ready, cGMP-Compatible Design |
| Pressure Monitoring | 4 Real-Time Channels |
| Sensor Integration | Temperature, UV Absorbance, pH, Conductivity, Air Flow, Transmembrane Pressure (TMP), Differential Pressure (ΔP), Crossflow & Permeate Flow Rates |
| Pump Types | Sanitary Diaphragm & Rotary Lobe Pumps |
| Integrity Test | Integrated Air Flow-Based Filter Integrity Testing |
| Cleaning | Fully Automated CIP Method Development & Water Flux Validation |
| Data Management | Audit Trail, Electronic Signatures, 10-Tier User Access Control, Logbook Generation |
| Brand | Cytiva |
|---|---|
| Origin | United Kingdom |
| Manufacturer Type | Authorized Distributor |
| Product Origin | Imported |
| Model | Omics Bundle |
| Separation Method | Automated |
| Cell Viability (%) | ≥90% |
| Compatibility | Requires Integration with VIA Freeze™ Uno Controlled-Rate Freezer |
| Intended Use | Research Use Only (RUO), Not for Diagnostic Purposes |
| Brand | Cytiva |
|---|---|
| Origin | Switzerland |
| Model | VIA Freeze Uno |
| Type | Fully Automatic |
| Cooling Method | Conductive |
| Compliance | cGMP, 21 CFR Part 11, ISO 13485 (via system integration) |
| Sample Capacity | 24–192 cryovials, up to 250 mL cryobags, cryostraws (with interchangeable trays) |
| Calibration | Supplied with NIST-traceable calibration certificate |
| Qualification | IQ/OQ documentation included |
| Software Integration | Chronicle™ Automation Platform (optional standalone purchase) |
| Brand | Cytiva |
|---|---|
| Origin | Switzerland |
| Model | VIA Thaw™ L1000 |
| Type | Fully Automated |
| Regulatory Classification | Import (CE-marked for IVD/medical device use in EU |
| Compliance | Designed to support GMP-compliant cell therapy manufacturing per ISO 13485, ASTM F3276-21 (Standard Guide for Cryopreserved Cellular Product Thawing), and 21 CFR Part 11–ready when used with Chronicle™ software |
| Brand | Cytiva |
|---|---|
| Origin | Sweden |
| Model | Wave Mixer |
| Type | Sterile, Non-invasive Oscillatory Mixing System |
| Capacity | Up to 10 L |
| Mixing Time | ≤6.5 s for full homogenization |
| Drive Mechanism | Programmable Rocking Platform with Sinusoidal Motion Profile |
| Sterility Assurance | Designed for use with single-use bioprocess bags (e.g., Cytiva HYPERSTACK®, Xcellerex™ XDR bags) |
| Compliance | Compatible with ISO 13485–certified manufacturing environments and supports GMP-aligned process validation |
| Brand | Cytiva |
|---|---|
| Origin | USA |
| Manufacturer Type | Authorized Distributor |
| Origin Category | Imported |
| Model | Xcellerex APS |
| Pricing | Available Upon Request |
| Brand | Cytiva |
|---|---|
| Origin | USA |
| Manufacturer Type | Authorized Distributor |
| Product Category | Imported |
| Model | Xcellerex XDR 50–2000 |
| Working Volumes | 50 L, 200 L, 500 L, 1000 L, 2000 L |
| Brand | Cytiva |
|---|---|
| Origin | USA |
| Configuration | Single-Vessel System |
| Working Volume | 4.5–10 L |
| Contact Materials | USP Class VI Low-Density Polyethylene (LDPE) |
| Compliance | cGMP-Compatible Design for Clinical & Commercial Biomanufacturing |
| Scalability | Direct Fluid Dynamic & Control Parity with Xcellerex XDR-50, XDR-200, XDR-500, XDR-1000, and XDR-2000 Systems |
| Impeller Type | Low-Shear, High-Power Magnetic Coupling Impeller with Integrated Sparger |
| Automation Architecture | Smart Process Control with Integrated Data Logging and Audit Trail Capability |
| Brand | Cytiva |
|---|---|
| Origin | USA |
| Manufacturer Type | Authorized Distributor |
| Product Category | Imported |
| Model | Xcellerex XDR-50 MO |
| Application Domain | Microbial Fermentation |
| Brand | Cytiva |
|---|---|
| Origin | USA |
| Manufacturer Type | Authorized Distributor |
| Product Origin | Imported |
| Model | Xcellerex™ |
| Pricing | Available Upon Request |
| Brand | Cytiva |
|---|---|
| Origin | USA |
| Manufacturer Type | Authorized Distributor |
| Product Category | Imported |
| Model | Xcellerex™ X-200 |
| Pricing | Upon Request |
| Application Domain | Biopharmaceutical Process Development & Manufacturing |
| Brand | Cytiva |
|---|---|
| Origin | USA |
| Manufacturer Type | Authorized Distributor |
| Product Category | Imported |
| Model | Xcellerex™ X-50 |
| Pricing | Upon Request |
| Application Domain | Biopharmaceutical Cell Culture |
| Brand | Cytiva |
|---|---|
| Origin | Switzerland |
| Manufacturer Type | Authorized Distributor |
| Product Origin | Imported |
| Model | Xuri Cell Expansion System W25 |
| Price | Upon Request |
| Temperature Range | 15°C–32°C |
| Working Volume | 25 L |
| Brand | Cytiva |
|---|---|
| Origin | Switzerland |
| Model | NanoAssemblr™ Blaze™ System |
| Volume Range | 1 L (Blaze™) / up to 10 L (Blaze+™) |
| System Architecture | Closed, single-use, GMP-ready process development platform |
| Core Technology | NxGen™ microfluidic mixing |
| Regulatory Alignment | Supports FDA 21 CFR Part 11–compliant data integrity, GLP/GMP process transfer readiness |
| Disposable Components | NxGen™ single-use chips (with/without inline dilution), sterile tubing kits for external vessel integration |
| Downstream Compatibility | Integrated TFF, sterile filtration, and fill-finish workflow support |
| Scalability Pathway | Direct CPP/CQA transfer from Blaze™ → Blaze+™ → NanoAssemblr™ Production System |
| Brand | Cytiva |
|---|---|
| Origin | Switzerland |
| Manufacturer Type | Authorized Distributor |
| Product Category | Imported |
| Model | NanoAssemblr™ Commercial Manufacturing System |
| Pricing | Upon Request |
| Flow Rate Range | 6–48 L/h (dependent on disposable flow path kit) |
| Brand | Cytiva |
|---|---|
| Origin | Switzerland |
| Model | NanoAssemblr™ GMP System |
| Flow Rate Range | 60–200 mL/min (3.6–12 L/h) |
| Brand | Cytiva |
|---|---|
| Origin | Switzerland |
| Model | NanoAssemblr™ Spark™ Instrument |
| Sample Volume Range | 25–250 µL |
| Mixing Technology | NxGen™ Microfluidic Vortex Mixing |
| Sterility Design | BSC-Compatible, Aseptic Operation Ready |
| Chip Type | Single-Use, Gamma-Irradiated Cartridge |
| Scalability Pathway | Seamless Transition to NanoAssemblr™ Ignite™ and NxGen™ Production Platforms |
| Regulatory Context | Designed for GLP-compliant early-stage development, supports 21 CFR Part 11–ready software integration (optional) |
| Brand | Cytiva |
|---|---|
| Origin | Sweden |
| Manufacturer Type | Authorized Distributor |
| Origin Category | Imported |
| Model | OligoPilot |
| Pricing | Available Upon Request |
| Brand | Cytiva |
|---|---|
| Origin | USA |
| Manufacturer Type | Authorized Distributor |
| Origin Category | Imported |
| Model | VIA capsule |
| Pricing | Available Upon Request |
| Brand | Cytiva |
|---|---|
| Origin | Sweden |
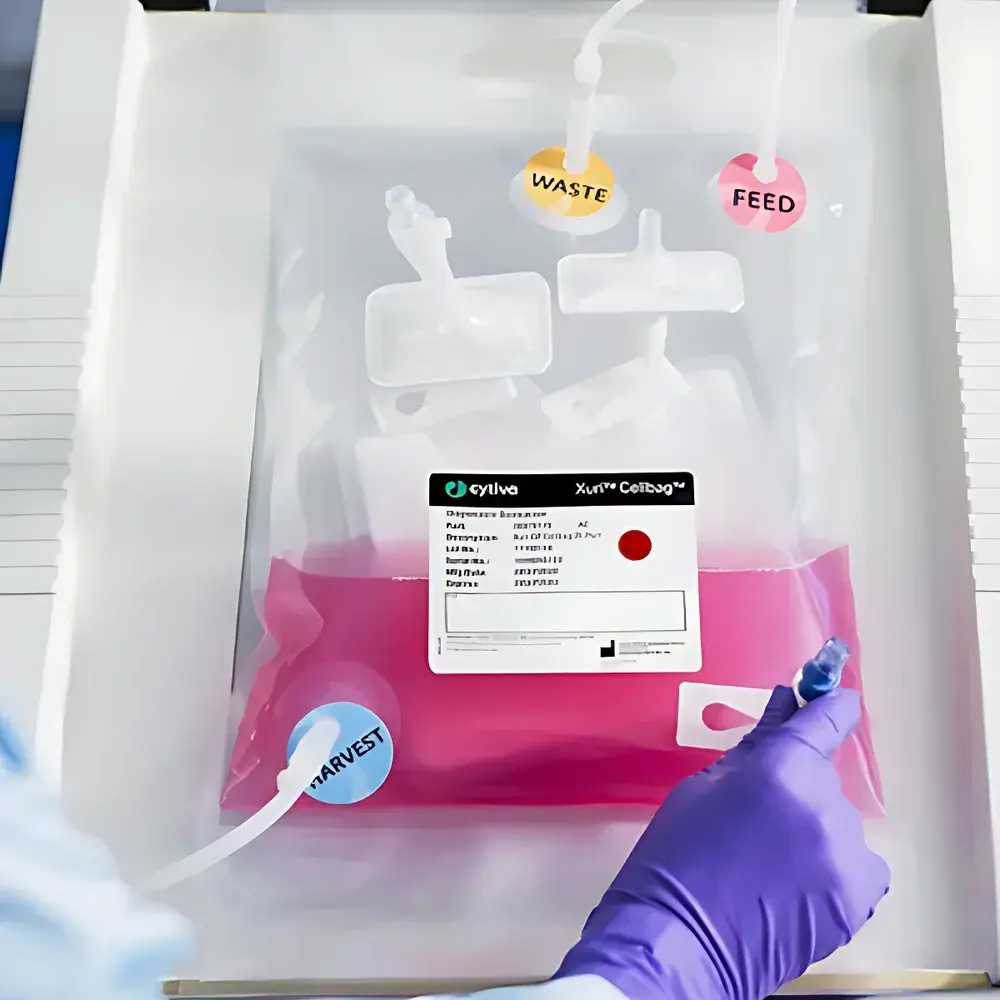
| Model | Wave Cellbag |
| Sterilization Method | Gamma Irradiation |
| Material Compliance | USP Class VI |
| Regulatory Documentation | FDA-registered DMF on file |
| Intended Use | Research, Process Development, cGMP Manufacturing, Cell Therapy Production |
| Bag Configuration | Single-use, pre-sterilized, disposable bioprocess bag |