- All
- Favorite
- Popular
- Most rated
| Brand | Bruker |
|---|---|
| Origin | Germany |
| Manufacturer Type | Authorized Distributor |
| Product Origin | Imported |
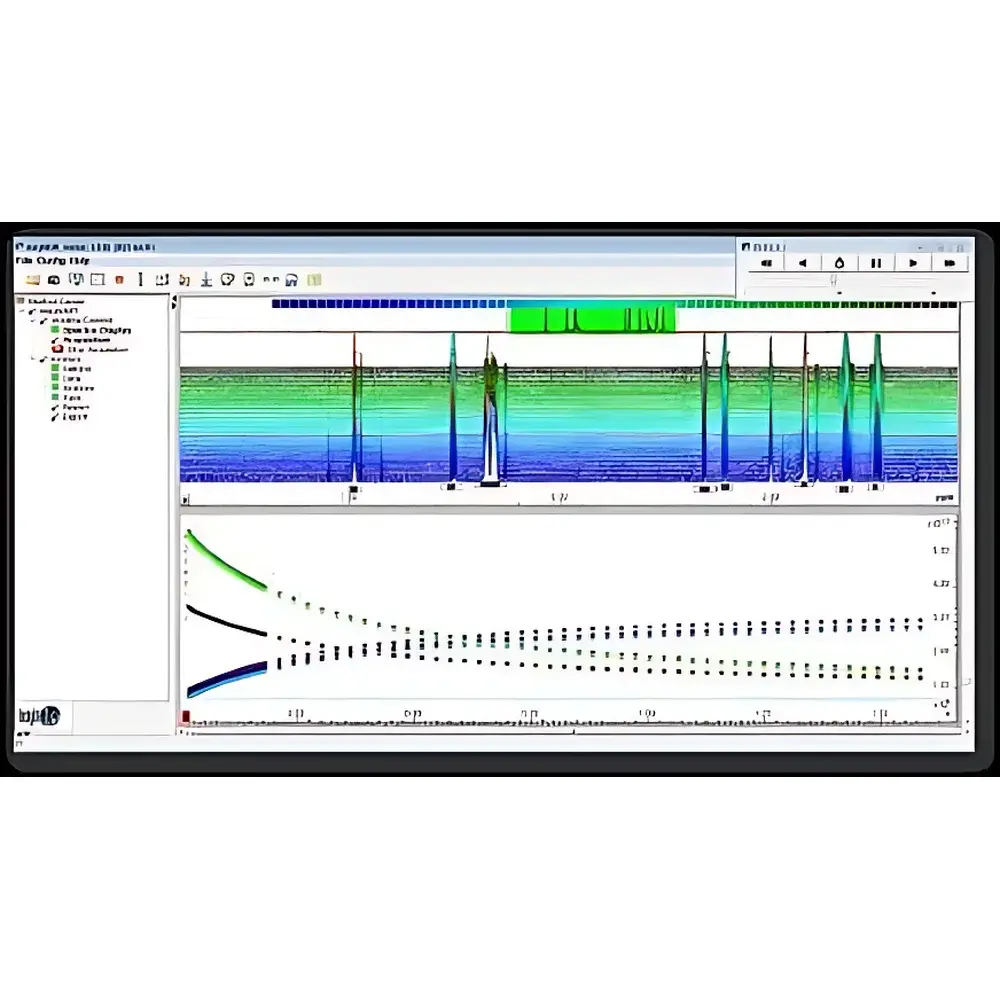
| Model | AVANCE NEO 600M NMR Spectrometer (Ascend Evo Magnet) |
| Instrument Type | High-Field NMR Spectrometer |
| Sample Compatibility | Solid and Liquid Samples |
| Operating Frequency | 600 MHz |
| Data Acquisition Method | Pulsed Fourier Transform |
| Brand | Bruker |
|---|---|
| Model | CryoProbe |
| Origin | Imported |
| Cooling Principle | Closed-cycle helium refrigeration (Gifford-McMahon cryocooler) |
| Operating Temperature | ~20 K (probe coil), ~25 K (preamp) |
| Signal-to-Noise (S/N) Gain | Up to 4× vs. room-temperature probes |
| Compatible Systems | Bruker AVANCE III/IV NMR platforms |
| Probe Configurations | Dual-tuned (e.g., ¹H/¹³C), triple-resonance (e.g., ¹H/¹³C/¹⁵N), with ²H lock and Z-gradient capability |
| Compliance | Designed for GLP/GMP-compliant labs |
| Brand | Bruker |
|---|---|
| Origin | Germany |
| Instrument Type | High-Field NMR Spectrometer Software Module |
| Sample Compatibility | Solid and Liquid Samples (Unified Workflow) |
| Sensitivity | Signal-to-Noise Ratio (SNR)-Driven Analysis Validation |
| Resolution | Linewidth-Based Spectral Fidelity Assessment |
| Deployment | Native Integration with Bruker TopSpin™ and IconNMR™ Platforms |
| Regulatory Alignment | Supports GLP/GMP Documentation Requirements, FDA 21 CFR Part 11 Compliant Audit Trail (Optional Configuration) |
| Brand | Bruker |
|---|---|
| Origin | Switzerland |
| Model | DNP-NMR |
| Field Strength Options | 263 GHz / 395 GHz (corresponding to ~9.4 T and ~14.1 T for ¹H, respectively) |
| Sample Type | Solid-state and semi-solid biological macromolecules |
| Operating Temperature | ~100 K |
| Microwave Power Source | High-power gyrotron |
| Sensitivity Enhancement | 20–80× over conventional ssNMR |
| Probe Type | Cryogenic Magic Angle Spinning (MAS) probe |
| Compliance | Designed for GLP/GMP-aligned workflows with audit-trail-capable data acquisition |
| Brand | Bruker |
|---|---|
| Origin | Germany |
| Model | minispec (mq-series platform) |
| Measurement Principle | Time-Domain Nuclear Magnetic Resonance (TD-NMR) |
| Sample Tube Options | 10 mm (mq40), 40 mm (mq-one), 50 mm (mq7.5) |
| Compliance | ASTM D8139, ISO 10565, AOAC 994.01, USP <1210> |
| Data Integrity | Audit trail-enabled software compliant with FDA 21 CFR Part 11 requirements for regulated QC/QA environments |
| Calibration | Minimal reference sample calibration |
| Brand | Bruker |
|---|---|
| Origin | Germany |
| Model | AVANCE NEO |
| Instrument Type | High-Field NMR Spectrometer |
| Sample Compatibility | Solid-State & Solution-State (Dual-Mode) |
| Operating Frequency Range | 300–800 MHz (scalable to ≥1.25 GHz) |
| Data Acquisition Method | Pulse Fourier Transform (PFT) |
| Architecture | Integrated Transceiver-Based Digital RF Platform |
| Software Platform | TopSpin 4.x or later with Embedded Acquisition Server |
| Compliance | Designed for GLP/GMP environments |
| Brand | Bruker |
|---|---|
| Origin | Germany |
| Manufacturer Type | Authorized Distributor |
| Origin Category | Imported |
| Model | SkyScan1278 |
| Instrument Type | Tomographic Imaging System |
| Energy Resolution | 17% |
| Spatial Resolution | 50 µm |
| Scan Time | 7.2 s per full-body mouse scan |
| Field of View Diameter | 80 mm |
| Scan Length | 200 mm |
| Sample Capacity | 1 animal per scan |
| X-ray Source | Adjustable 20–65 kV microfocus tube with patented beam shaping optics |
| Detector | High-sensitivity CMOS flat-panel detector |
| Dose Level | <6 mGy for whole-body murine scan |
| Physiological Gating | Integrated respiratory, ECG, temperature, and motion monitoring |
| Data Output Formats | DICOM, TIFF, JPG, BMP, PNG, AVI |
| Software Compliance | GLP-compliant acquisition and reconstruction suite |
| Brand | Bruker |
|---|---|
| Origin | France |
| Manufacturer Type | Authorized Distributor |
| Origin Category | Imported |
| Model | minispec Food & Beverage Droplet Size Analyzer |
| Automation Level | Semi-Automatic |
| Detection Scope | Milk Composition & Emulsion Droplet Size Distribution |
| Measurement Principle | Time-Domain Nuclear Magnetic Resonance (TD-NMR) |
| Validated For | Single Emulsions (O/W and W/O) and Double Emulsions (W/O/W) |
| Droplet Size Range | 0.5–10 µm (O/W), 1–30 µm (W/O) |
| Minimum Detectable Dispersed Phase | 2% (v/v) |
| Sample Prep | Minimal — No Dilution or Modification Required |
| Regulatory Alignment | Compliant with GLP practices |
| Brand | Bruker |
|---|---|
| Origin | Germany |
| Model | InsightMR™ |
| Instrument Type | High-Field Online Process NMR Spectrometer |
| Sample Compatibility | Solid-Liquid Hybrid (Flow & Batch) |
| Software Platform | TopSpin®-Based Dedicated Reaction Monitoring Suite |
| Regulatory Context | Designed for GLP/GMP-Compliant Process Analytical Technology (PAT) Environments |
| Integration | Fully Compatible with Bruker Avance™ III/IV NMR Platforms |
| Brand | Bruker |
|---|---|
| Origin | Germany |
| Manufacturer Type | Authorized Distributor |
| Product Origin | Imported |
| Model | ELEXSYS II |
| Pricing | Upon Request |
| Instrument Type | Continuous-Wave (CW) |
| Frequency Range Capability | 1 GHz to 263 GHz (CW and/or FT modes) |
| Optional Modules | DICE-II ENDOR, EPR Imaging, Pulsed FT-EPR, Multi-Frequency Resonators |
| Software Architecture | Open, Network-Enabled Client/Server Platform |
| Compliance Framework | Designed for GLP/GMP-aligned workflows |
| Brand | Bruker |
|---|---|
| Origin | Germany |
| Manufacturer Type | Authorized Distributor |
| Origin Category | Imported |
| Model | Ascend Evo 1.0 GHz NMR |
| Price Range | USD $10–20 million |
| Instrument Type | High-Field NMR Spectrometer |
| Sample Compatibility | Solid-State & Solution-State |
| Operating Frequency | 1.0 GHz |
| Acquisition Mode | Pulsed Fourier Transform (PFT) & Continuous Wave (CW) |
| Brand | Bruker |
|---|---|
| Origin | France |
| Manufacturer Type | Authorized Distributor |
| Origin Category | Imported |
| Model | minispec Automation |
| Pricing | Upon Request |
| Brand | Bruker |
|---|---|
| Origin | Germany |
| Model | BioSpec® 3T |
| Field Strength | 3 Tesla |
| Instrument Type | Preclinical Magnetic Resonance Imaging (MRI) and Magnetic Resonance Spectroscopy (MRS) System |
| Bore Diameter | 180 mm |
| RF Coil Options | >30 dedicated coils for mouse/rat head, brain, heart, body, multinuclear, and functional applications |
| Gradient Strength | Up to 900 mT/m |
| Gradient Linearity | ±3.5% within 50 mm DSV |
| Shim System | 6 high-order shim coils + high-power shim power supplies (up to 3750 Hz/cm² in XZ/YZ) |
| Magnetic Field Homogeneity | ±0.1 ppm within 50 mm DSV |
| Magnet Hold Time | >4 hours after power loss or coolant interruption |
| Software Platform | ParaVision® with >1000 optimized pulse sequences including IntraGate™, UTE, ZTE, and non-ECG-triggered cardiac imaging |
| Multi-Modal Compatibility | PET, optical imaging, CT |
| Regulatory Compliance | Designed for GLP-compliant preclinical research environments |
| Brand | Bruker |
|---|---|
| Origin | France |
| Instrument Type | Low-Field Time-Domain Nuclear Magnetic Resonance (TD-NMR) Analyzer |
| Sample Type | Liquid |
| Operating Frequency | 20 MHz |
| Sample Preparation | None required |
| Compliance | ASTM D 7171, ASTM D 4808, ASTM D 3701 |
| Brand | Bruker |
|---|---|
| Origin | France |
| Model | minispec LF90II |
| Instrument Type | In Vivo Rodent (Mouse/Rat) Body Composition Analyzer |
| Operating Frequency | 6.2 MHz |
| Measurement Time | < 120 s per animal |
| Sample Handling | Non-invasive, anesthesia-free, restraint-only in dedicated cylindrical holder |
| Compliance | Designed for GLP-compliant preclinical research environments |
| Brand | Bruker |
|---|---|
| Origin | France |
| Model | minispec LF50 / LF90 |
| Application | In vivo quantitative analysis of fat mass, lean body mass, and free fluid in live mice and rats |
| Measurement Principle | Pulsed low-field TD-NMR (1H) at 0.047 T (2 MHz) |
| Sample Orientation | Vertical or horizontal magnet configuration |
| Analysis Time | ≤ 120 s per animal |
| Animal Safety | Non-invasive, no ionizing radiation, no anesthesia required |
| Regulatory Context | Compliant with GLP-aligned data integrity requirements |
| Brand | Bruker |
|---|---|
| Origin | Germany |
| Instrument Type | Continuous-Wave (CW) EPR |
| Magnet System | Permanent Magnet |
| Cooling | Air-Cooled (No Water Required) |
| Form Factor | Benchtop |
| Automation Level | Fully Automated Tuning, Data Acquisition & Analysis |
| Application-Specific Configurations | e-scan A (Radiation Dosimetry), e-scan B (Beer Freshness QC), e-scan F (Food Irradiation Verification), e-scan M (Stable Radical & Spin Adduct Detection, e.g., ROS/RNS) |
| Brand | Bruker |
|---|---|
| Origin | Germany |
| Model | SkyScan 1276 |
| Imaging Modality | X-ray micro-computed tomography (micro-CT) |
| Spatial Resolution (nominal) | ≤2.8 µm |
| Scan Modes | Step-and-shoot, continuous gantry rotation with slip-ring interface |
| Scan Time per Reconstruction | as low as 3.9 s |
| Reconstructed Slice Count per Scan | >1600 slices |
| Max Image Matrix Size | 8000 × 8000 pixels |
| X-ray Source Voltage Range | 20–100 kV adjustable |
| Automatic Filter Changer | 6-position |
| Dose Reduction Technology | Beam-shaping collimator (patented by Bruker microCT) |
| Physiological Monitoring Integration | Respiratory gating, ECG synchronization, temperature stabilization, motion detection |
| Animal Handling | Interchangeable mouse/rat beds with integrated anesthesia mask and sensor interface |
| Real-time Dose Monitoring | On-screen dosimetry derived from projection image analysis |
| Reconstruction Acceleration | GPU-accelerated algorithms + InstaRecon® layered reconstruction (≥10× faster than conventional FBP) |
| Software Compliance | GLP-compliant software suite with audit trail, user access control, and electronic signature support |
| Data Export & Remote Viewing | Direct export to iOS/Android devices via dedicated viewer app |
| Brand | Bruker |
|---|---|
| Origin | Germany |
| Manufacturer Type | Authorized Distributor |
| Origin Category | Imported |
| Model | A2B |
| Instrument Type | High-Field NMR Spectrometer |
| Sample Type | Solid |
| Sensitivity | Signal-to-Noise Ratio (SNR) |
| Resolution | Spectral Linewidth |
| Brand | Bruker |
|---|---|
| Origin | Germany |
| Model | EMXplus, EMXmicro |
| Instrument Type | Continuous-Wave (CW) EPR |
| Configuration | Benchtop, Integrated Digital Architecture |
| Control Interface | Ethernet-connected, WinEPR Software Platform |
| Magnetic Field Resolution | 18-bit digital |
| Signal Digitization | 18-bit ADC |
| Microwave Bridge | Next-generation EMXplusX™ |
| Detection Modes | Simultaneous dual-phase (0° & 90° modulation), 1st- and 2nd-harmonic detection |
| Brand | Bruker |
|---|---|
| Origin | Germany |
| Instrument Type | High-Field NMR Spectrometer |
| Model | AVANCE NEO 500M |
| Sample Compatibility | Liquid and Solid-State (Dual-Mode) |
| Sensitivity | High Signal-to-Noise Ratio (SNR) |
| Resolution | High Spectral Resolution (Narrow Linewidth) |
| Magnet System | ASCEND™ 500 MHz Superconducting Magnet |
| Console Architecture | Integrated Embedded Acquisition Server (TopSpin 4+) |
| Compliance | GLP/GMP-ready, FDA 21 CFR Part 11–compatible software modules available |
| Brand | Bruker |
|---|---|
| Origin | Germany |
| Manufacturer Type | Authorized Distributor |
| Origin Category | Imported |
| Model | LabScape |
| Price Range | USD 14,000 – 70,000 (est.) |
| Instrument Type | High-Field NMR Spectrometer |
| Sample Compatibility | Solid-State and Solution-State NMR |
| Sensitivity | Optimized for high signal-to-noise ratio (SNR) across standard probe configurations |
| Resolution | Tuned for high spectral resolution (line shape fidelity) in both ¹H and multinuclear experiments |
| Brand | Bruker |
|---|---|
| Origin | Germany |
| Manufacturer Type | Authorized Distributor |
| Origin Category | Imported |
| Model | JuiceScreener |
| Instrument Type | NMR-Based Beverage Authenticity Analyzer |
| Application | Laboratory |
| Measurement Time | ≤4 min |
| Compliance | ASTM D8271-21, ISO 21569:2022 (NMR-based food authenticity), GLP-compliant workflow via SampleTrack™ |
| Software Platform | TopSpin 4.2 + JuiceScreener Module v3.1 |
| Database | >16,000 reference NMR spectra from authentic fruit juices across 50+ global production regions |
| Quantified Analytes | ≥30 compounds (glucose, fructose, sucrose, citric acid, malic acid, isocitric acid, quinic acid, ethanol, fumaric acid, lactic acid, HMF, galacturonic acid, chloride, etc.) |
| Brand | Bruker |
|---|---|
| Origin | USA |
| Manufacturer Type | Authorized Distributor |
| Origin Category | Imported |
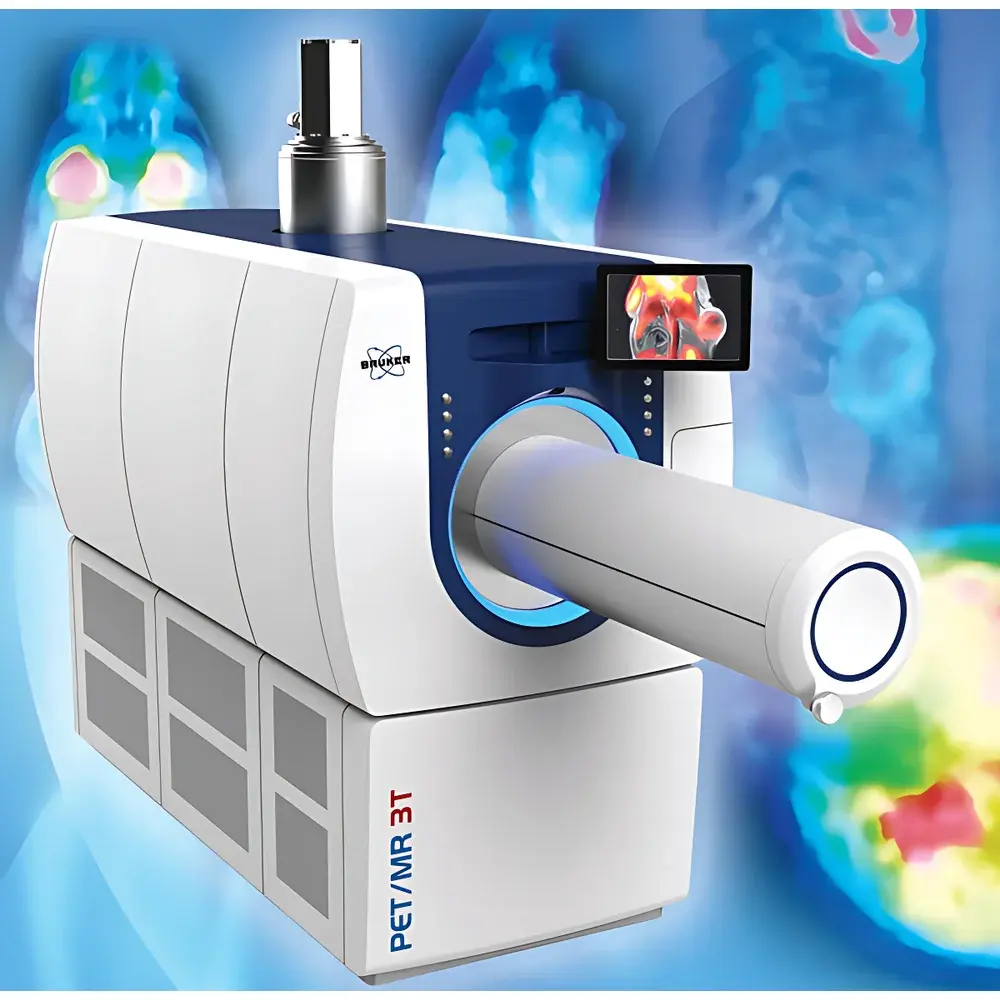
| Model | PET/MR 3T |
| Imaging Modality | Integrated Positron Emission Tomography / Magnetic Resonance Imaging |
| Energy Resolution | 17% FWHM at 511 keV |
| Spatial Resolution (PET) | <0.7 mm (NEMA NU-4) |
| Spatial Resolution (MRI) | ≤1.2 mm isotropic (with CryoProbe™) |
| Scan Speed | Up to 10 min per dynamic PET frame (variable by protocol) |
| Field of View (FOV) | 80 mm (PET transaxial), 148 mm (MRI transaxial), 285 mm (whole-body MRI longitudinal) |
| Animal Capacity | 1 subject per scan (mouse or rat) |
Show next