ATAGO PAL Series Pharmaceutical-Grade Digital Portable Refractometer
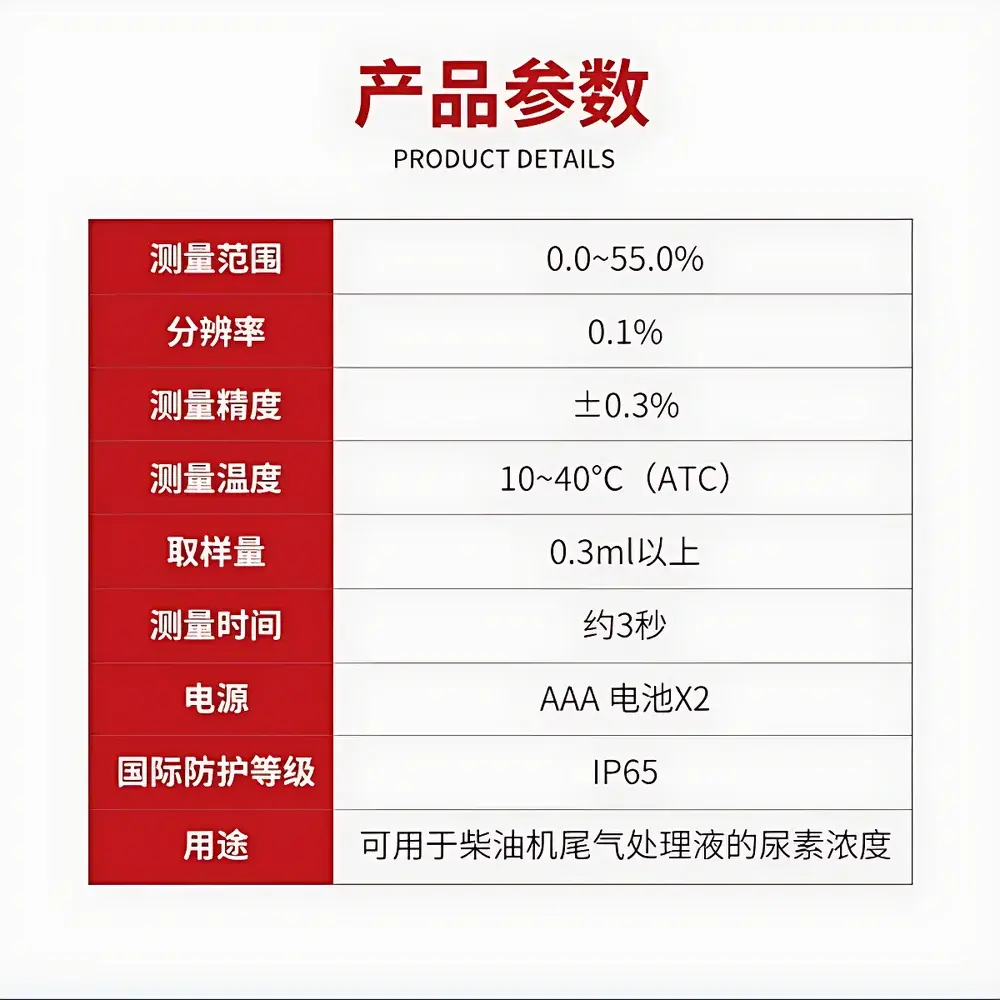
| Brand | ATAGO |
|---|---|
| Origin | Japan |
| Model | PAL Series (Pharmaceutical Formulation Dedicated) |
| Product Type | Handheld Refractometer |
| Display | Digital LCD |
| Temperature Compensation | Automatic (ATC), 10–40 °C |
| Measurement Range | 0.0–45.0% w/w (Brix equivalent) |
| Accuracy | ±0.4% w/w |
| Compliance | ISO 2173, ASTM D1074, USP <781>, JP XVII General Test for Refractive Index |
Overview
The ATAGO PAL Series Pharmaceutical-Grade Digital Portable Refractometer is a precision optical instrument engineered for rapid, on-site quantification of solute concentration in liquid pharmaceutical formulations—such as syrups, elixirs, suspensions, ophthalmic solutions, and injectable diluents—via measurement of refractive index. Based on the principle of total internal reflection at the prism–sample interface, the device converts refractive index values into mass concentration (% w/w) using pre-calibrated polynomial algorithms traceable to NIST-certified sucrose standards. Unlike generic food-grade refractometers, the PAL Series for pharmaceutical use features enhanced optical path stability, reduced thermal drift, and a dedicated calibration protocol aligned with pharmacopoeial requirements for excipient quantification and batch release testing. Its compact, single-hand operable form factor enables direct measurement in cleanroom environments, QC laboratories, or production line checkpoints without requiring sample dilution or temperature equilibration.
Key Features
- Pharmaceutical-specific calibration curve optimized for aqueous glycerol, propylene glycol, sorbitol, and sucrose-based vehicles—validated across 0.0–45.0% w/w concentration range
- Automatic Temperature Compensation (ATC) with high-stability thermistor (±0.2 °C accuracy) covering 10–40 °C operational range, eliminating manual correction per USP
- Digital LCD display with backlighting, resolution of 0.1% w/w, and real-time measurement stabilization indicator
- IP65-rated magnesium alloy housing resistant to ethanol, isopropanol, and common disinfectants; suitable for ISO Class 5–8 cleanrooms
- Non-invasive measurement: ≤0.3 mL sample volume required; prism surface designed for easy wipe-clean decontamination between batches
- Built-in START ZERO function enabling rapid zero-point verification with purified water prior to each measurement sequence
Sample Compatibility & Compliance
The PAL Series refractometer is validated for use with low-viscosity, optically clear to slightly opalescent aqueous solutions typical of oral liquids, nasal sprays, and topical gels. It excludes turbid suspensions, emulsions, or highly viscous semisolids (>500 mPa·s) where light scattering compromises refractive index fidelity. Instrument performance conforms to ISO 2173 (fruit juice analysis methodology), ASTM D1074 (standard test method for refractive index of asphaltic materials—adapted for pharmaceutical solvents), and USP Chapter “Refractive Index” for identity and assay verification. Calibration certificates are provided with traceability to JCSS (Japan Calibration Service System) and include uncertainty budgets compliant with ISO/IEC 17025 requirements for regulated laboratories.
Software & Data Management
While the PAL Series operates as a standalone instrument, optional RS-232 output (via ATAGO RS-232 Adapter Kit) enables integration with LIMS or electronic lab notebooks. All measurements store timestamp, operator ID (manually entered), and ambient temperature metadata. For GxP environments, the device supports audit-trail-ready workflows when paired with ATAGO’s optional PC software (ReflaSoft™ v3.2), which generates 21 CFR Part 11-compliant reports—including user authentication logs, calibration history, and raw refractive index vs. % w/w conversion tables. Data export formats include CSV, PDF, and XML for seamless import into statistical process control (SPC) platforms.
Applications
- Batch release testing of syrup formulations (e.g., dextromethorphan HBr syrup, paracetamol suspension vehicle)
- In-process monitoring of active pharmaceutical ingredient (API) concentration during solution-phase manufacturing
- Excipient verification (e.g., glycerin content in cough drops, propylene glycol in transdermal gels)
- Stability study support: tracking concentration drift over accelerated storage conditions (40 °C/75% RH)
- Raw material incoming inspection for polyol-based excipients against CoA specifications
- Supporting compendial compliance for JP XVII, EP 2.2.6, and USP refractive index acceptance criteria
FAQ
Does the PAL Series comply with FDA 21 CFR Part 11 for electronic records?
Yes—when used with ReflaSoft™ software and configured with user authentication, electronic signatures, and audit trail activation.
Can it measure non-sugar solutes such as sodium chloride or urea?
It provides accurate % w/w readings only for substances with refractive index response calibrated against the built-in pharmaceutical curve; custom calibration is not supported on-device.
Is periodic recalibration required?
Annual recalibration by an ATAGO-accredited service center is recommended; field verification with certified reference standards (e.g., 10.00 ± 0.02% w/w sucrose) should be performed daily in GMP settings.
What cleaning agents are compatible with the prism surface?
Deionized water, 70% isopropanol, or 0.1% sodium lauryl sulfate solution—avoid acetone, chlorinated solvents, or abrasive wipes.
How does ATC affect measurement uncertainty in controlled-room-temperature environments?
Within 20–25 °C, ATC contributes <±0.05% w/w to total measurement uncertainty; full uncertainty budget includes repeatability (±0.15% w/w) and calibration standard uncertainty (±0.08% w/w).