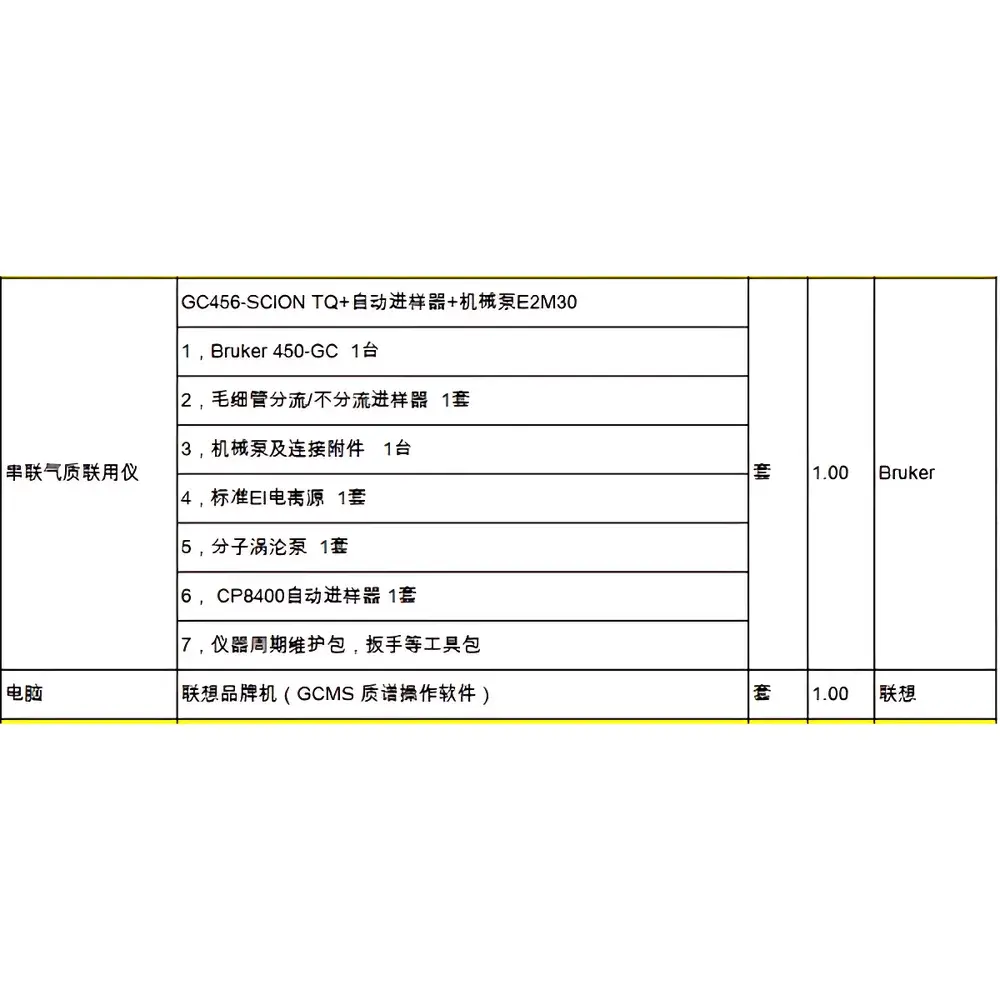
Bruker GC456-SCION TQ Triple Quadrupole Gas Chromatography-Mass Spectrometry System
| Brand | Bruker |
|---|---|
| Origin | USA |
| Model | GC456-SCION TQ |
| Configuration | Used, Refurbished & Fully Qualified Triple Quadrupole GC-MS/MS System |
| Compliance | Meets ASTM D5845, EPA Method 8270/8260, and ISO 17025-aligned operational readiness standards |
Overview
The Bruker GC456-SCION TQ is a high-performance, triple quadrupole gas chromatography–tandem mass spectrometry (GC-MS/MS) system engineered for targeted quantitative analysis in regulated and research environments. Built upon Bruker’s GC456 gas chromatograph platform and integrated with the SCION TQ mass spectrometer, this instrument delivers enhanced selectivity, sensitivity, and robustness for trace-level detection of volatile and semi-volatile organic compounds. The system operates on the principle of electron ionization (EI) and optional chemical ionization (CI), with tandem MS capability enabling precursor ion selection in Q1, collision-induced dissociation (CID) in Q2 (collision cell), and product ion detection in Q3—providing superior specificity in complex matrices. Designed for laboratories requiring compliance-ready performance, the GC456-SCION TQ supports method development, routine quantitation, and confirmatory analysis across environmental, food safety, forensic toxicology, and pharmaceutical QC applications.
Key Features
- Triple quadrupole architecture with independently controlled RF/DC voltages for optimized transmission and resolution across wide mass ranges (m/z 10–1000)
- High-efficiency EI source with dual-mode operation (EI/CI), thermally stable to 350 °C, and compatible with automated source cleaning protocols
- GC456 gas chromatograph featuring precision electronic pressure control (EPC), split/splitless injection, and programmable temperature vaporizing (PTV) inlet options
- Collision cell with inert gas (Ar or N₂) optimization for reproducible fragmentation and low chemical noise
- Refurbishment includes full system validation: vacuum integrity testing, mass calibration verification, detector gain adjustment, and tune report generation per Bruker SOPs
- Instrument qualification package provided: Installation Qualification (IQ), Operational Qualification (OQ), and documented Performance Verification (PV) data aligned with ISO/IEC 17025 requirements
Sample Compatibility & Compliance
The GC456-SCION TQ accommodates standard 1/8″ or 1/16″ capillary columns (e.g., DB-5ms, HP-5MS, Rxi-5Sil MS) and supports direct aqueous injection, thermal desorption (TD), purge-and-trap (P&T), and headspace (HS) interfaces via modular inlet expansion. It meets analytical requirements for EPA Methods 508.1, 525.3, 8270D, and 8260D; ASTM D5845 (gasoline oxygenates); and EU Commission Regulation (EU) No 37/2010 (veterinary drug residues). All refurbished units undergo rigorous leak testing, carrier gas purity verification, and baseline noise assessment to ensure compliance with GLP and GMP data integrity expectations. Full audit trail functionality—including user login logs, method change history, and raw data versioning—is enabled through Bruker’s Compass DataAnalysis software suite.
Software & Data Management
Controlled by Bruker’s Compass 2.1 software platform, the system provides intuitive method building, real-time acquisition monitoring, and automated quantitation workflows using internal standard calibration curves. Data files are stored in open-format .baf (Bruker Analysis Format), fully compatible with third-party processing tools including ACD/Labs MS Workbook Suite and SimGlycan. The software supports 21 CFR Part 11-compliant configurations when deployed with network authentication, electronic signatures, and role-based access control—enabling use in FDA-regulated laboratories. Raw data archiving, spectral library searching (NIST, Wiley, and Bruker’s own MassLib), and batch report generation are fully scriptable and audit-trail enabled.
Applications
- Environmental analysis: Quantification of PAHs, PCBs, organochlorine pesticides, and emerging contaminants (e.g., PFAS precursors) in soil, water, and air samples
- Food and beverage testing: Residue screening of mycotoxins, veterinary drugs, and process contaminants (e.g., furan, acrylamide) at sub-ppb levels
- Forensic toxicology: Confirmatory identification and quantification of drugs of abuse, metabolites, and novel psychoactive substances (NPS) in biological fluids
- Pharmaceutical impurity profiling: Detection and structural elucidation of genotoxic impurities per ICH M7 guidelines
- Industrial hygiene: Workplace air monitoring for VOCs and SVOCs per OSHA and NIOSH methods
FAQ
Is this unit covered by a warranty?
Yes—each refurbished GC456-SCION TQ is supplied with a 12-month comprehensive hardware and software support warranty, including remote diagnostics, firmware updates, and priority response for critical issues.
Can the system be validated for GxP use?
Yes—full IQ/OQ documentation, PQ protocol templates, and 21 CFR Part 11 configuration guidance are included. Optional on-site validation support is available upon request.
What maintenance history is available for this used instrument?
All units originate from decommissioned academic or contract research laboratories in North America and Europe. Full service logs, tune reports, and vacuum history are reviewed prior to refurbishment and made available to the buyer.
Does it include original consumables or spare parts?
A starter kit is included: one EI source, two GC column nuts, one septum, one liner pack, and one set of tuning standards (BFB, DFTPP). Additional consumables are available through Bruker-authorized channels.
Is training provided with purchase?
Yes—two days of on-site or virtual operator training covering instrument startup, method development, data processing, and routine maintenance procedures are included in the purchase agreement.