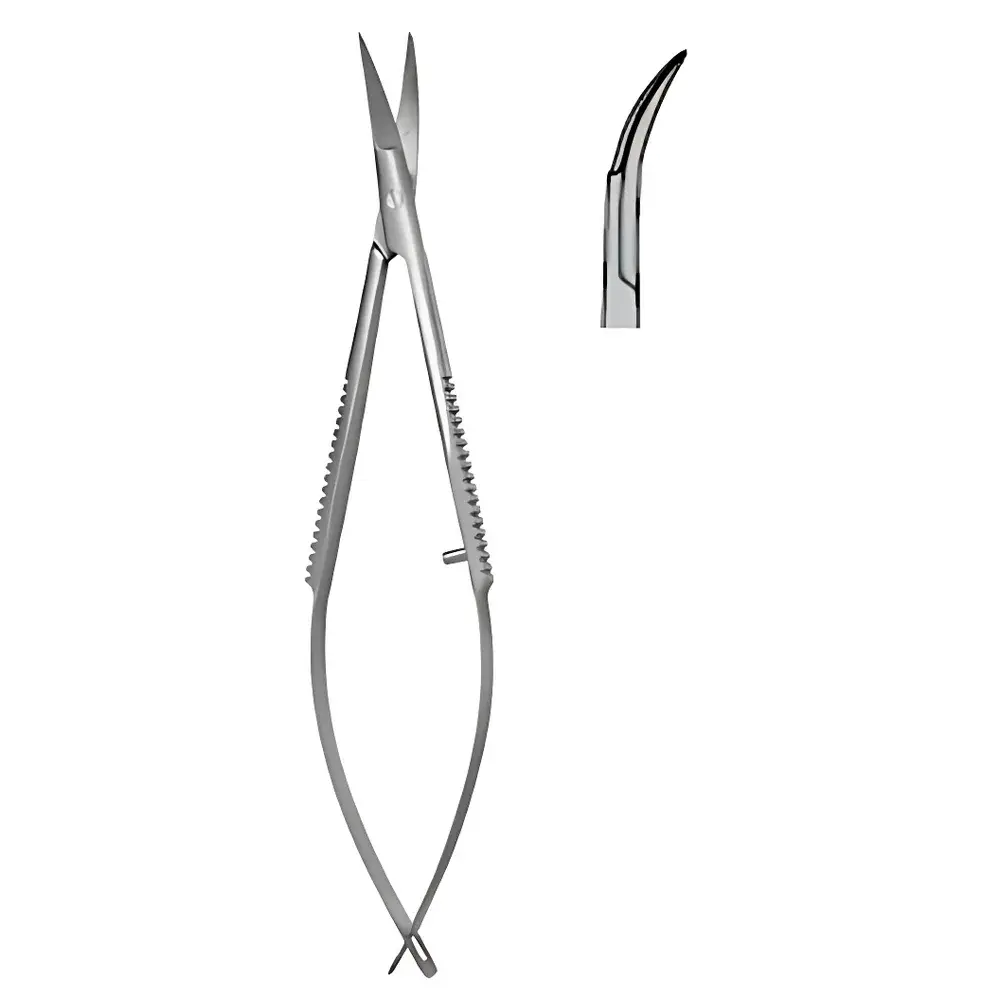
CASTROVIEJO-Style Micro Spring-Loaded Scissors – Curved, Blunt-Blunt Tip, S11011-11
| Brand | RWD |
|---|---|
| Origin | Shenzhen, China |
| Manufacturer Type | Direct Manufacturer |
| Country of Origin | China |
| Model | S11011-11 |
| Price | USD 60 |
Overview
The CASTROVIEJO-Style Micro Spring-Loaded Scissors – Curved, Blunt-Blunt Tip (Model S11011-11) is a precision surgical instrument engineered for delicate ophthalmic, neurosurgical, and microvascular procedures. Designed in accordance with the functional geometry and ergonomic principles of the classic Castroviejo scissor platform, this variant features a curved blade configuration and dual blunt tips to minimize tissue trauma during dissection, membrane peeling, or fine suture handling. The integrated stainless steel spring mechanism ensures consistent, fatigue-resistant actuation across thousands of cycles—critical for sustained intraoperative control under high-magnification visualization (e.g., operating microscopes or digital endoscopic systems). Constructed from medical-grade 420 stainless steel, the instrument meets ISO 7153-1:2016 requirements for surgical instruments and is fully compatible with standard autoclaving protocols (134 °C, 2–3 bar saturated steam, per ISO 17664).
Key Features
- Curved blade geometry optimized for improved visibility and access in confined anatomical spaces—particularly advantageous in anterior segment surgery and retinal membrane manipulation.
- Dual blunt tips prevent inadvertent puncture or laceration of fragile tissues such as Descemet’s membrane, internal limiting membrane (ILM), or dural layers.
- Precision-ground cutting edges maintain sharpness after repeated sterilization; edge retention validated per ASTM F1508-22 (Standard Test Method for Edge Retention of Surgical Blades).
- Ergonomic finger rings with textured matte finish enhance tactile control and reduce slippage during prolonged use under gloved conditions.
- Spring-loaded mechanism fabricated from corrosion-resistant phosphor bronze alloy, ensuring stable return force (±5% tolerance over 5,000 actuations) without plastic deformation.
- Total length of 115 mm (4.5 inches) balances maneuverability and leverage—ideal for both handheld and microscope-integrated workflows.
Sample Compatibility & Compliance
This instrument is intended for single-user, reusable application in sterile clinical environments. It is compatible with all standard sterilization methods including steam autoclaving (gravity and prevacuum cycles), ethylene oxide (EtO), and low-temperature hydrogen peroxide plasma. No material degradation or dimensional drift has been observed following 100 consecutive sterilization cycles under validated parameters. The device conforms to ISO 13485:2016 (Medical devices — Quality management systems) and carries CE marking under Regulation (EU) 2017/745 (MDR) for Class I non-sterile, reusable surgical instruments. Biocompatibility testing per ISO 10993-1:2018 confirms absence of cytotoxicity, sensitization, or intracutaneous reactivity.
Software & Data Management
As a manually operated mechanical instrument, the S11011-11 does not incorporate embedded electronics, firmware, or software interfaces. Its performance documentation—including sterilization logs, usage tracking, and maintenance records—is managed externally via institutional inventory systems compliant with FDA 21 CFR Part 11 (electronic records and signatures) and EU Annex 11 (Computerised Systems). Instrument traceability is supported through laser-etched alphanumeric serial numbering (per ISO 15223-1), enabling integration into hospital asset management platforms for GLP/GMP-aligned audit trails.
Applications
- Ophthalmology: Capsulorhexis support, ILM peeling, conjunctival dissection, and scleral flap preparation in trabeculectomy.
- Neurosurgery: Dural opening and closure, arachnoid dissection, and microvascular decompression adjunct use.
- Plastic & Reconstructive Surgery: Fine undermining in facial resurfacing, graft trimming, and dermal suture adjustment.
- Research Laboratories: Ex vivo tissue dissection in rodent ocular models, embryonic chick chorioallantoic membrane (CAM) studies, and organotypic slice preparation.
- Veterinary Microsurgery: Corneal transplant suturing, otoscopic-assisted tympanic membrane repair, and avian ophthalmic interventions.
FAQ
Is this instrument supplied sterile?
No. The S11011-11 is provided non-sterile and must be sterilized prior to first use per institutional protocol.
Can it be used with robotic surgical platforms?
It is designed for manual operation; compatibility with robotic instrument arms requires third-party mounting adapters and is not validated by RWD.
What is the recommended sharpening interval?
Sharpening is not advised; edge integrity is maintained through proper cleaning and sterilization. Resharpening alters blade geometry and voids compliance with ISO 7153-1.
Does RWD provide instrument traceability documentation?
Yes—each unit includes a Certificate of Conformance listing batch number, material certification (EN 10088-1), and sterilization validation summary upon request.
Is the spring mechanism user-serviceable?
No. The spring assembly is permanently crimped and not field-replaceable; instrument replacement is required if spring fatigue or loss of tension is observed.