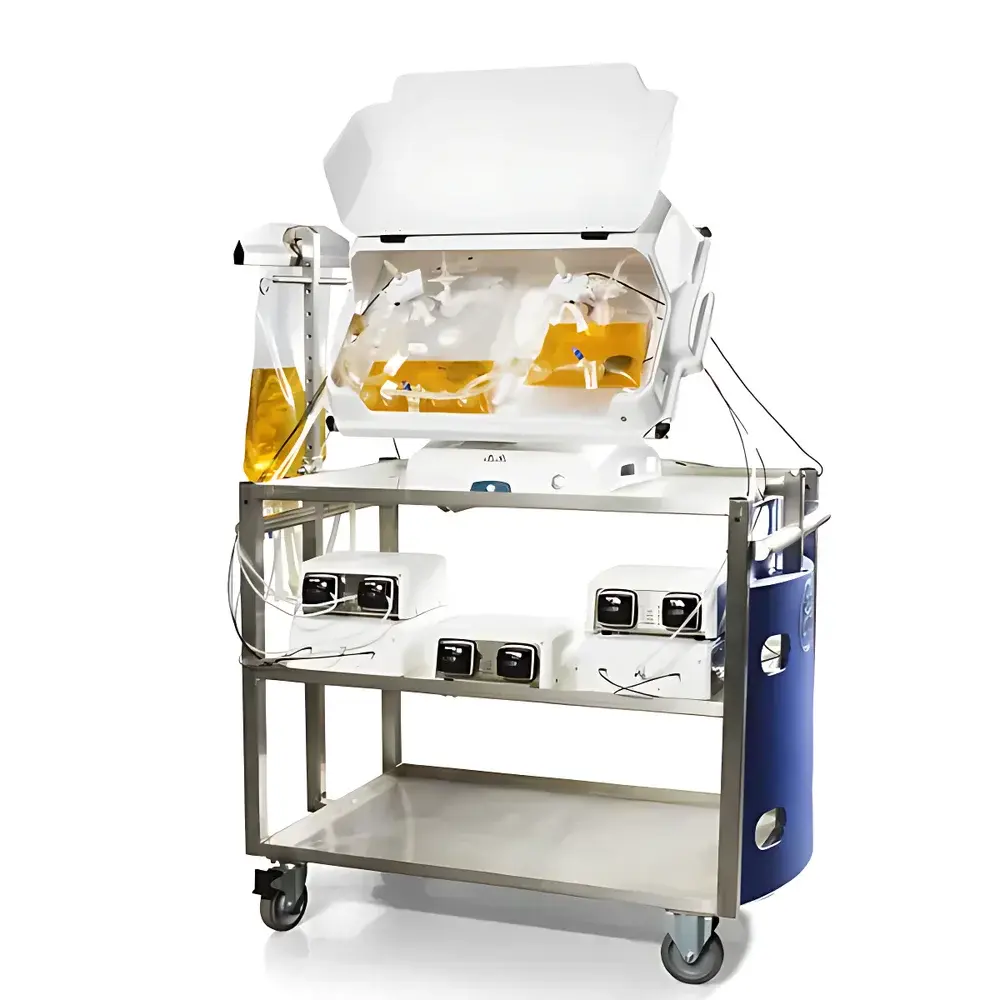
Cytiva ReadyToProcess WAVE 25 Wave Bioreactor
| Brand | Cytiva |
|---|---|
| Origin | Sweden |
| Model | WAVE 20/50 |
| Maximum Working Volume | 25 L |
| Tray Sizes | 5 L / 10 L / 25 L |
Overview
The Cytiva ReadyToProcess WAVE 25 Wave Bioreactor is an engineered platform for scalable, single-use mammalian and microbial cell culture under controlled physiological conditions. It operates on the well-established wave-mixing principle—where orbital rocking motion induces gentle, laminar fluid displacement within a sterile, pre-sterilized Cellbag™ bioprocess container. This hydrodynamic mechanism enables simultaneous oxygen transfer, CO₂ removal, and homogenous mixing without mechanical agitation or sparging, minimizing shear stress and foaming while maintaining high cell viability and productivity. Designed for process development, seed train expansion, and clinical-scale production (up to 25 L working volume), the system complies with current Good Manufacturing Practice (cGMP) design principles and supports regulatory submissions under ICH Q5D, Q5A(R2), and USP . Its modular architecture integrates seamlessly into end-to-end bioprocessing workflows—from upstream process characterization to tech transfer.
Key Features
- Gentle, low-shear wave mixing via programmable rocking parameters: adjustable speed (rpm), tilt angle (°), and acceleration profile (sinusoidal or asymmetric waveform)
- Integrated dual-mode temperature control: Peltier-heated tray with embedded RTD sensors; automatic power modulation based on real-time bag volume and internal temperature feedback
- Precision weight monitoring using factory-calibrated load cells (±0.1% FS accuracy); enables gravimetric feed/perfusion control and automated pump calibration
- Ergonomic tilting operation: 30°–45° inclination for simplified sampling, harvest, and bag handling without compromising sterility
- Modular tray system supporting three validated Cellbag™ sizes (5 L, 10 L, 25 L); spring-loaded quick-lock mechanism ensures repeatable bag alignment and sealing integrity
- Exhaust filter heater integrated as standard—prevents condensation-induced filter clogging during extended runs
- Redundant onboard data storage: mirrored solid-state drives (SSDs) retain all sensor logs, setpoints, alarms, and event timestamps with SHA-256 hash verification
Sample Compatibility & Compliance
The WAVE 25 accommodates Cytiva’s full portfolio of gamma-irradiated, USP Class VI-certified Cellbag™ bioreactors—including pH/DO optical sensor-integrated variants (e.g., Cellbag™ with Sartorius PreSens or Hamilton Arc sensors). All contact materials comply with ISO 10993-5 cytotoxicity standards and FDA 21 CFR Part 11 requirements for electronic records and signatures when paired with UNICORN 7.0 or later. The system meets IEC 61000-6-2/6-3 EMC specifications and carries CE marking per Machinery Directive 2006/42/EC. It supports GLP/GMP audit readiness through configurable user access levels, full audit trail logging (including parameter changes, login/logout events, and alarm acknowledgments), and time-stamped electronic signatures aligned with Annex 11 guidance.
Software & Data Management
Control and monitoring are executed via Cytiva’s UNICORN software platform (v7.2+), which provides deterministic real-time loop execution (<100 ms cycle time) and hierarchical alarm management (priority-based escalation, email/SMS notification integration). All operational data—including temperature, weight, rocking parameters, and external sensor inputs—are archived in ACID-compliant SQL Server databases with optional backup to network-attached storage (NAS). OPC UA server functionality enables bidirectional communication with MES (e.g., Siemens Opcenter, Rockwell FactoryTalk), SCADA systems, and digital twin environments. Raw data export adheres to ASTM E2500-07 and ISA-88 Part 1 standards for structured batch record generation.
Applications
- Monoclonal antibody (mAb) and recombinant protein production in CHO and HEK293 suspension cultures
- Viral vector manufacturing (AAV, lentivirus) requiring low-shear, high-viability environments
- Stem cell expansion under xeno-free, serum-free conditions
- Process characterization studies including DoE-based optimization of rocking parameters, gas overlay strategies, and feeding regimes
- Comparative scale-down modeling for bioreactor technology transfer (validated against stainless-steel stirred-tank reactors per ISPE Baseline Guide Vol. 4)
- Clinical batch manufacturing compliant with FDA IND/IMPD submission requirements
FAQ
What is the maximum recommended working volume for the WAVE 25 system?
The system is validated for reliable performance up to 25 L working volume using the 25 L Cellbag™; operation beyond this limit may compromise mixing uniformity and gas transfer efficiency.
Can the WAVE 25 operate under perfusion mode?
Yes—integrated gravimetric control enables closed-loop perfusion via peristaltic or diaphragm pumps, with real-time media replacement rate adjustment based on weight differential.
Is remote monitoring supported?
Remote access is available through UNICORN Web Client (HTTPS/TLS 1.2 encrypted), allowing secure visualization of live trends, historical data, and alarm status from any authorized workstation.
How is temperature uniformity verified across the Cellbag™?
Cytiva provides a qualified thermal mapping protocol (IQ/OQ documentation included) using calibrated thermocouples placed at nine predefined positions within representative 25 L bags under nominal operating conditions.
Does the system support PAT integration?
Yes—via analog/digital I/O modules and OPC UA, the WAVE 25 accepts input from third-party inline analyzers (e.g., Raman, NIR, capacitance probes) for advanced process analytical technology applications.