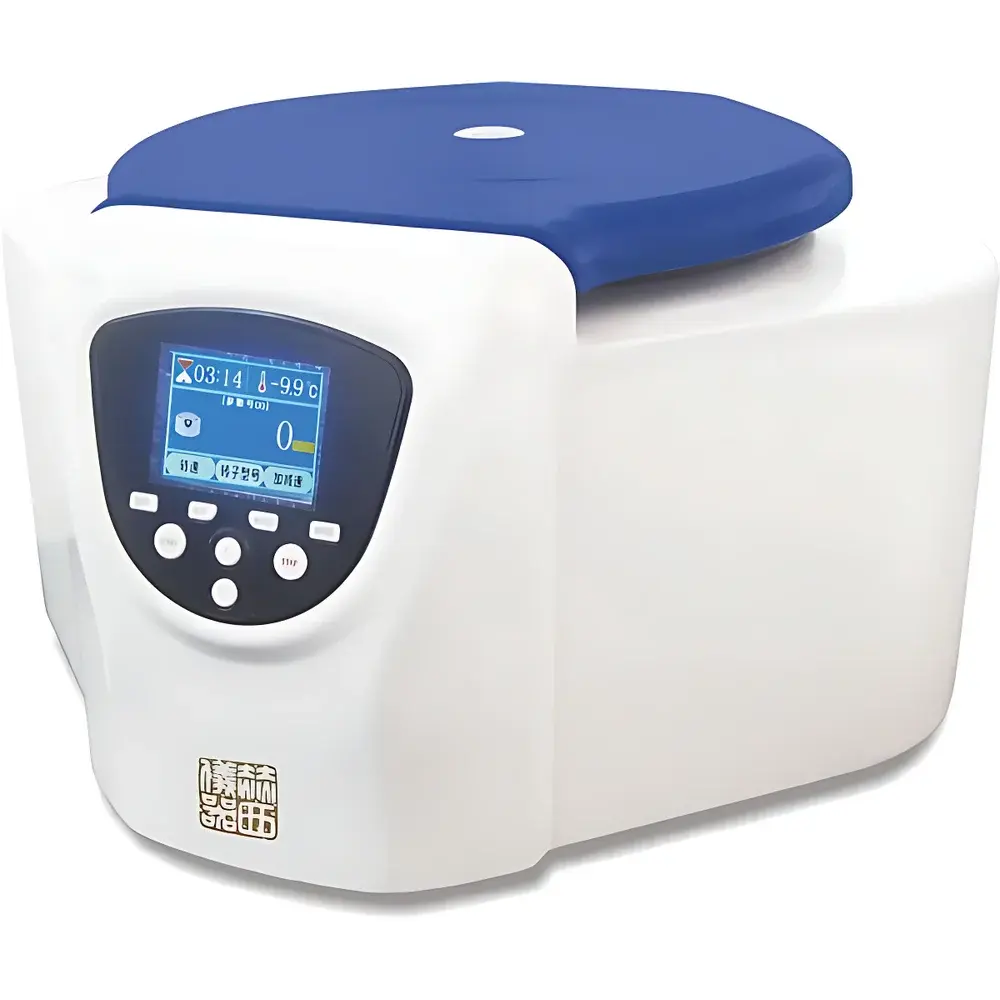
Herxi TDZ4-WS Benchtop Low-Speed Centrifuge
| Brand | Herxi |
|---|---|
| Origin | Hunan, China |
| Model | TDZ4-WS |
| Instrument Type | Benchtop Centrifuge |
| Speed Range | Up to 4000 rpm |
| Max RCF | 2650 ×g |
| Max Capacity | 4 × 100 mL |
| Dimensions (L×W×H) | 500 × 450 × 280 mm |
| Noise Level | ≤60 dB(A) |
| Power Supply | AC 220 V, 50 Hz |
| Motor | High-Torque Brushless DC Motor |
| Control System | Microprocessor-Based with TFT-LCD Touch + Keypad Interface |
| Acceleration/Deceleration | 10-step programmable |
| Programmable Methods | 20 user-defined protocols |
| Safety Certifications | ISO 9001:2015, ISO 13485:2016, CFDA Registered |
| Compliance | RoHS 2015/863 (Seal Material), GLP-Compatible Audit Trail Logging Enabled |
Overview
The Herxi TDZ4-WS Benchtop Low-Speed Centrifuge is a microprocessor-controlled, brushless DC motor-driven centrifugation system engineered for routine separation of suspended particles in liquid-phase samples across life science, clinical diagnostics, pharmaceutical quality control, food safety, and environmental testing laboratories. Operating on the principle of sedimentation under controlled gravitational force (RCF), this instrument delivers reproducible, low-shear separations ideal for pelleting cells, precipitating macromolecules, clarifying crude extracts, and preparing samples for downstream analysis such as ELISA, PCR, or chromatography. Its design prioritizes operational safety, acoustic comfort, and long-term mechanical stability—critical attributes for high-throughput core labs and regulated environments where equipment uptime and data traceability are essential.
Key Features
- High-torque brushless DC motor ensures consistent rotational performance with minimal thermal drift and extended service life; eliminates carbon brush wear and associated maintenance.
- Integrated dual-mode interface: 4.3-inch TFT-LCD color display supports both capacitive touch and physical keypad inputs, enabling real-time monitoring of setpoint vs. actual speed (rpm), RCF, time remaining, and acceleration/deceleration status.
- Programmable acceleration and deceleration profiles (10 steps each) reduce sample resuspension risk during spin-down—particularly critical for fragile pellets such as lymphocytes or bacterial cultures.
- Robust mechanical safety architecture includes automatic rotor imbalance detection, over-speed cutoff, over-temperature shutdown, and electromagnetic door interlock with stainless steel latch mechanism compliant with IEC 61010-2-020.
- Biocontainment-compliant angular rotor features full-silicone rubber gasketing (RoHS 2015/863 certified), minimizing aerosol generation during high-speed operation and supporting biosafety level 2 (BSL-2) workflows.
- Structural integrity reinforced by 304 austenitic stainless steel centrifuge chamber, triple-layer steel protective housing, and powder-coated steel chassis—designed for durability in shared lab spaces subject to frequent reconfiguration.
- Wide-voltage compatibility option (100–230 V AC, 50/60 Hz) enables deployment in international satellite labs without voltage conversion hardware.
Sample Compatibility & Compliance
The TDZ4-WS accommodates standard laboratory tubes including 10–15 mL conical, 50 mL polypropylene, and 100 mL glass or plastic centrifuge bottles. Compatible rotors include fixed-angle configurations optimized for rapid pelleting (e.g., 4 × 100 mL at 4000 rpm / 2490 ×g). All rotor assemblies undergo dynamic balancing verification per ISO 21501-4 and are validated for cyclic loading endurance exceeding 10,000 runs. The device meets CE marking requirements under Directive 2014/30/EU (EMC) and 2014/35/EU (LVD), carries CFDA registration (Class II medical device), and conforms to ISO 13485:2016 for medical device manufacturing quality systems. Full audit trail functionality—including operator ID, timestamped parameter changes, and session logs—is enabled via firmware and supports FDA 21 CFR Part 11 readiness when integrated with validated LIMS platforms.
Software & Data Management
While the TDZ4-WS operates as a standalone instrument, its embedded firmware supports GLP-aligned data integrity practices: all method executions are logged with immutable timestamps, user identifiers (via optional password-protected login), and checksum-verified parameter sets. The display interface provides immediate access to centrifugal force (×g) calculation using real-time speed and rotor radius—eliminating manual conversion errors. Twenty customizable protocols can be saved with descriptive naming (e.g., “PBMC Isolation – 22°C”), recalled instantly, and exported via USB port (optional firmware update) for external validation documentation. No proprietary software installation is required for basic operation; however, optional CSV export capability facilitates integration into electronic lab notebooks (ELN) and QA/QC reporting workflows.
Applications
- Clinical hematology: Separation of whole blood into plasma, buffy coat, and erythrocyte fractions prior to automated cell counting or coagulation assays.
- Pharmaceutical QC: Clarification of suspension formulations, recovery of active pharmaceutical ingredients (APIs) from reaction mixtures, and residual solvent removal.
- Food microbiology: Concentration of pathogens (e.g., Salmonella, Listeria) from enrichment broths prior to plating or molecular detection.
- Environmental testing: Sedimentation of suspended solids in wastewater or soil leachates for turbidity assessment or metal speciation analysis.
- Academic research: Routine isolation of nuclei, mitochondria, or membrane vesicles using differential centrifugation protocols within defined RCF ranges.
FAQ
What is the maximum relative centrifugal force (RCF) achievable with the TDZ4-WS?
The instrument achieves up to 2650 ×g at its rated maximum speed of 4000 rpm with the 4 × 100 mL angular rotor.
Does the TDZ4-WS support continuous operation mode?
Yes—it offers three run modes: timed (1 min to 99 h 59 min), continuous, and short-spin (hold until manually stopped).
Is rotor calibration required before first use?
No factory-calibrated rotors are supplied; however, users must verify rotor identification and load symmetry prior to each run per ISO 21501-4 guidelines.
Can the TDZ4-WS be validated for GMP environments?
Yes—the combination of ISO 13485 certification, audit-trail logging, and configurable user access controls enables IQ/OQ/PQ protocol development in alignment with Annex 11 and EU GMP Chapter 4.
What maintenance intervals are recommended?
Routine cleaning of the rotor chamber and gasket every 2 weeks is advised; annual inspection of motor bearings and door lock actuator is recommended for labs operating >8 h/day.