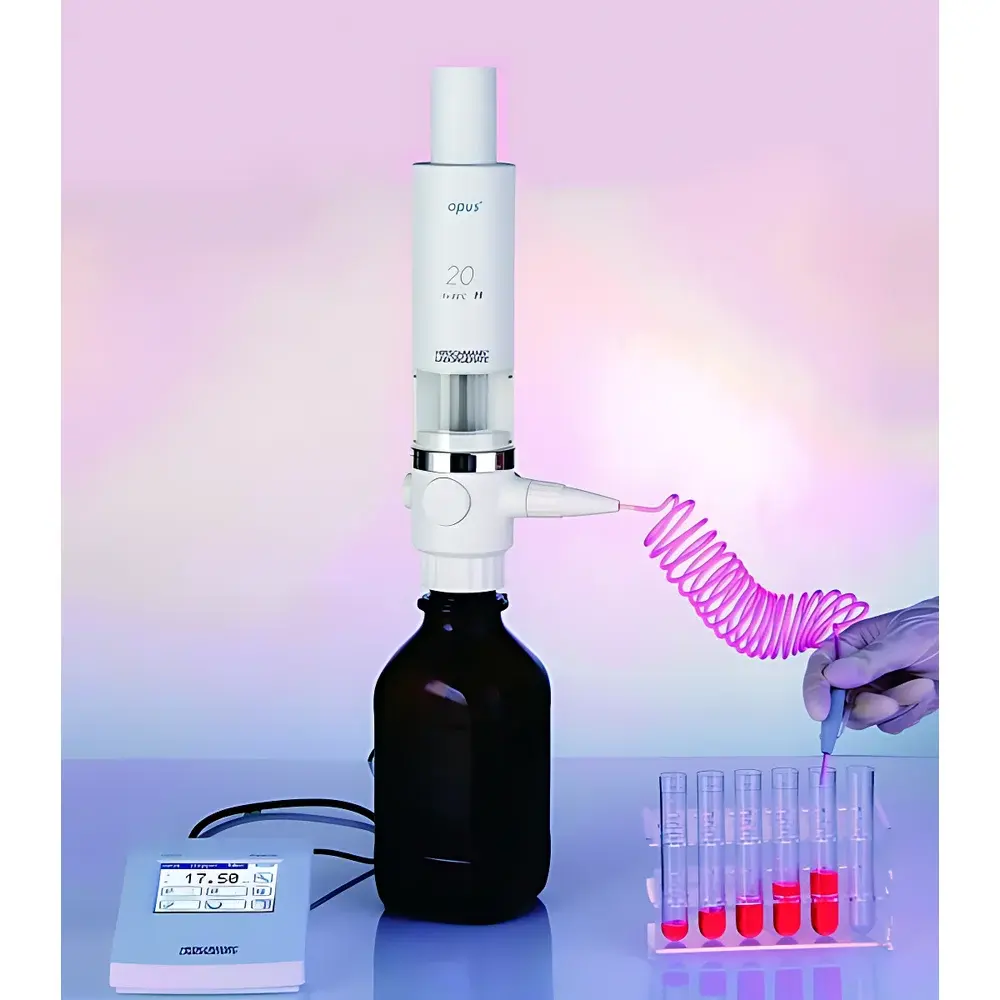
Hirschmann Opus Electronic Burette Model 9581020
| Brand | Hirschmann (Germany) |
|---|---|
| Origin | Germany |
| Model | 9581020 |
| Compliance | ISO, DIN EN ISO 9000, GLP, GMP |
| Display | TFT Touchscreen |
| Control Options | Touchscreen, External Mouse, Foot Switch, PC Interface (RS232/USB) |
| Fluid Path Materials | ECTFE, FEP, PTFE, Borosilicate Glass |
| Max. Volume | 20 mL |
| Minimum Graduation | 0.01 mL |
| Accuracy (A) | ≤0.2% |
| Coefficient of Variation (CV) | ≤0.07% |
| Resolution | 10 µL |
| Optional Accessories | Desiccant Drying Tube, Amber Viewing Window, Integrated Safety Valve |
| Calibration | Individual unit calibration with traceable serial-numbered QC certificate |
Overview
The Hirschmann Opus Electronic Burette Model 9581020 is a precision-engineered, motor-driven burette designed for high-reproducibility volumetric titration in regulated laboratory and manufacturing environments. Unlike manual or semi-automated burettes, the Opus employs a closed-loop stepper motor system coupled with optical position sensing to deliver precise, repeatable liquid dispensing without operator-dependent mechanical variables. Its measurement principle is based on displacement-controlled piston actuation within a chemically inert fluid path—ensuring volumetric accuracy independent of liquid density, viscosity, or surface tension effects typical in gravity-fed systems. The device operates under ISO 8655-3 (volumetric apparatus — burettes) and conforms to DIN EN ISO 9001 quality management requirements, making it suitable for use in GLP-compliant research, GMP-controlled QC labs, and pharmaceutical process validation workflows where auditability and traceability are mandatory.
Key Features
- Motor-driven dispensing with 10 µL resolution and ≤0.2% accuracy (A) per ISO 8655-3, validated at 20 mL nominal volume
- TFT touchscreen interface with intuitive icon-based navigation; supports multi-language UI (English, German, French, Spanish)
- Three dispensing modes: rapid fill, controlled slow-drip, and pre-titration approach—each programmable and recallable via user-defined method storage (up to 9 methods)
- Integrated safety valve prevents over-pressurization and accidental reagent backflow; optional desiccant drying tube maintains anhydrous conditions for moisture-sensitive titrants
- Modular fluid path constructed entirely from fluoropolymers (ECTFE, FEP, PTFE) and borosilicate glass—chemically resistant to all common titrants except hydrofluoric acid
- Amber viewing window option enables light-sensitive titration (e.g., iodometric, cerimetric, or photolabile redox systems)
- Air purge and auto-prime function eliminates bubble formation and ensures consistent meniscus positioning prior to dispensing
Sample Compatibility & Compliance
The Opus 9581020 accommodates aqueous, organic, and mixed-phase titrants—including strong acids (HCl, H₂SO₄), bases (NaOH, KOH), chelating agents (EDTA), oxidizing solutions (KMnO₄, KBrO₃), and non-aqueous reagents (perchloric acid in glacial acetic acid). Its inert fluid path eliminates adsorption, leaching, or catalytic interference during prolonged use. Each unit undergoes individual volumetric calibration against NIST-traceable standards, and ships with a serialized Certificate of Conformance compliant with ISO/IEC 17025 documentation practices. Full audit trails—including date/time stamps, operator ID, method parameters, and raw dispense logs—are retained locally and exportable for FDA 21 CFR Part 11–aligned electronic record systems when connected to validated LIMS or ELN platforms.
Software & Data Management
Data acquisition is supported via RS232 or USB interface using Hirschmann’s proprietary Opus Control Suite (Windows-compatible), which enables remote operation, method scripting, real-time dispense monitoring, and CSV/PDF report generation. All dispense events—including start/stop timestamps, volume delivered, motor step count, and error flags—are logged with immutable metadata. The software supports configurable user roles, electronic signatures, and automatic backup to network drives—facilitating alignment with ALCOA+ data integrity principles. Exported datasets include ISO 17025–compliant uncertainty budgets derived from factory calibration data and in-use verification records.
Applications
- Pharmaceutical assay validation (USP , EP 2.2.20) requiring strict volumetric repeatability
- Food & beverage acidity testing (AOAC 941.15, ISO 750) under routine QC protocols
- Environmental water analysis (EPA 300.0, ISO 9963-1) for chloride, hardness, and alkalinity titrations
- Chemical raw material certification where batch release depends on titrimetric purity assessment
- Academic teaching labs requiring robust, low-maintenance instrumentation with minimal training overhead
- Process analytical technology (PAT) integration in continuous manufacturing lines via OPC UA or Modbus TCP gateway adapters (optional)
FAQ
Does the Opus 9581020 support 21 CFR Part 11 compliance?
Yes—when used with validated Opus Control Suite v3.2+ and configured with electronic signature workflows, audit trail logging, and role-based access control.
Can the burette be calibrated in-house?
Yes—users may perform routine performance verification using certified reference weights and gravimetric procedures per ISO 8655-6; full recalibration requires return to an authorized Hirschmann service center.
Is the amber window compatible with all models?
The amber viewing window is an optional retrofit kit (Order No. 9581020-AMBER) and installs without tools on any Opus 9581020 unit.
What maintenance is required beyond periodic cleaning?
None—the brushless stepper motor and sealed optical encoder require no lubrication or adjustment; only routine rinsing of the fluid path and visual inspection of seals are recommended per DIN EN ISO 15195 guidelines.
How is data integrity ensured during power loss?
Internal non-volatile memory retains all method configurations and last 500 dispense events; unsaved real-time logs are buffered and written upon safe shutdown or recovery.