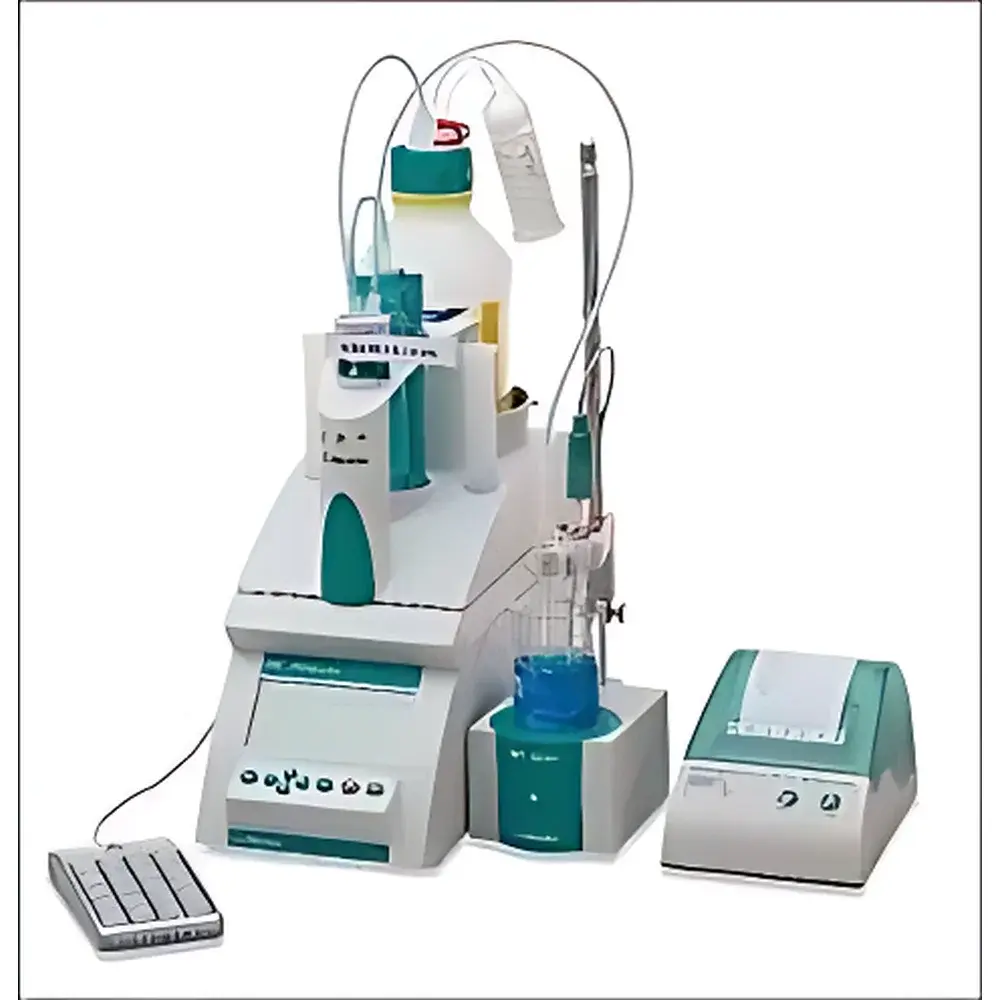
Metrohm 848 Titrino Plus Automatic Potentiometric Titrator
| Brand | Metrohm |
|---|---|
| Origin | Switzerland |
| Model | 848 |
| Titration Principle | Potentiometric Endpoint Detection |
| Burette Resolution | 1/10,000 |
| Input Signal Resolution | High-resolution analog-to-digital conversion |
| Control Interface | Keyboard & USB Mouse |
| Display | Integrated high-contrast LCD with real-time titration curve plotting |
| Stirring Options | Compatible with 801 Magnetic Stirrer or 802 Spiral Stirrer |
| Compliance | GLP/GMP-compliant data handling, ASTM E202–22, ISO 8655-3, USP <541>, FDA 21 CFR Part 11 ready (with optional software modules) |
| Connectivity | RS232, USB Host (for printer, keyboard, mouse, barcode scanner, USB storage), Remote I/O for autosamplers (e.g., 869 Compact Sample Processor) |
| Software Support | tiamo™ PC software for method development, data acquisition, audit trail, electronic signatures, and LIMS integration |
| Measurement Modes | SET (set endpoint), MET (equivalence point), DET (dynamic titration), pH/mV/T measurement, 5-point pH calibration |
| Optional Modules | Photometric titration (523 nm / 610 nm), Polarographic titration (Ipol/Upol) |
Overview
The Metrohm 848 Titrino Plus Automatic Potentiometric Titrator is a benchtop electrochemical analysis system engineered for precision, repeatability, and regulatory compliance in routine and quality-controlled laboratory environments. It operates on the fundamental principle of potentiometric titration—measuring the potential difference between a reference electrode and an indicator electrode as titrant is incrementally added to the analyte solution. The inflection point of the resulting E–V (potential vs. volume) curve is used to determine the equivalence point with high fidelity. Designed and manufactured in Herisau, Switzerland, the instrument integrates Metrohm’s legacy of electrode metrology and fluidic engineering into a modular, user-accessible platform. Its architecture supports both classical volumetric titrations and advanced modes including photometric and polarographic endpoints, enabling method flexibility without hardware reconfiguration.
Key Features
- High-resolution burette drive delivering 1/10,000 resolution (0.001 mL increments for a 10 mL burette), ensuring precise reagent delivery across wide dynamic ranges.
- Smart exchange units with embedded EEPROM chips storing calibration history, reagent lot information, and maintenance logs—enabling full traceability per ISO 8655-3 and GLP requirements.
- Real-time graphical display of titration curves directly on the built-in LCD screen, with simultaneous numeric readouts of pH, mV, temperature, and derivative dE/dV.
- Dual stirring compatibility: seamless integration with the 801 magnetic stirrer (for standard aqueous applications) or the 802 spiral stirrer (optimized for viscous, heterogeneous, or low-volume samples).
- Full USB host capability supporting peripheral devices—including thermal printers, barcode scanners, external keyboards/mice, and USB mass storage for immediate method/result archiving.
- Remote interface (TTL-level digital I/O) for synchronization with third-party sample processors such as the 869 Compact Sample Processor, enabling unattended batch analysis.
Sample Compatibility & Compliance
The 848 Titrino Plus accommodates a broad spectrum of sample matrices—aqueous, non-aqueous, turbid, suspended, and temperature-sensitive solutions—without requiring specialized cell modifications. Its electrode input stage supports all common Metrohm pH, redox, ion-selective, and polarographic sensors, as well as third-party electrodes meeting DIN 19262 or ASTM E202–22 electrical specifications. All data acquisition, storage, and reporting functions comply with Good Laboratory Practice (GLP) and Good Manufacturing Practice (GMP) frameworks. When paired with tiamo™ software and appropriate configuration, the system meets FDA 21 CFR Part 11 requirements for electronic records and signatures—including time-stamped audit trails, role-based access control, and immutable result archiving. Calibration records, method parameters, and raw titration curves are stored with metadata (operator ID, timestamp, instrument ID, electrode serial number) to satisfy ISO/IEC 17025 accreditation criteria.
Software & Data Management
Method development, execution, and reporting are managed via tiamo™ PC software—a validated, multi-user platform supporting method templates, parameter validation rules, and automated report generation in PDF, CSV, or XML formats. Raw data files (.TMD) retain full waveform fidelity and are digitally signed upon export. USB storage integration allows direct saving of methods, calibration reports, and individual titration results—including derivative plots and equivalence point calculations—without network dependency. For enterprise deployment, tiamo™ supports LIMS connectivity through ASTM E1384 or HL7-compatible interfaces, enabling bidirectional sample ID transfer and result ingestion. All software modules undergo periodic verification per ICH Q2(R2) guidelines for analytical procedure validation.
Applications
The 848 Titrino Plus performs standardized and custom titrations across pharmaceutical, food & beverage, environmental, and industrial chemistry workflows. Validated applications include acid-base titrations (e.g., total acidity in wine or free fatty acids in oils), redox determinations (e.g., iodine value in fats per AOAC 920.112, Fe²⁺ content in pharmaceutical intermediates), precipitation titrations (e.g., chloride in drinking water per ISO 9297), complexometric assays (e.g., Ca²⁺/Mg²⁺ hardness per ISO 6059), and photometric endpoints (e.g., dichromate-based COD analysis at 610 nm). Polarographic titration modes support trace metal speciation using dropping mercury or solid-state polarographic electrodes under controlled potential conditions.
FAQ
What electrode types are compatible with the 848 Titrino Plus?
It accepts all Metrohm pH, redox, ion-selective, and polarographic electrodes with standard BNC or S7 connectors, as well as third-party electrodes conforming to DIN 19262 voltage output specifications.
Can the instrument be integrated into a regulated QC environment?
Yes—when configured with tiamo™ software, electronic signature modules, and audit-trail-enabled USB storage, it satisfies FDA 21 CFR Part 11, EU Annex 11, and ISO/IEC 17025 documentation requirements.
Is non-aqueous titration supported out-of-the-box?
Yes—the system includes preconfigured methods for common non-aqueous media (e.g., glacial acetic acid, dimethylformamide) and supports temperature-compensated endpoint detection down to 5 °C.
How is burette calibration verified?
Calibration is performed using gravimetric procedures per ISO 8655-3; smart exchange units store calibration coefficients and expiration dates, which are automatically loaded during installation.
What sample throughput can be achieved with automation?
When coupled with the 869 Compact Sample Processor (60-position capacity), the system achieves fully unattended analysis of up to 45 samples per hour, depending on titration kinetics and endpoint stability.