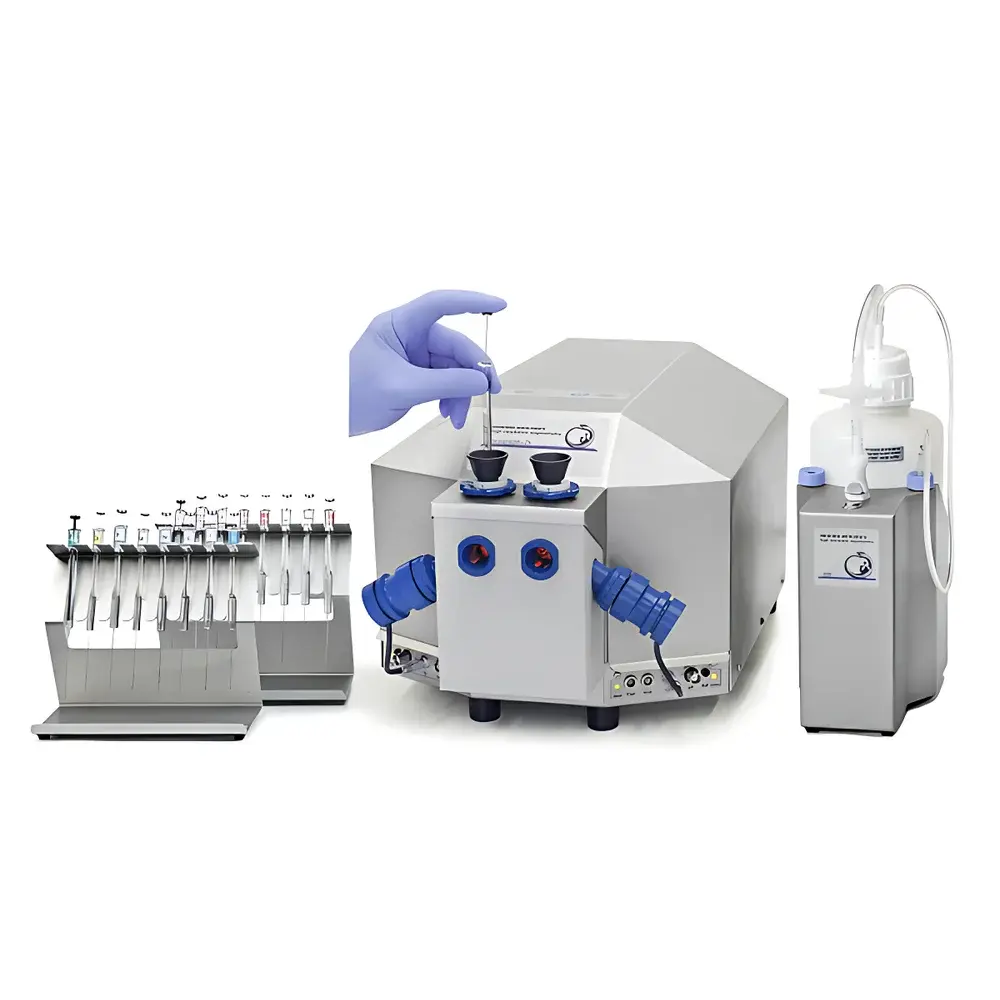
OROBOROS O2K High-Resolution Respirometry System
| Brand | OROBOROS Instruments |
|---|---|
| Origin | Austria |
| Model | O2K |
| Temperature Range | 4–47 °C |
| Chamber Volume | 1.5–3.5 mL |
| Oxygen Resolution | 1 pmol O₂·s⁻¹·mL⁻¹ |
Overview
The OROBOROS O2K High-Resolution Respirometry System is a dual-chamber, modular platform engineered for quantitative analysis of mitochondrial respiratory function and cellular bioenergetics in real time. Based on Clark-type polarographic oxygen sensors with sub-picomolar resolution (1 pmol O₂·s⁻¹·mL⁻¹), the O2K measures oxygen flux (JO₂) as the primary thermodynamic variable—directly reflecting electron transfer system (ETS) activity across physiological oxygen partial pressures (pO₂). Unlike indirect assays or endpoint measurements, the O2K enables continuous, label-free monitoring of substrate-uncoupler-inhibitor titration (SUIT) protocols, supporting mechanistic dissection of oxidative phosphorylation (OXPHOS), proton leak, non-mitochondrial oxygen consumption, and redox-coupled processes. Its open-system architecture allows full reagent flexibility, eliminating proprietary consumables while maintaining strict environmental control (temperature, stirring, pO₂, pH) within chemically inert Duran glass and titanium chambers. Designed for compliance with international standards for preclinical and translational research—including ISO/IEC 17025, CLSI EP26-A, and FDA 21 CFR Part 11 requirements for electronic records—the O2K serves as the de facto reference instrument in mitochondrial physiology, pharmacology, toxicology, and metabolic disease modeling.
Key Features
- Dual independent respirometry chambers (1.5–3.5 mL volume), each equipped with high-stability polarographic oxygen sensors calibrated in real time against air-saturated and anoxic reference solutions
- Integrated fluorescence detection module supporting simultaneous measurement of mitochondrial membrane potential (ΔΨm), H₂O₂, ATP, Ca²⁺, Mg²⁺, NAD(P)H, and NO using excitation/emission filters compatible with common fluorophores (e.g., safranine, Amplex Red, Calcium Green)
- TIP2k microtitration pumps enabling precise, programmable injection and aspiration (±0.1 µL accuracy) with up to 15 sequential titrations per chamber—fully synchronized with oxygen and fluorescence acquisition
- Active temperature control via Peltier-based heating/cooling modules (4–47 °C), achieving ±0.02 °C stability over extended experiments
- Magnetically driven stirrer ensuring homogeneous sample suspension and eliminating diffusion gradients—critical for accurate kinetic assessment of permeabilized tissues and isolated mitochondria
- Modular hardware expansion capability: optional modules include NO electrode, pH electrode, H₂S sensor, and photo-biology add-ons (O2k-PhotoBiology) for light-controlled respiration studies
Sample Compatibility & Compliance
The O2K accommodates a broad spectrum of biological samples without labeling or genetic modification: intact cells (adherent/suspension), permeabilized fibers, isolated mitochondria, tissue homogenates (e.g., skeletal muscle, heart, liver), plant protoplasts, yeast, bacteria, and even small invertebrates. Its low-background chamber design minimizes autoxidation artifacts, enabling reliable measurements under hypoxic (pO₂ < 1 kPa), normoxic (21 kPa), or hyperoxic conditions. All hardware and software components comply with CE marking requirements and support Good Laboratory Practice (GLP) workflows: DatLab generates timestamped, tamper-evident audit trails; user access levels are configurable; electronic signatures and protocol versioning meet 21 CFR Part 11 criteria for regulated environments. Instrument qualification documentation (IQ/OQ/PQ) templates are provided for GMP-aligned laboratories.
Software & Data Management
DatLab software (v8.x) is the central analytical engine—developed in-house by Oroboros Instruments and continuously refined through peer-reviewed methodological publications. It provides live visualization of all parameters (JO₂, fluorescence intensity, temperature, stirrer speed), automatic background correction, and dynamic calibration routines (oxygen solubility, sensor slope, fluorescence quenching). The Protocol Editor supports drag-and-drop experimental design, including conditional logic (e.g., “inject FCCP when JO₂ drops below X nmol·s⁻¹·mL⁻¹”) and iterative titration loops. Raw data are stored in open-format .dat files (XML-based), fully exportable to CSV, MATLAB, or Python for secondary analysis. Built-in analysis tools compute key bioenergetic indices: LEAK respiration, OXPHOS capacity, ETS capacity, respiratory control ratio (RCR), coupling efficiency, and flux control ratios (FCR)—all traceable to original SUIT protocols.
Applications
The O2K is routinely applied in academic and industrial settings to investigate: mitochondrial dysfunction in neurodegeneration (Parkinson’s, Alzheimer’s), metabolic inflexibility in type 2 diabetes and obesity, cardioprotection mechanisms following ischemia-reperfusion injury, drug-induced mitochondrial toxicity (e.g., antiretrovirals, chemotherapeutics), cancer cell metabolic reprogramming (Warburg effect vs. reverse Warburg), aging-related bioenergetic decline, and environmental stress responses in plants and aquatic organisms. Published studies leveraging O2K data appear in high-impact journals including Nature Metabolism, Cell Metabolism, EMBO Journal, and Journal of Clinical Investigation. Its adaptability to standardized SUIT protocols ensures inter-laboratory reproducibility—a cornerstone for multi-center clinical trials evaluating mitochondrial biomarkers.
FAQ
What sample types can be measured directly in the O2K?
Intact cells, permeabilized tissue fibers, isolated mitochondria, bacterial cultures, plant leaves or roots (as discs), and small invertebrate tissues—all without fluorescent labels or genetic engineering.
Does the O2K require proprietary consumables?
No. The system uses standard reagents, glass syringes, and reusable chambers. Oxygen sensors are field-calibratable and have multi-year lifespans.
How does the O2K ensure measurement reproducibility across labs?
Through standardized SUIT protocols, certified calibration procedures, open-data formats, and participation in international quality assurance programs (e.g., Mitochondrial Physiology Network, MitoEAGLE).
Can DatLab software be validated for use in regulated environments?
Yes. Oroboros provides validation packages including IQ/OQ documentation, test scripts, and 21 CFR Part 11 configuration guides for audit-ready deployment.
Is technical support available outside Europe?
Oroboros maintains regional application specialists and certified service engineers across North America, Asia-Pacific, and Latin America, with remote diagnostics and on-site training options.