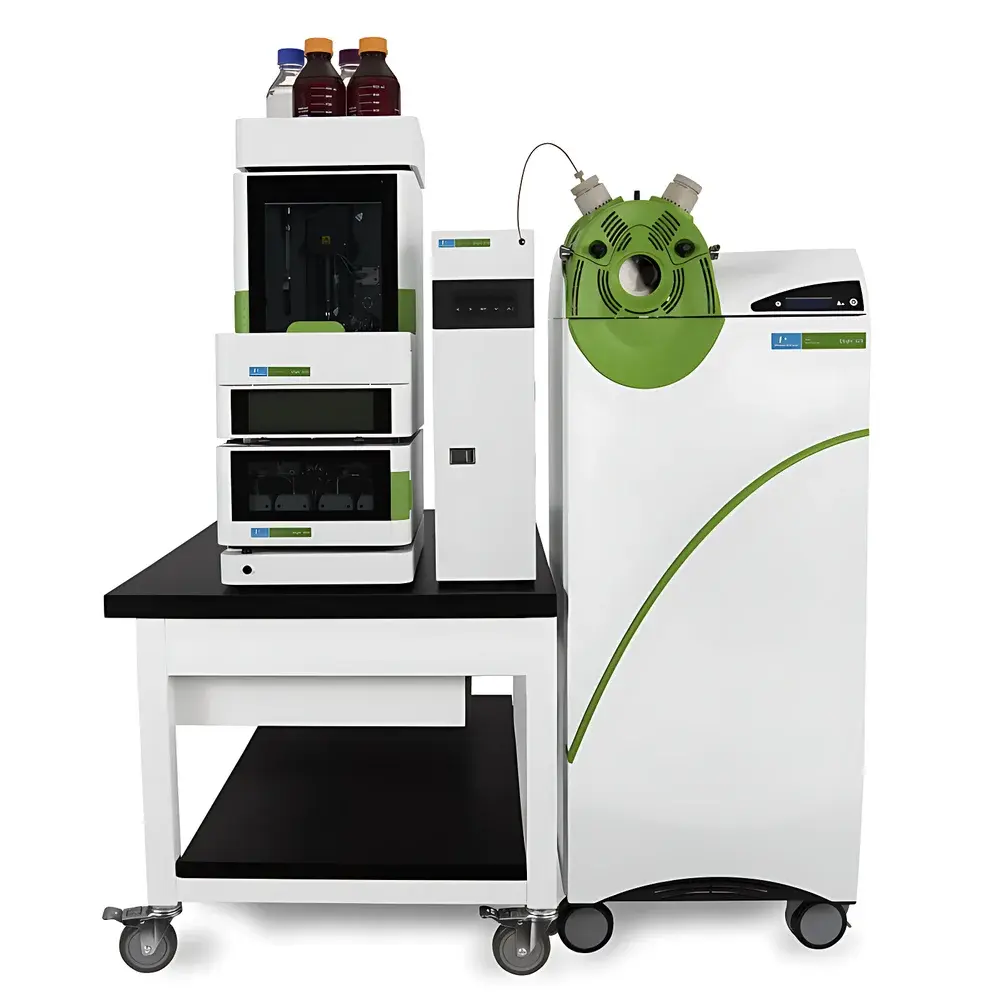
PerkinElmer QSight 400 Triple Quadrupole Liquid Chromatography Mass Spectrometer
| Brand | PerkinElmer |
|---|---|
| Origin | Canada |
| Manufacturer Type | Original Equipment Manufacturer (OEM) |
| Import Status | Imported |
| Model | QSight 400 |
| Instrument Type | Triple Quadrupole (TQ) LC-MS/MS |
| Application Scope | General-Purpose |
| Mass Range | Available upon consultation |
| Sensitivity | Available upon consultation |
| Resolution | Available upon consultation |
| Accurate Mass Capability | Not applicable (unit-mass resolution TQ system) |
Overview
The PerkinElmer QSight 400 is a high-performance triple quadrupole liquid chromatography tandem mass spectrometer (LC-MS/MS) engineered for robust, high-throughput quantitative analysis in regulated and research-intensive laboratories. Unlike high-resolution accurate-mass (HRAM) platforms, the QSight 400 operates as a unit-mass resolution tandem MS system optimized for selected reaction monitoring (SRM) and multiple reaction monitoring (MRM) workflows—delivering exceptional specificity, reproducibility, and linear dynamic range across complex sample matrices. Its architecture integrates seamlessly with the QSight LX50 ultra-high-performance liquid chromatography (UHPLC) system, forming a fully synchronized LC-MS/MS platform designed to meet stringent regulatory requirements in food safety, environmental monitoring, clinical toxicology, and agrochemical residue testing. The system leverages proven ion optics, thermally stabilized vacuum environments, and intelligent source design to ensure long-term signal stability without routine recalibration.
Key Features
- Dual Ionization Source (ESI/APCI): Simultaneous electrospray ionization (ESI) and atmospheric pressure chemical ionization (APCI) capability within a single injection—enabling comprehensive compound coverage without source swapping or method revalidation.
- Hot Surface Induced Desolvation (HSID) Interface: A patented, self-cleaning ion source interface utilizing precisely controlled high-temperature laminar gas flow to enhance desolvation efficiency, reduce chemical noise, and improve signal-to-noise ratio—without mechanical moving parts or consumable components.
- Laminar Flow Ion Transmission Optics: Engineered ion path geometry minimizes ion losses and wall collisions under high-flow conditions, maintaining sensitivity across wide gradient elution profiles and extended run times.
- Unified Field Detector (UFD): A rugged, low-noise electron multiplier detector with extended dynamic range (>6 orders), enabling quantitation from sub-pg/mL to µg/mL levels in a single acquisition.
- Modular UHPLC Integration: Direct coupling with the QSight LX50 UHPLC system—including integrated column oven, low-dead-volume valve switching, and real-time pressure monitoring—ensures retention time stability and method transferability.
- Maintenance-Free Operation: Designed for >95% instrument uptime; typical maintenance intervals exceed 72 hours of continuous operation before scheduled cleaning—reducing downtime and technician dependency.
Sample Compatibility & Compliance
The QSight 400 demonstrates broad compatibility with diverse biological, environmental, and food-derived matrices—including plasma, urine, soil extracts, water concentrates, fruit homogenates, and cereal digests—without requiring extensive sample cleanup. Its MRM-based selectivity enables reliable quantification even in high-background samples where isobaric interferences are common. The system supports full compliance with internationally recognized analytical standards, including ISO/IEC 17025:2017 method validation requirements, FDA Bioanalytical Method Validation Guidance (2018), EPA Methods 1694 and 8321B, and EU Commission Regulation (EU) No 2021/808 for pesticide residue analysis. Data integrity is ensured via software audit trails compliant with 21 CFR Part 11 and Annex 11 (GxP environments), with configurable user roles, electronic signatures, and immutable raw data archiving.
Software & Data Management
Operation is managed through the unified QSight Software Suite—a modular, workflow-driven platform supporting method development, acquisition control, quantitative processing, and report generation in a single interface. Pre-loaded application-specific method libraries—including >200 validated MRM transitions for mycotoxins, pharmaceuticals, pesticides, and veterinary drugs—accelerate method deployment and reduce qualification effort. All methods include built-in calibration curve fitting (linear/log-linear with weighting), internal standard normalization, and QC flagging per ICH Q2(R2) guidelines. Raw data files (.qdb) are stored in an encrypted, timestamped format with full metadata traceability. Export options include CSV, PDF, and XML formats compatible with LIMS integration (e.g., LabWare, Thermo SampleManager) and third-party statistical packages.
Applications
The QSight 400 delivers validated performance in multi-residue quantitative workflows across regulated sectors: detection and quantification of veterinary drug residues (e.g., fluoroquinolones, sulfonamides) in meat and milk per EU Maximum Residue Levels (MRLs); screening of emerging contaminants (PFAS, microplastic additives, flame retardants) in wastewater and sediment; targeted metabolite profiling in pharmacokinetic studies; and rapid confirmation of allergenic proteins or adulterants in processed foods. Its dual-source versatility allows simultaneous polar (ESI+) and semi-volatile (APCI+) analyte detection—ideal for labs managing heterogeneous compound portfolios without hardware reconfiguration.
FAQ
Is the QSight 400 capable of high-resolution mass measurement?
No. The QSight 400 is a unit-mass resolution triple quadrupole system optimized for MRM quantitation—not high-resolution accurate-mass analysis. For exact mass determination, PerkinElmer offers the TOF-based Altus Q-TOF platform.
What is the recommended maintenance schedule for the HSID interface?
Under typical operating conditions (≤100 injections/day), no routine cleaning is required. Visual inspection and optional source wipe-down are recommended every 3–6 months, depending on matrix complexity.
Does the system support GLP-compliant audit trail functionality?
Yes. QSight Software includes configurable audit trail settings, electronic signature enforcement, and immutable raw data storage—all aligned with FDA 21 CFR Part 11 and EMA Annex 11 requirements.
Can existing LC methods developed on other vendors’ systems be transferred to the QSight LX50 UHPLC?
Yes. The QSight LX50 supports standard 0.001–5.0 mL/min flow rates, 0.01–1000 bar pressure range, and common column dimensions (2.1 mm × 50–150 mm). Gradient delay volume compensation and dwell time modeling facilitate straightforward method porting.
Is remote monitoring and control supported?
Yes. Secure web-based remote access is available via QSight Connect—a TLS-encrypted interface allowing real-time status monitoring, queue management, and diagnostic logging from authorized devices outside the local network.