Pilotech YC-2100 Pilot-Scale Vacuum Spray Dryer
| Brand | Pilotech |
|---|---|
| Origin | Shanghai, China |
| Manufacturer Type | Direct Manufacturer |
| Country of Origin | China |
| Model | YC-2100 |
| Sample Type | Aqueous Solutions Only |
| Max Feed Rate | 3000 mL/h |
| Temperature Range | 50–200 °C |
| Particle Size Range | 1–100 µm |
| Vacuum Range | −0.05 to −0.08 MPa |
| Heating Accuracy | ±1 °C (PID Controlled) |
| Nozzle Standard Orifice | 2.0 mm (Optional: 0.5 / 0.8 / 1.0 / 1.5 mm) |
| Minimum Sample Volume | 50 mL (varies with solids content) |
| Power Supply | 9 kW / 380 V / 3-phase 5-wire |
| Construction | Full 316L Stainless Steel |
| Control Interface | Color LCD Touchscreen (Chinese UI) |
Overview
The Pilotech YC-2100 Pilot-Scale Vacuum Spray Dryer is an engineered solution for laboratory-to-pilot transition of thermolabile, oxidation-sensitive, and high-value aqueous formulations. Unlike conventional atmospheric spray dryers, the YC-2100 operates under controlled vacuum conditions (−0.05 to −0.08 MPa), enabling rapid solvent evaporation at significantly reduced thermal load. This vacuum-assisted drying mechanism leverages the principle that lowering ambient pressure depresses the boiling point of water and other volatile solvents—allowing complete drying at inlet air temperatures as low as 50 °C while maintaining throughput up to 3000 mL/h. The system employs a two-fluid (air-liquid) nozzle design with concentric geometry to ensure symmetrical atomization, eliminating wall impingement and improving powder yield uniformity. Its fully stainless-steel (316L) construction meets hygienic design standards for pharmaceutical, nutraceutical, and biotechnology applications where material traceability and cleanability are critical.
Key Features
- Vacuum-enabled low-temperature drying: Enables processing of heat-sensitive biomolecules (e.g., enzymes, peptides, probiotics), polysaccharides, and amorphous sugar matrices without degradation or Maillard browning.
- High-precision PID temperature control: Real-time inlet air temperature regulation with ±1 °C accuracy across the full 50–200 °C operating range—critical for method transfer and process validation.
- Adjustable concentric two-fluid nozzle: Standard 2.0 mm orifice; optional nozzles (0.5, 0.8, 1.0, 1.5 mm) support optimization of droplet size distribution and final particle morphology.
- Vertical nozzle positioning adjustment: Allows fine-tuning of spray trajectory relative to the drying chamber axis—improving residence time distribution and reducing wet-wall deposition.
- Integrated noise-dampening architecture: Engineered mounting and acoustic isolation for vacuum pump and compressed air system reduce operational noise to ≤65 dB(A) at 1 m distance.
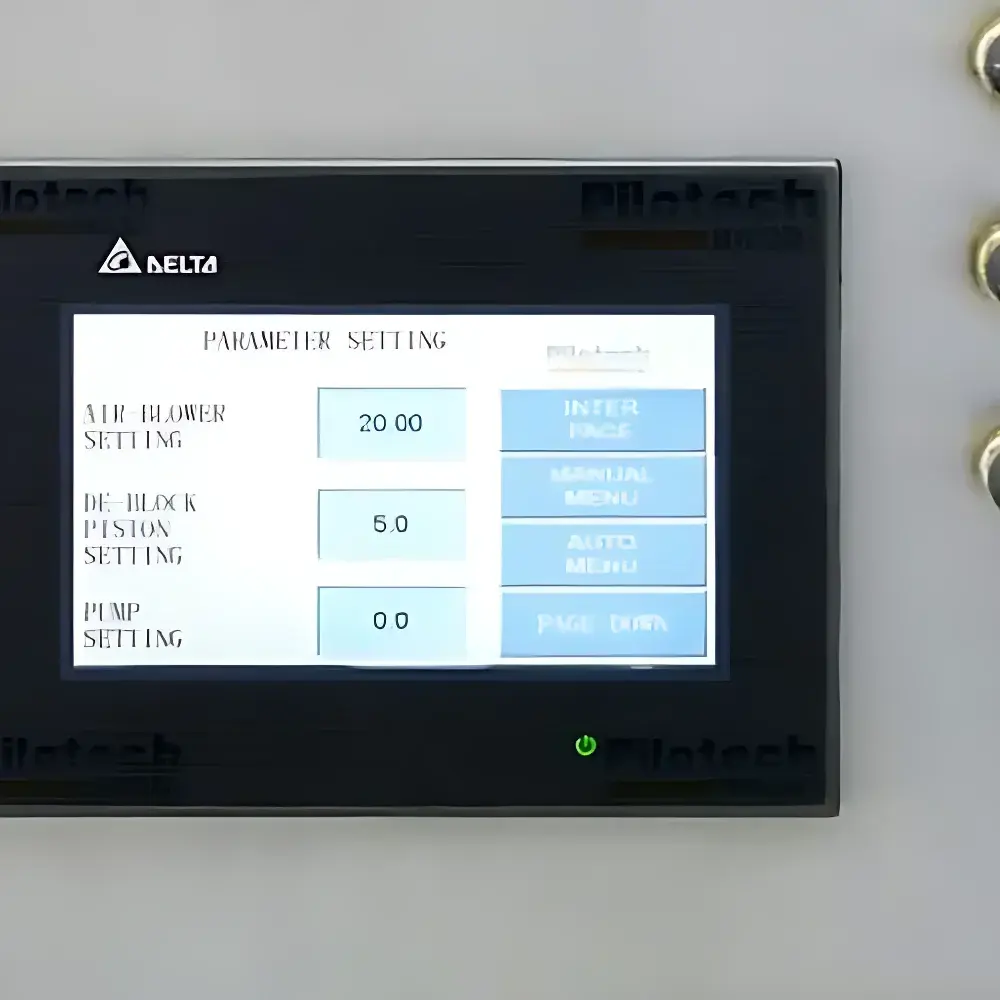
- Full-touch color LCD interface: Intuitive Chinese-language HMI with real-time display of inlet/outlet temperature, vacuum level, feed rate, and system status—designed for GLP-compliant operation in R&D labs.
Sample Compatibility & Compliance
The YC-2100 is validated for aqueous-based feedstocks only—including protein solutions, colloidal suspensions, polymer emulsions, and botanical extracts—with total solids content typically ranging from 2% to 30% w/w. It is not suitable for organic solvents or high-viscosity non-aqueous systems. The unit complies with CE mechanical safety directives (2006/42/EC) and conforms to ISO 8573-1:2010 for compressed air purity when used with certified oil-free air sources. While the touchscreen interface supports audit-ready parameter logging, full 21 CFR Part 11 compliance requires external data acquisition software and user-defined electronic signature protocols. All wetted parts meet ASME BPE surface finish specifications (Ra ≤ 0.8 µm), supporting cleaning validation per ASTM E3012-16.
Software & Data Management
The embedded controller records time-stamped operational parameters—including inlet temperature, outlet temperature, vacuum pressure, feed pump RPM, and system alarms—at 1-second intervals. Data export is supported via USB flash drive in CSV format for post-processing in MATLAB, JMP, or Python-based statistical analysis tools. Though the native interface does not include cloud synchronization or multi-user role management, the system architecture permits integration with LIMS via Modbus RTU (RS-485) for centralized batch record archiving. Process methods can be saved and recalled with version timestamps, supporting reproducible experimental design under ICH Q5C and Q8(R2) guidelines.
Applications
- Pharmaceutical development: Lyophilization precursor preparation, inhalable dry powder formulation (DPI), and nanoparticle carrier drying.
- Food & nutrition: Encapsulation of omega-3 oils, probiotic powders, anthocyanin-rich fruit extracts, and vitamin-fortified dairy matrices.
- Biotechnology: Recovery of labile monoclonal antibody fragments, viral vector intermediates, and CRISPR-Cas9 ribonucleoprotein complexes.
- Materials science: Synthesis of metal-organic framework (MOF) precursors, ceramic nanopowders, and conductive polymer microspheres.
- Academic research: Fundamental studies on droplet evaporation kinetics, glass transition behavior of amorphous solids, and scale-up correlation between lab-scale and production spray dryers.
FAQ
Can the YC-2100 handle organic solvents such as ethanol or acetone?
No. The system is designed exclusively for aqueous feed solutions. Organic solvents require explosion-proof construction, inert gas purging, and specialized condensate recovery—features not integrated into this model.
What is the typical residual moisture content of dried powder?
Under optimized vacuum and temperature conditions (e.g., 50 °C inlet, −0.07 MPa), residual moisture is consistently ≤1.0% w/w for standard carbohydrate- and protein-based feeds, as verified by Karl Fischer titration (ASTM D6304).
Is GMP documentation available for the YC-2100?
Yes—Factory Acceptance Test (FAT) reports, material traceability certificates (EN 10204 3.1), and 316L weld logbooks are provided upon request. IQ/OQ protocol templates compatible with EU Annex 15 and FDA guidance are also available for customer execution.
How is cleaning validation performed on this system?
The fully drainable, crevice-free chamber design supports rinse sampling per USP . Swab recovery studies for common actives (e.g., bovine serum albumin, trehalose) demonstrate ≥95% removal efficiency after a validated CIP cycle using 0.1 N NaOH at 60 °C for 15 minutes.