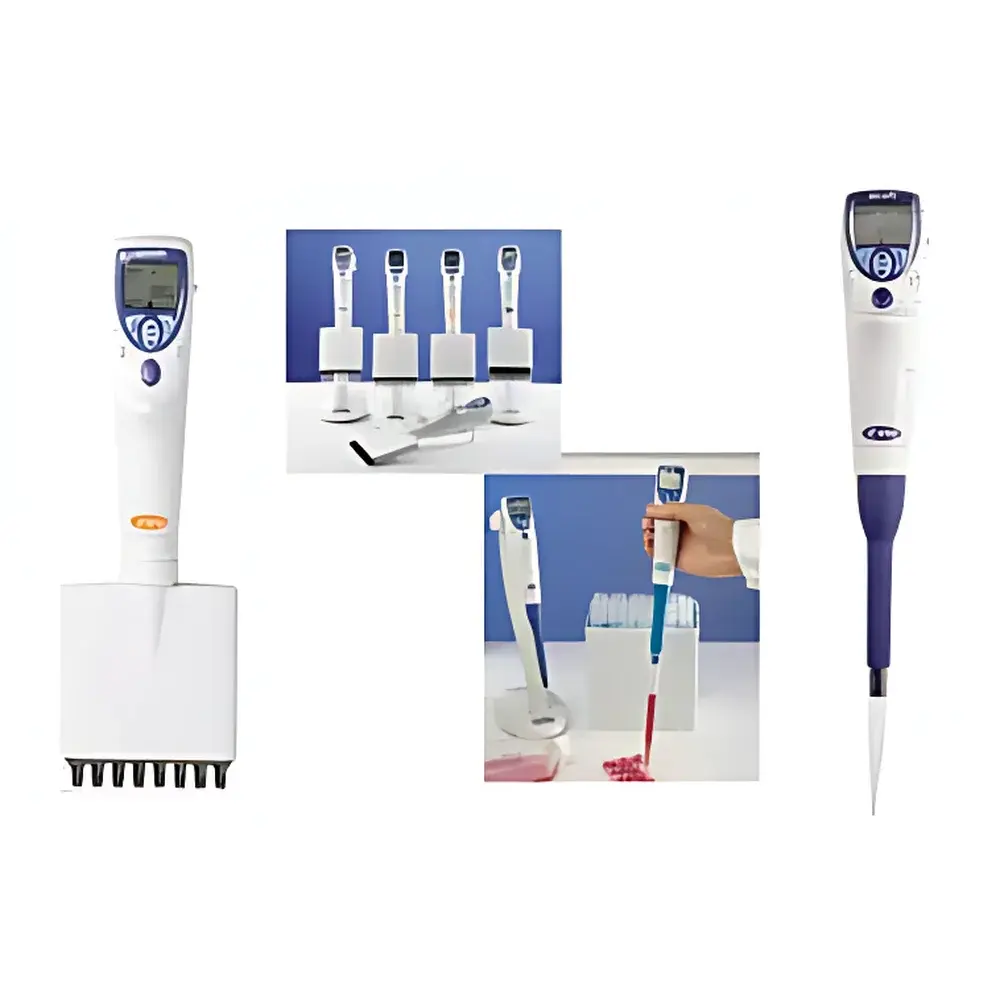
Sartorius eLine Series Electronic Pipettes
| Brand | Sartorius |
|---|---|
| Origin | Finland (Manufactured) |
| Model | eLine |
| Type | Motorized Single-Channel Electronic Pipette |
| Power | Rechargeable Lithium-Ion Battery |
| Display | Backlit LCD with Icon-Based Interface |
| Pipetting Modes | 11 Predefined (e.g., Standard, Reverse, Dilution, Dispense, Repeat, Mix, Variable Volume, Stepwise, Programmed Aspirate/Dispense, Calibration Mode, Error Log Review) |
| User Programs | Up to 6 Custom Protocols with Audit Trail |
| Tip Ejection | Motorized, One-Touch Electronic Eject |
| Charging Options | Dedicated eLine Charging Station (Docking Cradle) or Optional AC Adapter (Fast Charge Support) |
| Ergonomics | Ambidextrous Design with Adjustable Grip Angle and Low Actuation Force |
| Compliance | ISO 8655-2:2022 (Pipettes), ISO 13485:2016 (QMS), CE Marked for IVDD & MDR |
Overview
The Sartorius eLine Series Electronic Pipettes represent a rigorously engineered evolution in precision liquid handling—designed and manufactured in Finland by Sartorius (acquiring the former Biohit eLine platform). These motorized single-channel pipettes operate on closed-loop microprocessor-controlled piston actuation, ensuring consistent volumetric delivery across the full range (0.1–10 mL depending on model variant) without operator-dependent mechanical variability. Unlike manual or semi-automated alternatives, the eLine integrates real-time force feedback, adaptive motor torque regulation, and position-sensing encoders to maintain accuracy and repeatability under varying environmental conditions—including temperature gradients, tip resistance variability, and user fatigue. The system is calibrated per ISO 8655-2:2022 and traceably certified against NIST-traceable standards at point of manufacture. Its architecture supports both routine QC workflows and regulated environments where documentation integrity is critical.
Key Features
- Ambidextrous ergonomic design with adjustable grip angle and low-force activation (<0.8 N trigger force), minimizing repetitive strain injury (RSI) risk during high-throughput operations.
- Backlit graphical LCD interface with intuitive icon-driven navigation—no language dependency; all menus and error states rendered via standardized symbols compliant with IEC 62366-1 usability engineering requirements.
- Eleven built-in pipetting modes including reverse pipetting for viscous or volatile liquids, stepwise dispensing for serial dilutions, mix cycles with programmable speed and duration, and calibration verification mode with integrated self-test diagnostics.
- Motorized tip ejection mechanism eliminates inconsistent mechanical pressure during tip removal—reducing aerosol generation and cross-contamination risk while preserving pipette longevity.
- On-device storage for up to six user-defined protocols, each retaining full parameter history (volume, speed, pause intervals, mode selection) with timestamped execution logs.
- Dual charging architecture: primary docking station enables overnight trickle charging with battery health monitoring; optional AC adapter supports rapid charge (0–100% in ≤90 min) for emergency deployment without workflow interruption.
Sample Compatibility & Compliance
The eLine series accommodates standard conical polypropylene tips (0.1–10 mL range) from major OEMs (including Sartorius, Eppendorf, and Gilson), with automatic tip recognition via mechanical coding not required. All models comply with ISO 8655-2:2022 for gravimetric performance validation and are CE-marked under both In Vitro Diagnostic Medical Device Regulation (IVDR 2017/746) and Medical Device Regulation (MDR 2017/745). Full audit trail functionality—including protocol creation, modification, and execution timestamps—is configurable to meet FDA 21 CFR Part 11 requirements when paired with Sartorius LabX software. The device’s firmware revision history, calibration date, and service log entries are exportable in CSV format for GLP/GMP documentation packages.
Software & Data Management
While fully functional as a standalone instrument, the eLine integrates natively with Sartorius LabX Liquid Handling software (v3.5+). LabX provides centralized protocol deployment, remote firmware updates, electronic signature capture, and automated report generation—including ISO 8655-compliant calibration certificates with uncertainty budgets. All user actions—mode selection, volume entry, program launch, error acknowledgment—are recorded with immutable timestamps and operator ID linkage. Data export adheres to ASTM E2500-18 guidelines for analytical instrument data integrity, supporting 21 CFR Part 11 compliance through role-based access control and encrypted local database storage.
Applications
The eLine platform serves laboratories requiring metrologically robust, auditable liquid handling across diverse domains: molecular biology (qPCR setup, NGS library prep), clinical diagnostics (serum dilution, ELISA plate filling), pharmaceutical QC (dissolution testing, standard curve preparation), and academic research (cell culture passaging, reagent aliquoting). Its reverse pipetting mode ensures reliable handling of glycerol stocks, DMSO solutions, and protein-containing buffers. The programmable repeat dispensing function accelerates microplate-based assays such as kinase activity screening or cytotoxicity profiling—where inter-run consistency directly impacts Z’-factor reliability.
FAQ
Does the eLine require regular recalibration?
Yes—per ISO 8655-5:2022, electronic pipettes must undergo periodic verification (typically every 3–6 months depending on usage frequency and risk assessment). Calibration can be performed in-house using certified weights and distilled water or outsourced to an ISO/IEC 17025-accredited lab.
Can multiple eLine units be synchronized via LabX?
LabX supports concurrent management of up to 32 eLine devices on a single network segment, enabling batch protocol deployment, unified firmware updates, and consolidated audit trail aggregation.
Is tip compatibility limited to Sartorius-branded consumables?
No—the eLine accepts any industry-standard conical tip meeting ISO 8655-3 dimensional tolerances; however, optimal accuracy and tip seal integrity are validated using Sartorius OptiTip or Biohit Finntip consumables.
What happens if the battery depletes mid-pipetting cycle?
The device enters safe-hold mode: remaining volume is retained in memory, and the piston halts in its current position. Upon recharge, the last executed step resumes automatically without data loss.
How is firmware updated?
Firmware updates are delivered via LabX or manually loaded from Sartorius’ secure support portal (requires .hex file import and digital signature verification before installation).