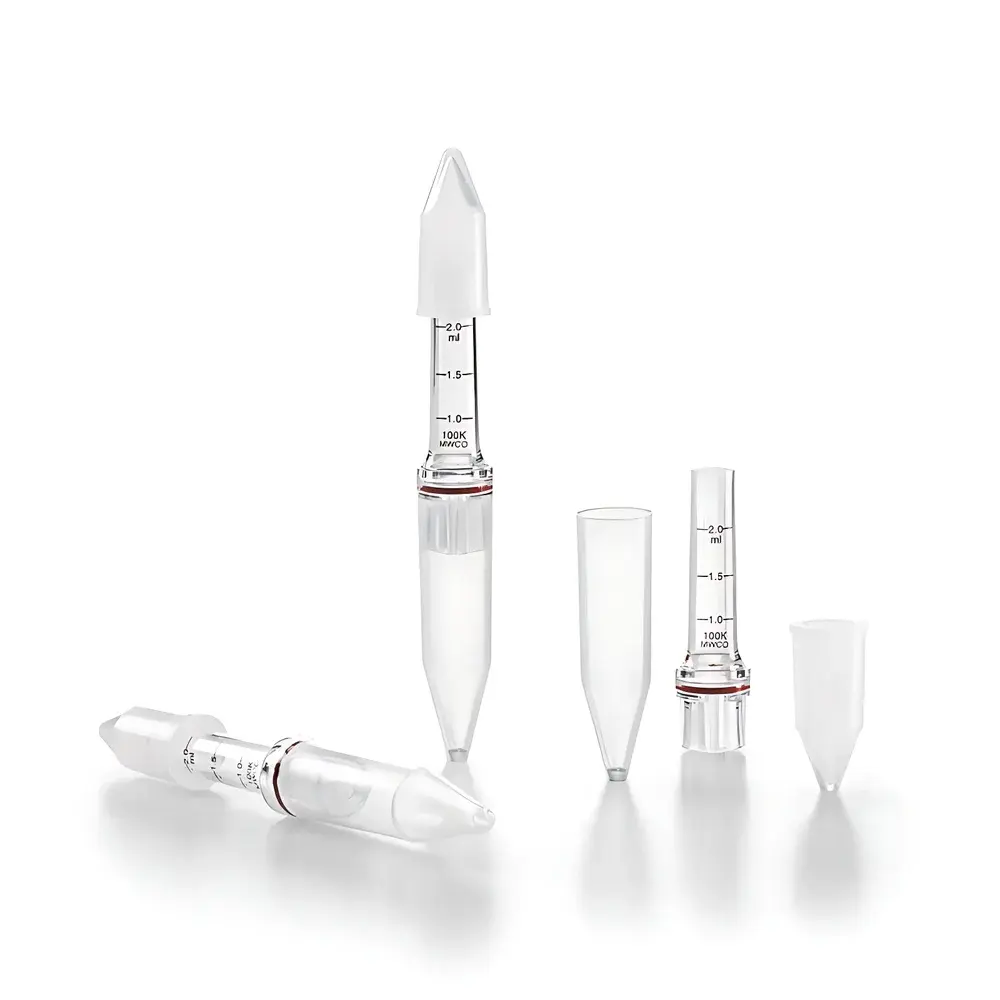
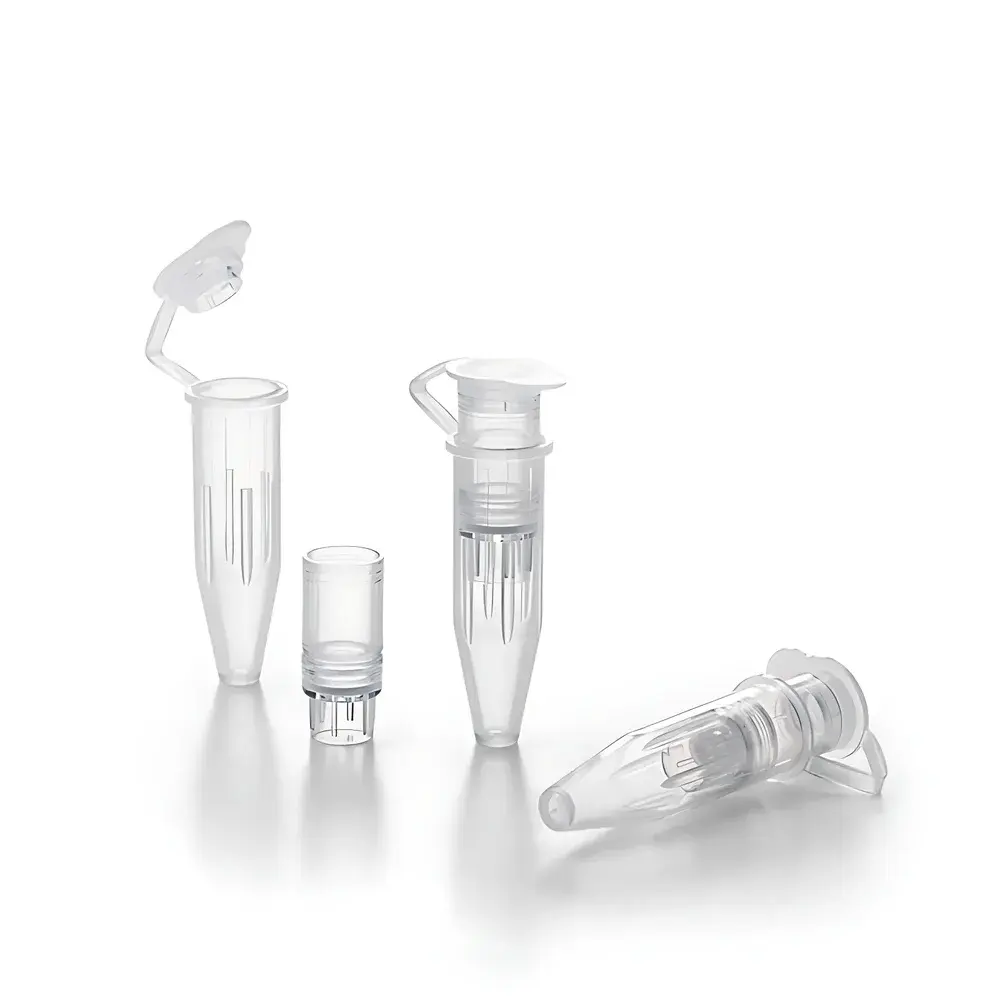
Sartorius Vivacon® 500-PCR and Vivacon® 2-PCR Ultrafiltration Concentrator Devices
| Brand | Sartorius |
|---|---|
| Origin | Germany |
| Product Type | Imported Life Science Consumables |
| Model | Vivacon® 500-PCR / Vivacon® 2-PCR |
| Membrane Material | Hydrosart® regenerated cellulose |
| MWCO Options | 2 kDa, 10 kDa, 30 kDa, 50 kDa, 100 kDa, 125 kDa |
| Sterilization | Ethylene Oxide (ETO) treated for PCR-grade applications |
| Sample Volume | 0.5 mL |
| Max. Centrifugal Force | 3,000–14,000 × g depending on MWCO |
| DNA Recovery (30× conc.) | 85–94% |
| Protein Recovery (30× conc.) | 81–95% |
| Compliance | DNase/RNase-free, endotoxin-tested (<0.5 EU/mL), certified PCR-inhibitor-free |
Overview
The Sartorius Vivacon® 500-PCR and Vivacon® 2-PCR ultrafiltration concentrator devices are engineered for high-efficiency, low-loss concentration and buffer exchange of nucleic acids and proteins in molecular biology and bioprocessing workflows. Based on tangential-flow ultrafiltration principles adapted to centrifugal operation, these devices utilize a patented horizontal membrane orientation combined with a reversible centrifugation protocol—enabling near-complete sample recovery without membrane clogging or concentration polarization. The core membrane material, Hydrosart®—a highly hydrophilic, low-protein-binding regenerated cellulose—ensures exceptional flux stability and minimal nonspecific adsorption across a broad pH and ionic strength range. Designed specifically for demanding applications such as next-generation sequencing library prep, qPCR sample enrichment, and low-abundance protein purification, the PCR-grade variants undergo validated ethylene oxide (ETO) sterilization to eliminate trace DNA contamination, ensuring compatibility with stringent amplification protocols where background interference must be reduced to sub-femtogram levels.
Key Features
- Horizontal membrane geometry minimizes concentration polarization and maximizes convective mass transfer during centrifugation.
- Reversible spin protocol: initial concentration followed by inverted centrifugation ensures >90% recovery of retained analytes—including fragmented DNA and labile proteins.
- Hydrosart® membrane exhibits superior wetting characteristics, rapid equilibration, and consistent performance across variable sample viscosities and salt concentrations.
- PCR-grade versions (Vivacon® 500-PCR / Vivacon® 2-PCR) are ETO-sterilized and independently verified for absence of amplifiable DNA, RNase, DNase, and PCR inhibitors per ISO 13485 and CLSI EP17-A2 guidelines.
- Endotoxin levels <0.5 EU/mL; certified non-pyrogenic for cell-based assays and therapeutic protein processing.
- Wide MWCO selection (2–125 kDa) supports diverse biomolecular targets—from oligonucleotides and peptides to monoclonal antibodies and viral capsid proteins.
Sample Compatibility & Compliance
Vivacon® concentrators demonstrate robust compatibility with aqueous biological matrices including serum, plasma, cell lysates, column eluates, and enzymatic digests. They tolerate common additives such as EDTA, DTT, glycerol (≤20%), and urea (≤4 M), though organic solvents and strong chaotropes are not recommended. All devices comply with ISO 10993-5 (cytotoxicity) and USP (in vitro reactivity). PCR-grade units meet requirements for ISO/IEC 17025-accredited laboratories performing diagnostic nucleic acid testing and are documented for GLP/GMP-aligned workflows under FDA 21 CFR Part 11-compliant data management systems when paired with validated instrumentation.
Software & Data Management
While Vivacon® devices are consumable-only components and do not incorporate embedded electronics or firmware, their use is fully supported within LIMS-integrated environments. Batch-specific certificates of analysis (CoA), including MWCO verification, endotoxin assay results, and ETO sterilization validation reports, are provided digitally via Sartorius’ QbD Portal. Traceability is maintained through unique lot numbering aligned with ISO 9001:2015 quality records, enabling full audit trail generation for regulatory submissions (e.g., IND, BLA, CE-IVDR). Integration with electronic lab notebooks (ELNs) is facilitated via standardized SDTM-compatible metadata export.
Applications
- Concentration of dilute DNA fragments (e.g., sheared genomic DNA, ChIP-seq libraries) prior to adapter ligation or quantification by fluorometry.
- Desalting and buffer exchange of purified antibodies prior to conjugation or formulation development.
- Rapid removal of free dNTPs, primers, and polymerase from PCR reactions without phenol-chloroform extraction.
- Preparative-scale enrichment of extracellular vesicles and exosomes from conditioned media using sequential MWCO filtration strategies.
- Downstream processing of CRISPR-Cas ribonucleoprotein complexes requiring nuclease-free handling and minimal aggregation.
- Concentration of low-yield recombinant proteins expressed in E. coli or insect cells, particularly those prone to shear-induced denaturation.
FAQ
What distinguishes Vivacon® 500-PCR from standard Vivacon® 500 devices?
Vivacon® 500-PCR units undergo additional ethylene oxide sterilization and are tested for residual DNA using quantitative PCR targeting human β-globin and bacterial 16S rRNA loci—ensuring no detectable amplifiable template remains post-processing.
Can Vivacon® concentrators be reused?
No. These are single-use, sterile-filtered consumables designed for maximum reproducibility and contamination control. Reuse risks membrane integrity compromise, carryover, and deviation from published recovery metrics.
Is Hydrosart® compatible with high-salt buffers like 1 M NaCl?
Yes. Hydrosart® maintains structural integrity and rejection performance in physiological and high-ionic-strength solutions; however, prolonged exposure to >2 M ammonium sulfate may reduce flux over extended runs.
What is the maximum recommended centrifugation speed for Vivacon® 2-PCR with 100 kDa MWCO?
14,000 × g for up to 15 minutes at 4°C; exceeding this may cause seal failure or membrane delamination.
Do Vivacon® devices require pre-wetting or priming before use?
No. Hydrosart® membranes are pre-hydrated and ready-to-use; direct loading of samples is validated and recommended to preserve low-binding surface properties.