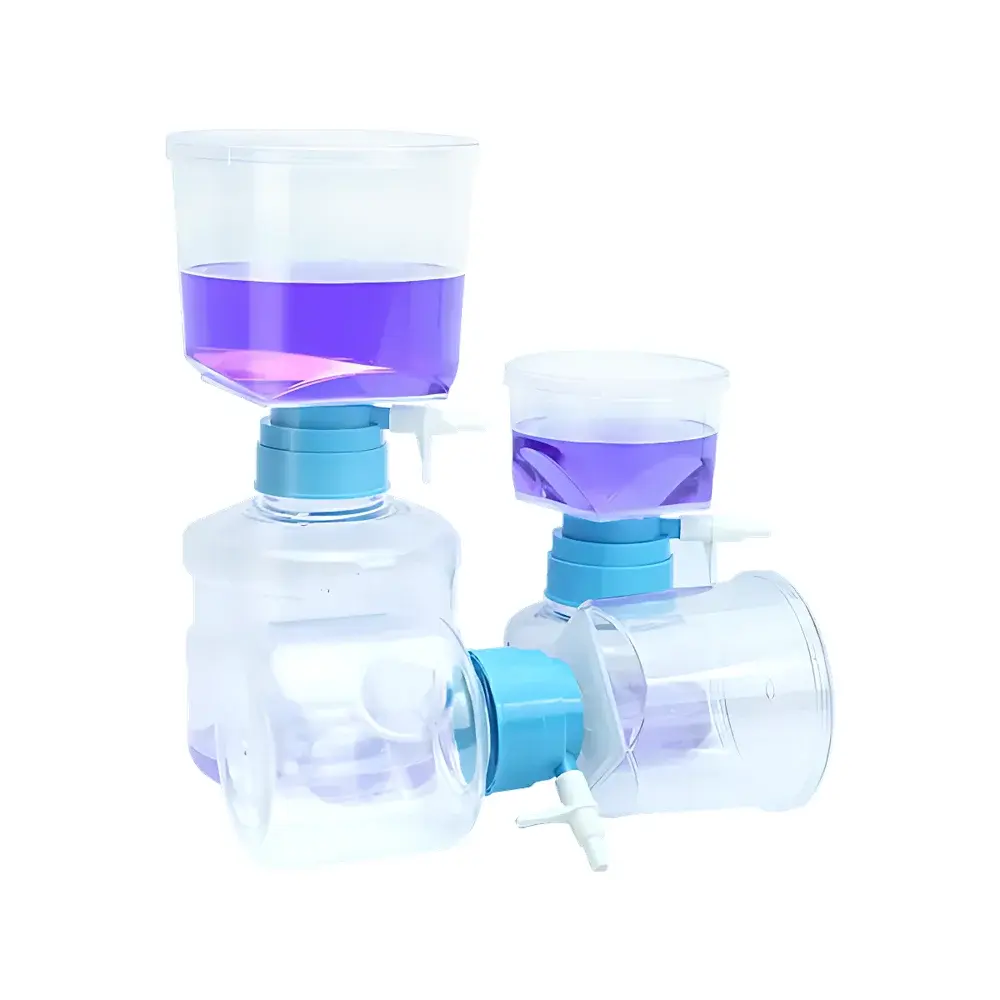
Tailin Biotech Vacuum Filtration Unit for Sterile Microbial Collection
| Brand | Tailin Biotech |
|---|---|
| Origin | Zhejiang, China |
| Manufacturer Type | Direct Manufacturer |
| Product Category | Domestic |
| Model | Vacuum Filtration Unit |
| Pricing | Available Upon Request |
Overview
The Tailin Biotech Vacuum Filtration Unit is a purpose-built sterile filtration system engineered for quantitative microbial collection (e.g., membrane filtration-based sterility testing) in compliance with pharmacopoeial standards including USP , EP 2.6.1, and JP 4.07. It operates on the principle of vacuum-driven cross-flow filtration through certified low-protein-binding, hydrophilic polyethersulfone (PES) or mixed cellulose ester (MCE) membranes—enabling efficient retention of viable microorganisms from large-volume aqueous samples without thermal or chemical denaturation. Designed specifically for pharmaceutical quality control laboratories, this unit maintains sample integrity under ambient conditions, eliminating phase change, enzymatic degradation, or endotoxin leaching—critical for subsequent colony enumeration, identification, or molecular analysis.
Key Features
- Manufactured in ISO Class 7 (Class 10,000) cleanroom environment to ensure pyrogen-free, DNase-/RNase-free construction;
- Pre-sterilized via validated gamma irradiation (25–35 kGy), with full sterility assurance documentation (SAL ≤ 10⁻⁶) and bioburden traceability per ISO 11137;
- Square-format filtration surface increases effective membrane area by ≥20% compared to conventional round housings—enhancing throughput and reducing filter clogging risk;
- Integrated support base with optimized drainage geometry minimizes residual liquid volume (<0.1 mL per filter), improving recovery efficiency for low-biomass samples;
- Dedicated pressure-relief mechanism prevents membrane rupture during vacuum ramp-up or sudden pressure equalization—preserving filter integrity and assay validity;
- One-piece upper cup design eliminates multi-component assembly, minimizing potential leak paths and simplifying pre-use integrity verification (e.g., bubble point or diffusion test);
- High-clarity, autoclavable polycarbonate housing with laser-etched volumetric graduations (±2% accuracy) enables real-time volume monitoring and precise endpoint determination.
Sample Compatibility & Compliance
The unit accommodates standard 47 mm or 50 mm diameter sterile filters (including 0.22 µm and 0.45 µm pore sizes) and supports filtration of diverse pharmaceutical matrices: injectables, ophthalmic solutions, irrigation fluids, raw material buffers, and cell culture supernatants. All wetted components comply with USP Class VI biological safety testing and pass cytotoxicity (ISO 10993-5), hemolysis (ISO 10993-4), and systemic toxicity (ISO 10993-11) evaluations. The system is compatible with GLP- and GMP-regulated workflows, supporting audit-ready documentation including lot-specific CoA, irradiation certificates, and endotoxin test reports (<0.03 EU/mL).
Software & Data Management
While the vacuum filtration unit itself is hardware-only, it integrates seamlessly into regulated laboratory environments via companion digital workflows. When used with Tailin’s optional vacuum manifold controllers (e.g., VF-MC200), users gain programmable vacuum ramp profiles, real-time pressure logging, and timestamped event records—exportable in CSV or PDF format. These data streams are structured to align with FDA 21 CFR Part 11 requirements when deployed within validated LIMS or ELN platforms, enabling electronic signature support, audit trail generation, and user access controls.
Applications
- Sterility testing of finished drug products and excipients per USP/EP/JP;
- Bioburden monitoring of purified water, WFI, and clean steam condensate;
- Environmental monitoring of Grade A/B cleanroom rinse waters;
- Recovery validation studies for low-concentration microbial suspensions;
- Pre-filtration conditioning prior to MALDI-TOF MS or next-generation sequencing workflows;
- Endotoxin removal verification using depyrogenated filter assemblies.
FAQ
Is the filtration unit supplied as a complete sterile kit or individual components?
Each unit ships as a fully assembled, gamma-irradiated, individually wrapped sterile kit—including upper cup, base, gasket, and pre-attached membrane holder—ready for immediate use in ISO 5 laminar flow hoods.
Can the unit be reused after sterilization?
No. This is a single-use, disposable device designed for one-time application to eliminate cross-contamination risk and ensure consistent performance across batches.
What regulatory documentation accompanies each lot?
Each shipment includes Certificate of Analysis (CoA), Certificate of Irradiation, Endotoxin Test Report, and Biological Safety Evaluation Summary—all traceable to manufacturing lot and irradiation batch.
Does the unit support compatibility with third-party vacuum manifolds?
Yes. Standardized 1/4″ NPT vacuum port and modular base geometry ensure interoperability with common laboratory vacuum systems (e.g., KNF, Welch, Gardner Denver), provided vacuum stability remains within −0.8 to −0.95 bar range.
How is filter integrity verified post-filtration?
Users perform in-situ non-destructive integrity tests (bubble point or forward flow) using Tailin-certified test fixtures; validation protocols align with ASTM F838 and ISO 16889 for porous membrane devices.