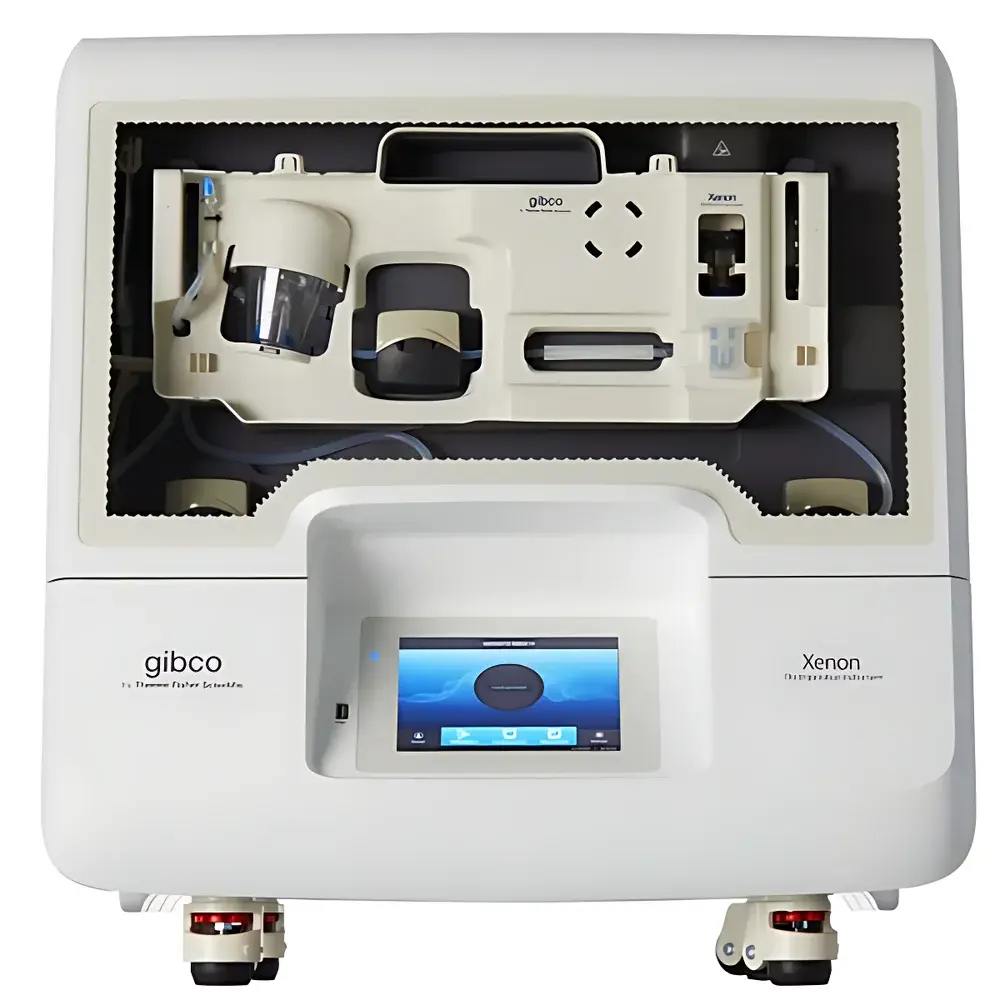
Thermo Fisher Gibco CTS Xenon Large-Scale Electroporation System
| Brand | Thermo Fisher |
|---|---|
| Origin | Singapore |
| Manufacturer Type | Original Equipment Manufacturer (OEM) |
| Product Origin | Imported |
| Model | CTS Xenon |
| High Voltage Range | 2500 V |
| Low Voltage Range | 500 V |
| Pulse Count | 1–10 |
| Pulse Duration | 1–30 ms |
| Output Waveform | Square Wave |
Overview
The Thermo Fisher Gibco CTS Xenon Large-Scale Electroporation System is a closed, modular, GMP-compatible platform engineered for scalable, non-viral nucleic acid and protein delivery into primary human cells—including T cells, NK cells, CD34+ hematopoietic stem cells (HSCs), and other difficult-to-transfect cell types. It operates on the principle of controlled electroporation: transient, reversible membrane permeabilization induced by precisely regulated square-wave electrical pulses. Unlike viral vector-based delivery, the CTS Xenon system enables physical, footprint-free genetic modification without integration risk, supporting plasmid DNA, mRNA, siRNA/miRNA, ribonucleoprotein (RNP) complexes (e.g., TrueCut Cas9/gRNA), and recombinant proteins. Designed explicitly for cell therapy process development and commercial manufacturing, it bridges the gap between research-scale Neon electroporation and clinical-grade production—maintaining identical pulse configuration logic while enabling volumetric scalability from 1 mL to 25 mL per run. Its architecture supports seamless integration into end-to-end closed workflows, including upstream cell activation, buffer exchange via CTS Rotea counterflow centrifugation, and downstream expansion.
Key Features
- Full parameter control via embedded graphical user interface (eGUI): adjustable voltage (500–2500 V), pulse width (1–30 ms), pulse count (1–10), and inter-pulse delay
- Closed-system processing enabled by sterile-weldable MultiShot (MS) consumables compatible with DEHP-free PVC and C-Flex tubing
- Two validated consumable formats: SingleShot (SS) chamber for process development (1 mL, 2×10⁷–1×10⁸ cells); MultiShot (MS) kit for GMP manufacturing (5–25 mL, 1×10⁸–2.5×10⁹ cells)
- Tritan™ MX711 copolyester chambers, gamma-irradiated and certified for use in ISO Class C cleanrooms
- No licensing fees, milestone payments, or royalty obligations—designed for cost-effective technology transfer and commercial deployment
- Consistent performance across scale: validated equivalency in editing efficiency (75–82% TCR/CD34/B2M knockout) and post-electroporation viability (>80–90%) between Neon (100 µL) and Xenon (1–25 mL) platforms
Sample Compatibility & Compliance
The CTS Xenon system is qualified for use with human primary immune cells under current Good Manufacturing Practice (cGMP) conditions. It supports three proprietary, serum-free, low-conductivity electroporation buffers—CTS Xenon EP Buffer (broad-spectrum transfection), GE Buffer (optimized for CRISPR/Cas9 and TALEN RNP delivery), and LC Buffer (for high-energy applications requiring enhanced membrane poration). All buffers are supplied in sterile, single-use 100 mL bottles or bags compatible with closed-system fluid management. The platform complies with ISO 13485:2016 for medical device quality management systems and is designed to support regulatory submissions aligned with FDA 21 CFR Part 11 (electronic records/signatures), ICH Q5A(R2) (viral safety), and EMA Guideline on Human Cell-Based Medicinal Products. Documentation packages include IQ/OQ/PQ protocols, traceable calibration records, and audit-ready change control logs.
Software & Data Management
The embedded eGUI provides intuitive, touch-enabled protocol creation, execution, and archiving. Each electroporation run generates timestamped, user-attributed metadata—including all pulse parameters, chamber ID, buffer lot number, operator ID, and environmental monitoring flags (if integrated with facility SCADA). Audit trails are immutable and exportable in CSV or PDF format, satisfying GLP/GMP data integrity requirements per ALCOA+ principles. When paired with Thermo Fisher’s Cellmation digital integration software, the CTS Xenon system interfaces bi-directionally with CTS Rotea and DynaCellect platforms, enabling synchronized workflow orchestration, real-time performance analytics, and electronic batch record (EBR) generation. All software modules adhere to IEC 62304 for medical device software lifecycle management.
Applications
The CTS Xenon system is deployed across autologous and allogeneic cell therapy pipelines for: CAR-T and TCR-T engineering (e.g., TCRαβ knockout + CAR insertion), NK cell enhancement (B2M/CIITA knockdown), HSC gene correction (e.g., β-globin locus editing), and transient protein expression for functional screening. Published case studies demonstrate robust performance in multi-donor validation batches—achieving >80% viability and >75% editing efficiency in CD3+ T cells electroporated with Cas9 RNP at 2.5×10⁹ total cells per run. Its compatibility with TrueCut Cas9 protein and synthetic gRNA enables rapid, off-the-shelf genome editing without plasmid amplification steps. The system also supports non-integrating mRNA delivery for transient antigen presentation or cytokine modulation in dendritic cell therapies.
FAQ
Is the CTS Xenon system compliant with FDA 21 CFR Part 11?
Yes—the eGUI enforces role-based access control, electronic signatures, and tamper-evident audit trails meeting Part 11 requirements for electronic records.
Can I transfer protocols directly from the Neon system to Xenon?
Yes—identical pulse configuration logic allows direct parameter mapping; optimization typically requires only minor voltage/pulse width adjustments for volume scaling.
What buffer options are available for CRISPR RNP delivery?
The CTS Xenon GE Buffer is specifically formulated and validated for high-efficiency RNP delivery into primary T and NK cells.
Does the system support automated integration with upstream/downstream equipment?
Yes—via standardized analog/digital I/O and OPC UA connectivity, it integrates with CTS Rotea, DynaCellect, bioreactors, and fill-finish stations.
Are consumables supplied sterile and ready for use in Class C environments?
All SingleShot and MultiShot chambers undergo gamma irradiation (25–35 kGy) and are certified for aseptic welding and cleanroom handling per ISO 14644-1 Class 5–8 specifications.