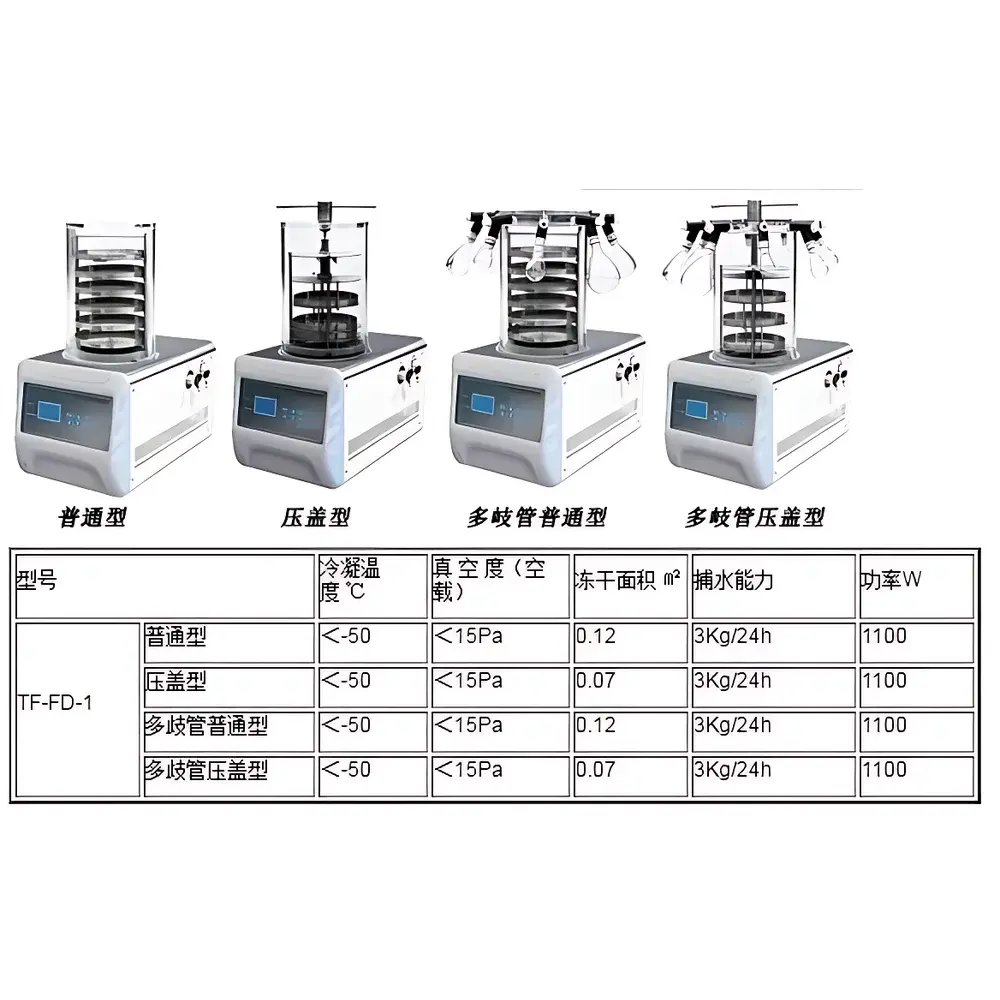
Tianfeng TF-FD-1 (Stoppering Type) Benchtop Laboratory Freeze Dryer
| Brand | Tianfeng |
|---|---|
| Origin | Shanghai, China |
| Manufacturer | OEM Producer |
| Type | Stoppering-Type Lyophilizer |
| Application | Laboratory-Scale Freeze Drying |
| Shelf Area | 0.07 m² |
| Ultimate Vacuum (No Load) | <15 Pa |
| Condenser Temperature | <−50 °C |
| Ice Capacity | 3 kg/24 h |
| Power Consumption | 1100 W |
| Dimensions (W×D×H) | 370 × 650 × 360 mm |
| Chamber Material | 304 Stainless Steel |
| Viewport | Transparent Acrylic Door |
| Control System | Touchscreen LCD with PID Regulation |
| Pre-freezing | In-situ via condenser |
| Optional | Inert Gas Purge Valve, Temperature Data Logger, Eutectic Point Tester |
Overview
The Tianfeng TF-FD-1 (Stoppering Type) Benchtop Laboratory Freeze Dryer is a compact, in-situ pre-freezing lyophilization system engineered for precise, reproducible freeze drying of heat-sensitive biological and pharmaceutical samples under controlled vacuum and low-temperature conditions. Based on the principle of sublimation—where ice transitions directly from solid to vapor phase without passing through liquid—this system preserves structural integrity, bioactivity, and chemical stability of labile compounds such as proteins, enzymes, vaccines, antibiotics, and herbal extracts. Unlike conventional thermal drying methods that induce denaturation, oxidation, or collapse, freeze drying occurs predominantly below the eutectic temperature, maintaining porous matrix architecture and enabling rapid reconstitution with minimal loss of functionality. Designed for benchtop use in R&D labs, QC/QA environments, and small-batch process development, the TF-FD-1 integrates a −50 °C condenser, stainless-steel chamber, and transparent acrylic viewport to support real-time visual monitoring of primary and secondary drying stages.
Key Features
- In-situ pre-freezing capability: Samples are frozen directly on the shelf or within the condenser chamber, eliminating manual transfer and minimizing thermal shock or contamination risk.
- Stoppering-compatible design: Equipped with a hydraulic or pneumatic stoppering mechanism (optional) for sterile vial sealing under vacuum—critical for aseptic pharmaceutical formulation development and GLP-compliant studies.
- High-efficiency dual-stage refrigeration: Air-cooled, single-compressor system achieves stable condenser temperatures ≤−50 °C, ensuring reliable water vapor capture and high ice capacity (3 kg/24 h).
- Intuitive touchscreen interface: Real-time display of shelf temperature, chamber pressure, condenser temperature, and drying curve; supports user-defined PID tuning for optimized cycle control.
- Corrosion-resistant construction: Entire drying chamber and condenser housing fabricated from electropolished 304 stainless steel; compliant with ISO 14644-1 Class 7 cleanroom compatibility requirements.
- Inert gas purge port: Integrated valve enables backfilling with nitrogen or argon post-drying to prevent oxidation and stabilize oxygen-sensitive formulations.
- Modular expandability: Compatible with optional accessories including calibrated temperature probes (±0.5 °C), eutectic point measurement kits, and 21 CFR Part 11–compliant data loggers with audit trail functionality.
Sample Compatibility & Compliance
The TF-FD-1 accommodates standard Φ180 mm sample trays across three shelf levels, supporting aqueous, buffered, or low-concentration organic solvent formulations (e.g., ethanol/water mixtures ≤10% v/v). It meets essential performance benchmarks referenced in USP , ISO 22054:2021 (freeze-drying of biopharmaceuticals), and ASTM F2375-04 (standard guide for lyophilization process validation). While not intrinsically rated for Class I Division 1 hazardous locations, its sealed refrigeration circuit, grounded chassis, and absence of internal spark sources make it suitable for general laboratory use—including GMP-aligned pilot-scale preparation of nutraceuticals, botanical extracts, and probiotic cultures—provided ambient ventilation and electrical grounding conform to IEC 61000-6-3 EMC standards.
Software & Data Management
The embedded controller records time-stamped process parameters at user-selectable intervals (1–60 s), exporting CSV-formatted datasets for offline analysis in MATLAB, Python, or Excel. Optional firmware upgrades support automated cycle recall, password-protected parameter locking, and electronic signature workflows aligned with FDA 21 CFR Part 11 Annex 11 expectations. When paired with a validated external data logger, the system satisfies ALCOA+ data integrity principles (Attributable, Legible, Contemporaneous, Original, Accurate) required for regulatory submissions to EMA, PMDA, and NMPA.
Applications
- Pharmaceutical R&D: Lyophilization of monoclonal antibodies, peptide APIs, and sterile injectables in glass vials.
- Biotechnology: Stabilization of cell lysates, viral vectors, CRISPR-Cas9 ribonucleoprotein complexes, and lyophilized diagnostics reagents.
- Nutraceutical & Functional Food Development: Preservation of probiotics (e.g., Lactobacillus spp.), enzymes (bromelain, papain), and phytochemical-rich botanical concentrates (ginseng, goji, turmeric).
- Academic Research: Structural biology sample preparation for cryo-EM grid freezing, microbiology culture banking, and long-term archiving of clinical specimens (serum, plasma, tissue homogenates).
- Quality Control Laboratories: Accelerated stability testing per ICH Q1A(R2), residual moisture analysis (Karl Fischer correlation), and reconstitution time validation.
FAQ
What is the maximum batch volume supported by the TF-FD-1’s 0.07 m² shelf area?
Typical loading capacity is 1–2 L of 5–10% w/v solution across three Φ180 mm trays, assuming uniform layer thickness ≤8 mm and optimal cake formation.
Can this unit handle samples containing acetonitrile or methanol?
Yes—when operated with extended condenser dwell time and reduced primary drying ramp rates, the −50 °C cold trap effectively captures low-boiling solvents up to 15% v/v concentration; however, solvent-specific validation is recommended.
Is the stoppering function automated and vacuum-integrated?
The optional stoppering module actuates under full vacuum (<15 Pa), enabling hermetic seal formation without air exposure—essential for oxygen-labile compounds and sterile product development.
Does the system comply with Good Manufacturing Practice (GMP) documentation requirements?
Out-of-the-box, it supports traceable operation via timestamped logs; full GMP compliance requires integration with validated third-party software for electronic records, user access controls, and change management protocols.
What maintenance intervals are recommended for sustained performance?
Oil change every 2,000 operating hours; vacuum pump oil replacement every 500 hours; condenser coil cleaning quarterly; O-ring inspection and lubrication biannually per ISO 13485 preventive maintenance guidelines.