Tianfeng TF-FD-1PF Manifold Freeze Dryer with Stoppering Function
| Brand | Tianfeng |
|---|---|
| Origin | Shanghai, China |
| Manufacturer Type | Direct Manufacturer |
| Instrument Type | Manifold Freeze Dryer |
| Application | Laboratory-Scale Lyophilization |
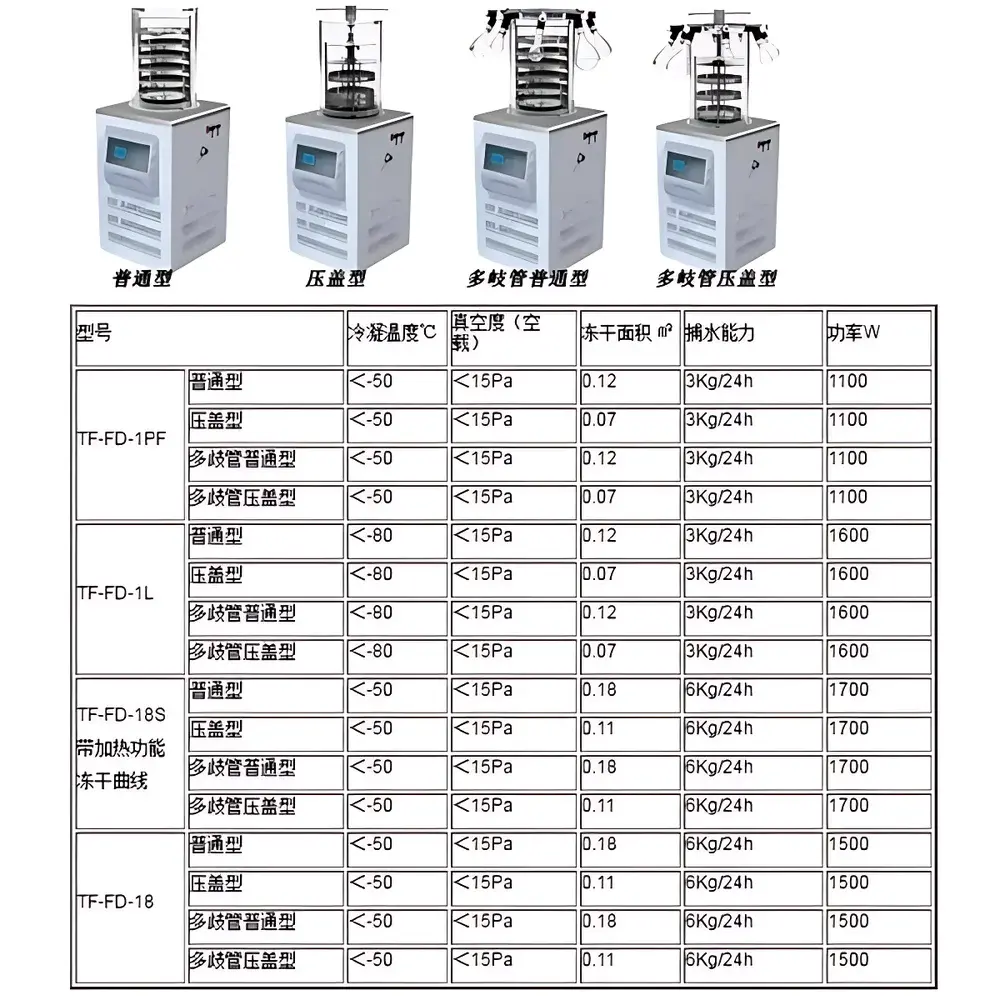
| Shelf Area | 0.07 m² |
| Ultimate Vacuum | <15 Pa (empty chamber) |
| Condenser Temperature | ≤−50 °C |
| Ice Capacity | 3 kg/24 h |
| Power Consumption | 1100 W |
| Electrical Supply | 220 V, 50 Hz |
| Sample Vials | Compatible with 50–1000 mL flasks (8 ports) |
| Chamber Design | Vertical stainless steel body with transparent acrylic drying chamber |
| Condenser Type | Air-cooled single-stage refrigeration system |
| Control Interface | Analog or basic digital panel (no programmable shelf heating) |
Overview
The Tianfeng TF-FD-1PF is a compact, vertical manifold freeze dryer engineered for laboratory-scale lyophilization of heat-sensitive biological and pharmaceutical samples. It operates on the fundamental principle of sublimation—removing water from frozen materials under deep vacuum while maintaining product integrity. Unlike conventional thermal drying methods, freeze drying preserves structural morphology, enzymatic activity, immunogenicity, and chemical stability by avoiding liquid-phase transitions and elevated temperatures. The TF-FD-1PF employs a single-stage air-cooled refrigeration system to achieve condenser temperatures ≤−50 °C, enabling efficient capture of water vapor during primary drying. Its manifold configuration supports simultaneous processing of up to eight independently sealed glass vessels (e.g., round-bottom flasks), making it ideal for comparative batch trials, solvent-sensitive formulations, or multi-sample screening where individual vial isolation is required.
Key Features
- Manifold design with eight independent stainless steel ports and silicone gasket-sealed stopcocks for precise vacuum control per vessel
- Integrated stoppering function allows in-chamber vacuum sealing of vials post-drying—critical for sterile handling of biologics and GMP-aligned workflows
- Stainless steel condenser and chamber housing ensure corrosion resistance and long-term vacuum integrity
- Transparent acrylic drying chamber enables real-time visual monitoring of ice formation, sublimation front progression, and cake structure development
- Pre-frosting capability: Samples may be pre-frozen directly on the condenser surface prior to main drying cycle initiation
- Robust air-cooled refrigeration architecture eliminates dependency on external cooling water, reducing infrastructure requirements and operational complexity
- Compliant with ISO 22033 (freeze-drying terminology) and aligned with core principles of USP <1211> and ICH Q5C guidelines for biopharmaceutical stability
Sample Compatibility & Compliance
The TF-FD-1PF accommodates a broad spectrum of aqueous and mildly volatile solvent-based formulations common in preclinical R&D environments—including monoclonal antibody solutions, plasmid DNA suspensions, bacterial lysates, enzyme preparations, and vaccine candidates. Its −50 °C condenser provides adequate trapping efficiency for water and low-volatility solvents (e.g., tert-butanol, acetic acid), though highly volatile organics (e.g., ethanol, acetone) require supplementary cold traps or process adaptation. The unit meets CE marking requirements for electrical safety (EN 61000-6-3, EN 61000-6-4) and mechanical design (EN ISO 12100). While not certified to 21 CFR Part 11 out-of-the-box, its analog/digital interface supports integration with validated data logging systems compliant with GLP and GMP audit trails when deployed within controlled laboratory environments.
Software & Data Management
The TF-FD-1PF is equipped with a dedicated control panel featuring vacuum gauge readout, condenser temperature display, and cycle timing functions. Though it lacks embedded programmable logic controllers (PLCs) or Ethernet connectivity, its operational parameters—including vacuum level, elapsed time, and condenser status—are fully observable and manually recordable. For laboratories requiring electronic data capture, third-party pressure transducers and temperature probes (e.g., PT100 sensors at shelf and condenser locations) can be interfaced via standard 4–20 mA or 0–5 V analog outputs. This facilitates traceable documentation compatible with internal SOPs and regulatory submissions under FDA, EMA, or PMDA review frameworks.
Applications
- Lyophilization of clinical trial materials including recombinant proteins, peptides, and oligonucleotides
- Stabilization of diagnostic reagents, reference standards, and ELISA kit components
- Preservation of microbial cultures, bacteriophages, and cell line stocks
- Routine drying of HPLC fractions, natural product isolates, and synthetic intermediates
- Method development and cycle optimization studies prior to scale-up on production-grade lyophilizers
- Academic research in biomaterials science, nanocarrier formulation, and controlled-release matrix design
FAQ
What types of flasks are compatible with the TF-FD-1PF manifold?
Standard ground-glass jointed round-bottom flasks (50 mL, 100 mL, 250 mL, 500 mL, and 1000 mL) with matching stopcocks and O-ring seals are supported. Custom adapters are available for non-standard joints.
Can the TF-FD-1PF handle organic solvents?
It effectively traps water and low-volatility solvents such as tert-butanol and dimethyl sulfoxide (DMSO) at ≤−50 °C. Highly volatile solvents like methanol or ethyl acetate require supplemental cold trapping or reduced loading volumes to prevent condenser overload.
Is pre-freezing required before loading samples?
Pre-freezing is strongly recommended—either in a separate ultra-low temperature freezer (≤−40 °C) or on the condenser surface. In-chamber pre-freezing minimizes thermal shock and ensures uniform nucleation.
Does the unit include validation documentation?
Factory calibration certificates for vacuum and temperature sensors are provided. IQ/OQ protocols and installation qualification templates are available upon request for integration into regulated quality systems.