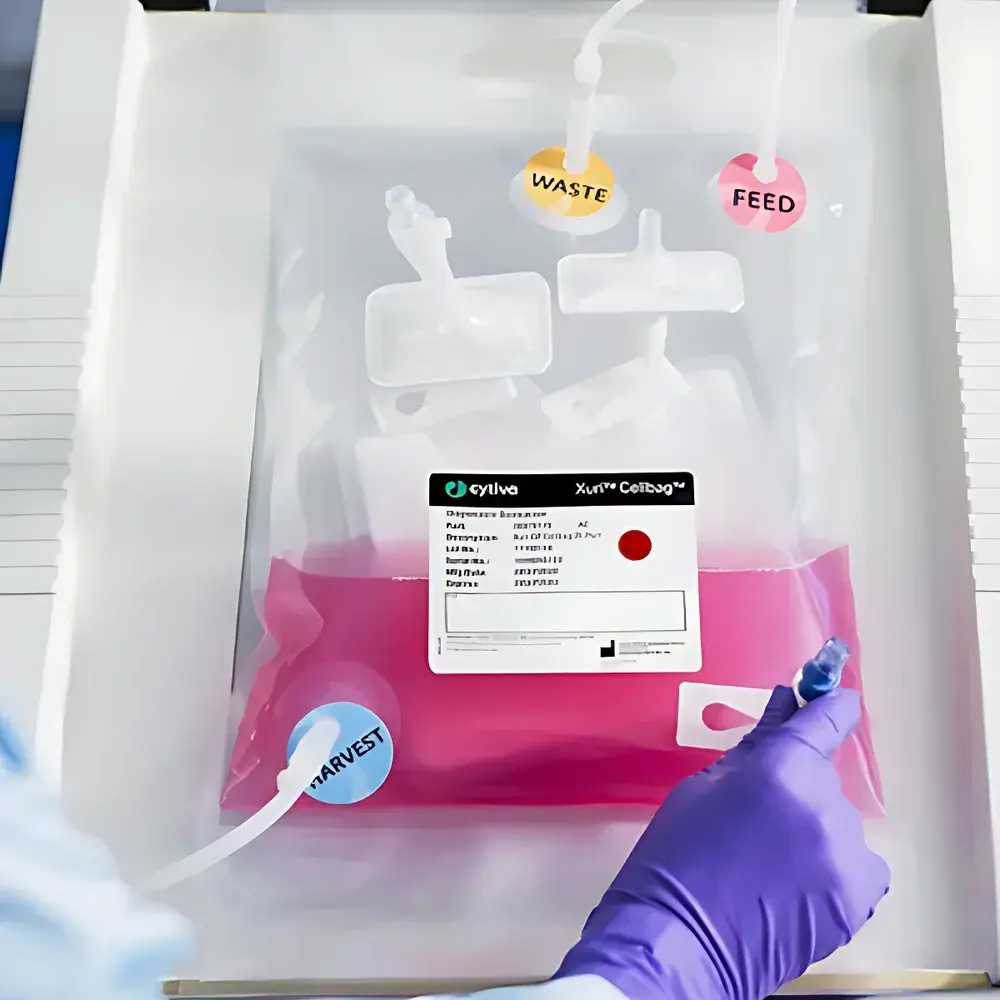
Wave Cellbag by Cytiva
| Brand | Cytiva |
|---|---|
| Origin | Sweden |
| Model | Wave Cellbag |
| Sterilization Method | Gamma Irradiation |
| Material Compliance | USP Class VI |
| Regulatory Documentation | FDA-registered DMF on file |
| Intended Use | Research, Process Development, cGMP Manufacturing, Cell Therapy Production |
| Bag Configuration | Single-use, pre-sterilized, disposable bioprocess bag |
Overview
The Wave Cellbag by Cytiva is a single-use, gamma-irradiated cell culture bag engineered for scalable, sterile mammalian and stem cell expansion in wave-mixed bioreactor systems—specifically the WAVE™ Bioreactor platform. Designed around the principles of gentle, low-shear mixing via orbital rocking motion, the Cellbag provides a closed, controlled environment that maintains physiological gas exchange (O₂/CO₂), pH stability, and uniform nutrient distribution without mechanical agitation or sparging-induced shear stress. Its construction adheres to stringent pharmaceutical manufacturing requirements, supporting applications ranging from early-stage research through clinical-grade cell therapy manufacturing under cGMP conditions.
Key Features
- Manufactured from multi-layer, transparent, pharmaceutical-grade film compliant with USP Class VI biological reactivity standards—ensuring full biocompatibility and extractables/leachables profiles suitable for sensitive primary and therapeutic cell lines.
- Pre-sterilized using validated gamma irradiation (25–35 kGy), delivering sterility assurance level (SAL) of 10⁻⁶ and endotoxin levels <0.25 EU/mL—certified non-pyrogenic per USP & EP monographs.
- Integrated fluid pathways with sterile-welded ports (e.g., sampling, inoculation, harvest, gas inlet/outlet) configured for aseptic connection to tubing manifolds and peristaltic pumps—enabling seamless integration into closed-process workflows.
- Optimized film elasticity and tensile strength support consistent bag geometry during rocking motion across volumes from 0.5 L to 250 L—minimizing localized stress concentrations and ensuring reproducible mixing dynamics.
- Supplied with comprehensive regulatory documentation, including a Drug Master File (DMF) on file with the U.S. FDA, material traceability records, and Certificate of Analysis (CoA) for each lot.
Sample Compatibility & Compliance
The Wave Cellbag supports suspension, adherent (microcarrier-based), and hybrid cell culture formats—including CHO, HEK293, CAP-T, iPSCs, T cells, and mesenchymal stromal cells (MSCs). It complies with ISO 10993-5 (cytotoxicity), ISO 10993-10 (irritation/sensitization), and ISO 10993-11 (hemocompatibility) testing protocols. All raw materials are sourced under strict supplier qualification programs aligned with ICH Q5A(R2) and Q5D guidelines. The bag system is compatible with GMP-compliant process validation activities, including installation qualification (IQ), operational qualification (OQ), and performance qualification (PQ) as defined in ASTM E2500 and EU Annex 15.
Software & Data Management
While the Cellbag itself is a passive consumable, its use is fully integrated into Cytiva’s broader digital bioprocessing ecosystem. When operated within WAVE™ Bioreactor systems equipped with BioPAT® MFCS software, users benefit from real-time monitoring of rocking angle, frequency, temperature, dissolved oxygen (DO), and pH—enabling automated feedback control loops and complete audit trails. Data capture meets FDA 21 CFR Part 11 requirements, including electronic signatures, user access controls, and immutable event logs—supporting GLP/GMP compliance and regulatory submissions.
Applications
- Process development and scale-down modeling for monoclonal antibody (mAb) and recombinant protein production
- Expansion of clinical-grade CAR-T and NK cells under cGMP conditions
- Stem cell differentiation and organoid formation in serum-free, xeno-free media systems
- Seed train intensification and bioreactor inoculum preparation
- Comparative studies of shear-sensitive cell phenotypes under varying mixing parameters
FAQ
Is the Wave Cellbag supplied with a Certificate of Conformance and CoA?
Yes—each lot is accompanied by a Certificate of Conformance and a Certificate of Analysis specifying sterilization dose, endotoxin levels, biocompatibility test summaries, and film lot traceability.
Can the Cellbag be used for cryopreservation storage after harvest?
No—the Wave Cellbag is designed exclusively for active culture; it is not rated for long-term frozen storage. Harvested cells should be transferred to cryobags or vials qualified for freezing.
Does Cytiva provide extractables and leachables data for regulatory filings?
Yes—comprehensive E&L reports generated per USP Chapter <1663> and <1664> are available upon request for specific bag sizes and contact conditions.
What is the maximum recommended rocking speed and angle for different bag volumes?
Rocking parameters must be validated per application; typical ranges are 10–30 rpm and 3–12° for 1–50 L bags, and 6–18 rpm and 4–10° for 100–250 L configurations—always referencing Cytiva’s validated operating envelopes.
Is the Cellbag compatible with single-use sensor integration (e.g., optical DO/pH patches)?
Yes—select Wave Cellbag models feature pre-installed, gamma-sterilized optical sensor patches (e.g., PreSens or Hamilton) with calibration traceability and compatibility with standard reader interfaces.