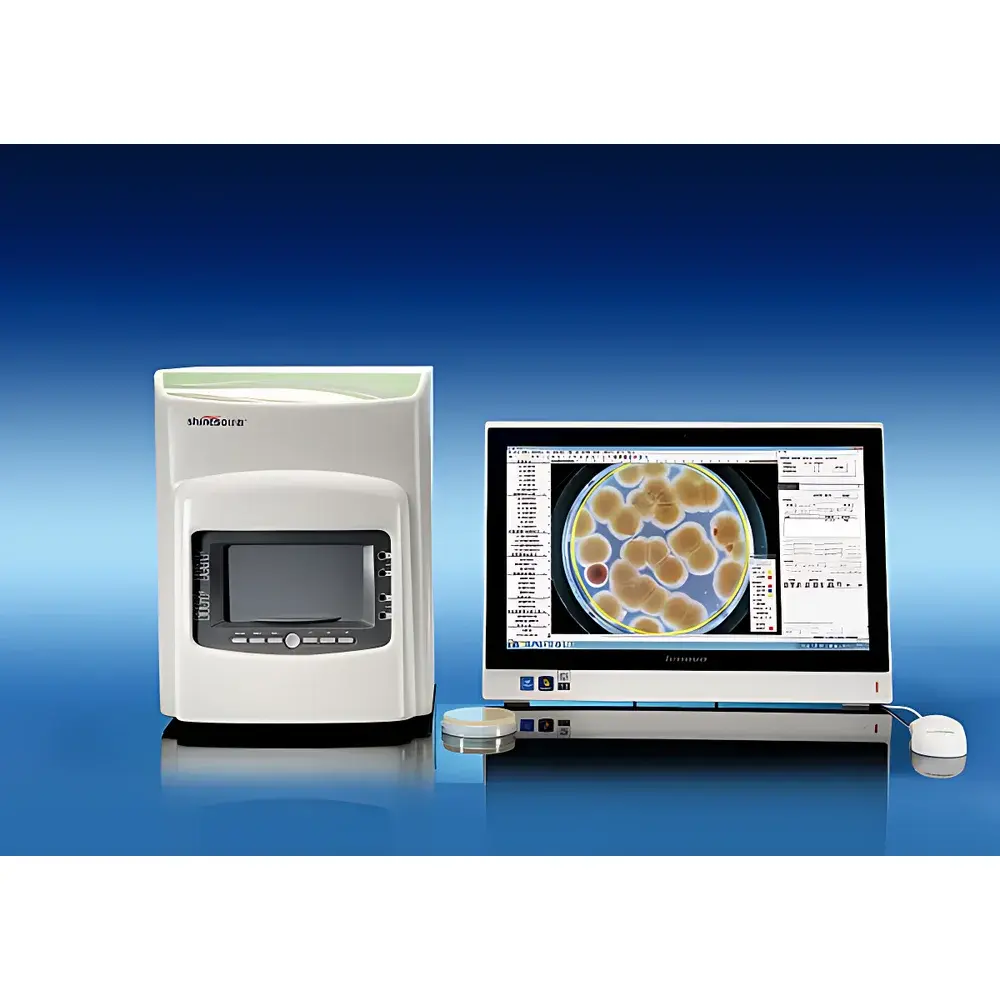
Xunshu iCount 50 Fully Automated Colony Counter
| Brand | Xunshu |
|---|---|
| Origin | Zhejiang, China |
| Manufacturer Type | OEM Manufacturer |
| Instrument Type | Fully Automated Colony Counter |
| Model | iCount 50 |
| Price Range | USD 7,000 – 11,200 (FOB) |
Overview
The Xunshu iCount 50 Fully Automated Colony Counter is an ISO/IEC 17025- and GLP-aligned digital imaging system engineered for high-fidelity microbial colony enumeration in regulated food, pharmaceutical, and clinical microbiology laboratories. It operates on the principle of high-resolution digital image acquisition followed by algorithm-driven morphological segmentation and classification—leveraging topological watershed analysis, level-set multi-model optimization, and adaptive color-space thresholding to distinguish viable colonies from agar matrix artifacts, bubbles, scratches, or insoluble particulates. Designed in compliance with China’s National Medical Products Administration (NMPA) requirements for computerized laboratory information systems—and fully compatible with FDA 21 CFR Part 11 audit-trail mandates—the iCount 50 delivers traceable, tamper-evident electronic records without compromising analytical rigor. Its core architecture integrates hardware-level illumination control, a scientific-grade imaging subsystem, and a four-tier permissioned software framework to enforce data integrity across the entire workflow: from sample imaging through statistical analysis to final report generation and archival.
Key Features
- 20-megapixel Sony 1-inch CMOS sensor with C-mount interface and USB 3.0 output—optimized for sub-millimeter colony resolution and chromatic fidelity under variable agar backgrounds.
- Patented dual-illumination system: top-mounted 360° diffuse trichromatic LED array for uniform surface reflectance; bottom-mounted “CrystalSharp” suspended dark-field illumination for enhanced contrast of subsurface or translucent colonies.
- Six一键 intelligent counting modes: Planar Perception, Volumetric Perception, Small-Colony Priority, Large-Colony Priority, Homochromatic Colony Isolation, and Medium Suppression—each preconfigured for specific plating methods and microbial morphologies.
- Advanced segmentation engine incorporating watershed-based adhesion splitting, dynamic parameter tuning, and inverse-statistical modeling for heterogeneous colony populations on non-uniform media.
- Dedicated algorithms for grid-filter membranes (solid/dashed black grids), 3M Petrifilm™ products (Total Aerobic Count, Staphylococcus aureus, Coliforms, E. coli/Coliform Rapid Count), and spiral plates compliant with FDA-BAM and SN/T 2098-2008 standards.
- Integrated microbial limit analysis module aligned with Chinese Pharmacopoeia (2020 Edition) for culture medium suitability testing and indicator organism morphology verification.
Sample Compatibility & Compliance
The iCount 50 supports all standard plate formats: pour plates, spread plates, membrane filters (including nitrocellulose and polycarbonate), spiral plates, multi-well plates (e.g., OPKA, SBA), and commercial chromogenic test films. It accommodates petri dish diameters from 35 mm to 150 mm and automatically applies dilution factor compensation upon user input. Regulatory alignment includes adherence to ISO 4833-1:2013 (microbiology of food and animal feeding stuffs), USP /, and EU Annex 1 requirements for data integrity. All raw images, processing parameters, operator actions, timestamps, and electronic signatures are immutably logged in an encrypted relational database—enabling full reconstruction of analytical conditions during internal audits or regulatory inspections.
Software & Data Management
The proprietary iCount Analysis Suite implements a four-role hierarchical access model: System Administrator, Data Administrator, Operator, and Reviewer—each with strictly segregated permissions and no cross-role override capability. Audit trails record every action—including image capture, tool selection, parameter adjustment, manual correction, and report submission—with machine-generated timestamps and embedded digital watermarks. Final reports export as PDF/A-1b or Excel (.xlsx) with embedded metadata (sample ID, dilution, plate dimensions, algorithm version, operator ID, reviewer ID, approval timestamp). Database backups support scheduled automated archiving and external media export. The system satisfies FDA 21 CFR Part 11 requirements for electronic records and signatures, including role-based authentication, biometric-ready login options, and immutable log retention for ≥15 years.
Applications
The iCount 50 is deployed in QC/QA laboratories for routine enumeration in food safety (ISO 4833-1, ISO 6887), pharmaceutical sterility testing (USP , EP 2.6.12), environmental monitoring (ISO 14698), antimicrobial efficacy (ISO 22196, ASTM E2149), and antifungal resistance screening (CLSI M38). Its specialized modules support mold quantification per GB/T 27405, serial dilution validation, and parallel analysis of multi-analyte assay plates. In research settings, it enables longitudinal colony morphology tracking, biofilm edge detection, and semi-quantitative assessment of inhibition zones when coupled with zone measurement tools.
FAQ
Does the iCount 50 comply with FDA 21 CFR Part 11?
Yes—the system enforces electronic signature authentication, audit-trail logging, and data immutability through cryptographic hashing and role-based access controls.
Can it process spiral plates according to FDA-BAM guidelines?
Yes—it supports exponential, slow-exponential, uniform, proportional, and lawn-pattern spiral counting modes and is validated against SBI and IUL spiral inoculators.
How does the system prevent unauthorized data modification?
All processing steps—including image acquisition, parameter settings, corrections, and reporting—are time-stamped, digitally signed, and stored in a write-once database; no post-submission edits are permitted.
Is calibration traceable to national standards?
The system includes built-in calibration routines using NIST-traceable stage micrometers and supports user-defined reference standards for diameter and area measurement.
What file formats are supported for data export?
Raw images (TIFF, PNG), processed results (CSV, Excel), and finalized reports (PDF/A-1b) with embedded metadata and electronic watermarks.
Does it support multi-language UI and documentation?
The software interface is English-native with optional Chinese language packs; all regulatory documentation (IQ/OQ/PQ protocols, SOP templates, and validation reports) is provided in English.