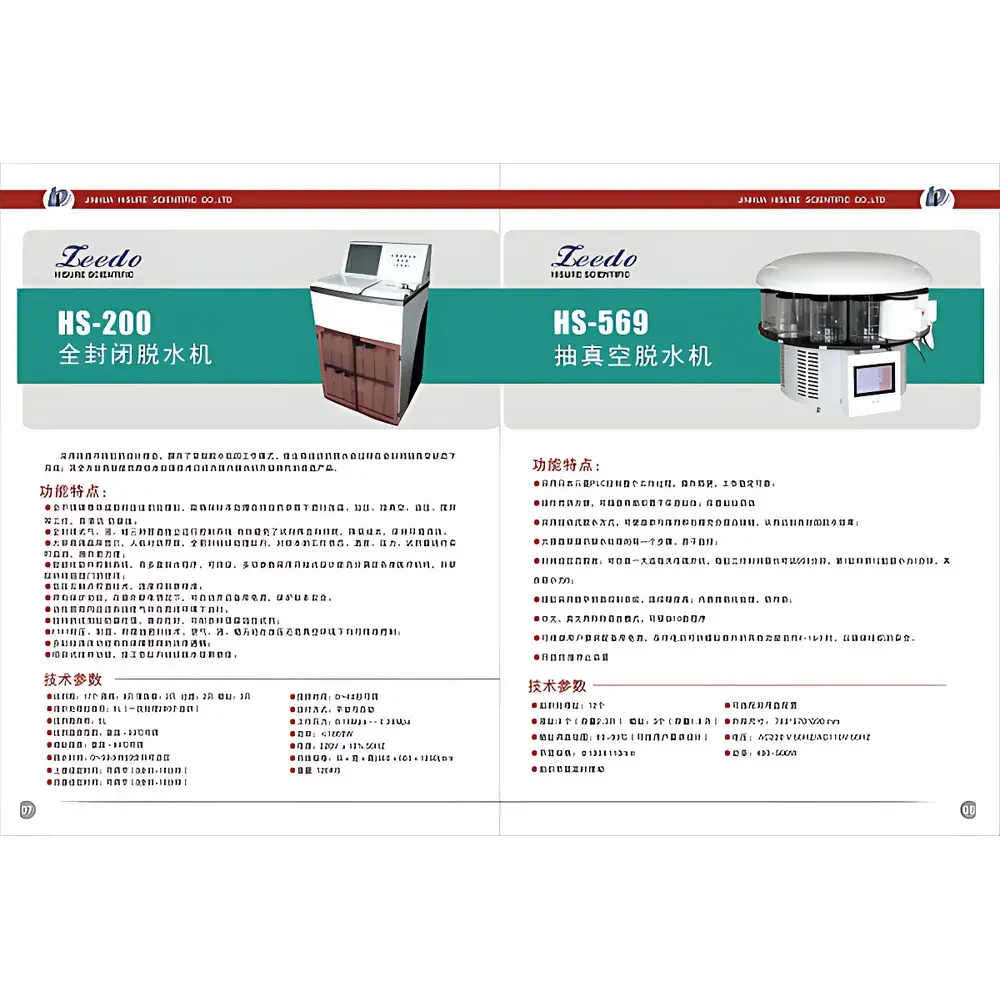
ZEEDO HS-200 Fully Enclosed Vacuum Tissue Dehydrator
| Brand | ZEEDO |
|---|---|
| Origin | Zhejiang, China |
| Manufacturer Type | Authorized Distributor |
| Country of Origin | China |
| Model | HS-200 |
| Price | USD 17,500 (FOB Zhejiang) |
| Tissue Processing Capacity | 200 specimens per cycle |
| Processing Chamber Volume | 5 L |
| Reagent Bottles | 17 (9 liquid, 3 cleaning, 2 filtration, 3 paraffin wax) |
| Reagent/Wax Temperature Range | Ambient to 99 °C |
| Vacuum/Pressure Range | 0.15–0.20 MPa |
| Dehydration Time Setting | 0–99 h 59 min |
| Fluid Transfer Duration | 3–10 min (fill/empty) |
| Stirring Duration | 0–60 s |
| Power Consumption | ≤1800 W |
| Dimensions (L×W×H) | 660 × 600 × 1250 mm |
| Net Weight | 120 kg |
Overview
The ZEEDO HS-200 Fully Enclosed Vacuum Tissue Dehydrator is an advanced, programmable laboratory instrument engineered for automated, high-fidelity tissue processing in histopathology workflows. It operates on a closed-loop vacuum-assisted dehydration principle—utilizing controlled pressure differentials, precise thermal regulation, and sequential reagent exchange—to replace conventional open-air or semi-automated methods. Unlike traditional dehydrators relying on gravity-based fluid transfer or atmospheric-pressure incubation, the HS-200 performs all dehydration, clearing, and infiltration steps within a hermetically sealed processing chamber under dynamically regulated vacuum and positive pressure conditions. This architecture minimizes solvent evaporation, eliminates airborne exposure to hazardous reagents (e.g., ethanol, xylene, and chloroform), and ensures consistent tissue morphology preservation—critical for diagnostic-grade sectioning and downstream immunohistochemical analysis.
Key Features
- Fully stainless-steel conical pressure-rated processing chamber (5 L volume), designed for safe operation at up to 0.20 MPa and compatible with repeated thermal cycling from ambient to 99 °C.
- Triple-isolated fluidic pathways for reagents, cleaning solutions, and molten paraffin wax—preventing cross-contamination and reducing reagent consumption by up to 35% compared to non-enclosed systems.
- Integrated dual-activated carbon gas filtration system enabling continuous recirculation and purification of vapors during vacuum and pressure phases—meeting OSHA and EU Directive 2004/40/EC occupational exposure limits for volatile organic compounds.
- PTFE-reinforced sealing technology across all valves, manifolds, and cylinder interfaces—ensuring long-term integrity under cyclic thermal stress and chemical exposure to aggressive solvents including absolute ethanol, isopropanol, and xylene substitutes.
- Intelligent “no-cylinder-swap” fluid management: 17 dedicated reservoirs (9 reagent, 3 cleaning, 2 filter, 3 wax) enable fully automated multi-step protocols without manual intervention or hardware reconfiguration.
- “Fountain-style” agitation mechanism providing uniform fluid dynamics across the entire specimen basket—enhancing diffusion kinetics and eliminating edge-effect dehydration artifacts in dense or fibrous tissues.
- Real-time monitoring interface with 7-inch TFT-LCD display showing chamber temperature, internal pressure, elapsed time, fluid levels, and active protocol stage—supporting GLP-compliant audit trails via optional RS-232/USB data logging.
Sample Compatibility & Compliance
The HS-200 accommodates standard histology cassettes (up to 200 specimens per run) and supports diverse tissue types—including soft tissues (liver, kidney), calcified specimens (bone biopsies with decalcification pre-treatment), and delicate neural samples—without structural distortion. Its vacuum-controlled infiltration minimizes air entrapment in porous or fatty tissues, improving paraffin penetration uniformity. The system complies with ISO 13485:2016 (medical device quality management), meets electrical safety requirements per IEC 61010-1:2010, and supports implementation of FDA 21 CFR Part 11–compliant electronic records when paired with validated software modules. All wetted materials conform to USP Class VI biocompatibility standards.
Software & Data Management
The embedded control firmware provides 12 preloaded tissue processing protocols aligned with CAP and RCPA histopathology guidelines, plus 8 user-definable custom programs. Each protocol allows independent adjustment of temperature ramp rates, dwell times, vacuum hold durations, pressure application sequences, and agitation intervals. Parameter changes are logged with timestamp, operator ID, and reason-for-change fields—enabling full traceability required under GLP/GMP environments. Optional Ethernet connectivity enables remote status monitoring and centralized fleet management via secure HTTPS API. Data export supports CSV and XML formats compatible with LIS/HIS integration (e.g., Sunquest, Cerner, EPIC).
Applications
- Routine diagnostic histology in hospital pathology labs and reference laboratories.
- Preclinical research tissue processing in academic and pharmaceutical settings—particularly for longitudinal studies requiring inter-batch reproducibility.
- High-throughput tissue banking where standardized dehydration minimizes variability in archival quality.
- Specialized applications including electron microscopy sample preparation (via graded ethanol/xylene substitution) and RNA/DNA preservation workflows using low-temperature, low-vacuum protocols.
- Training and education platforms in histotechnology programs—featuring intuitive workflow visualization and error-state diagnostics.
FAQ
Does the HS-200 support validation documentation for ISO 13485 or GMP compliance?
Yes—the system includes a Factory Acceptance Test (FAT) report, IQ/OQ templates, and calibration certificates for temperature and pressure sensors. Full PQ support is available upon request.
Can the HS-200 be integrated into existing laboratory information systems (LIS)?
Yes—via optional HL7 or ASTM E1384-compliant interface modules supporting bidirectional specimen ID synchronization and result status reporting.
What maintenance is required for the vacuum pump and filtration system?
The oil-free diaphragm vacuum pump requires no routine oil changes; activated carbon filters should be replaced every 6 months or after 500 cycles, whichever occurs first.
Is training provided for operators and biomedical engineers?
ZEEDO offers on-site installation qualification and operator certification courses, including troubleshooting, preventive maintenance, and software configuration—delivered by certified field service engineers.
How does the HS-200 handle power interruptions during active processing?
The built-in UPS module sustains controller operation for ≥15 minutes, preserving current protocol state and initiating safe chamber venting if recovery exceeds threshold duration.