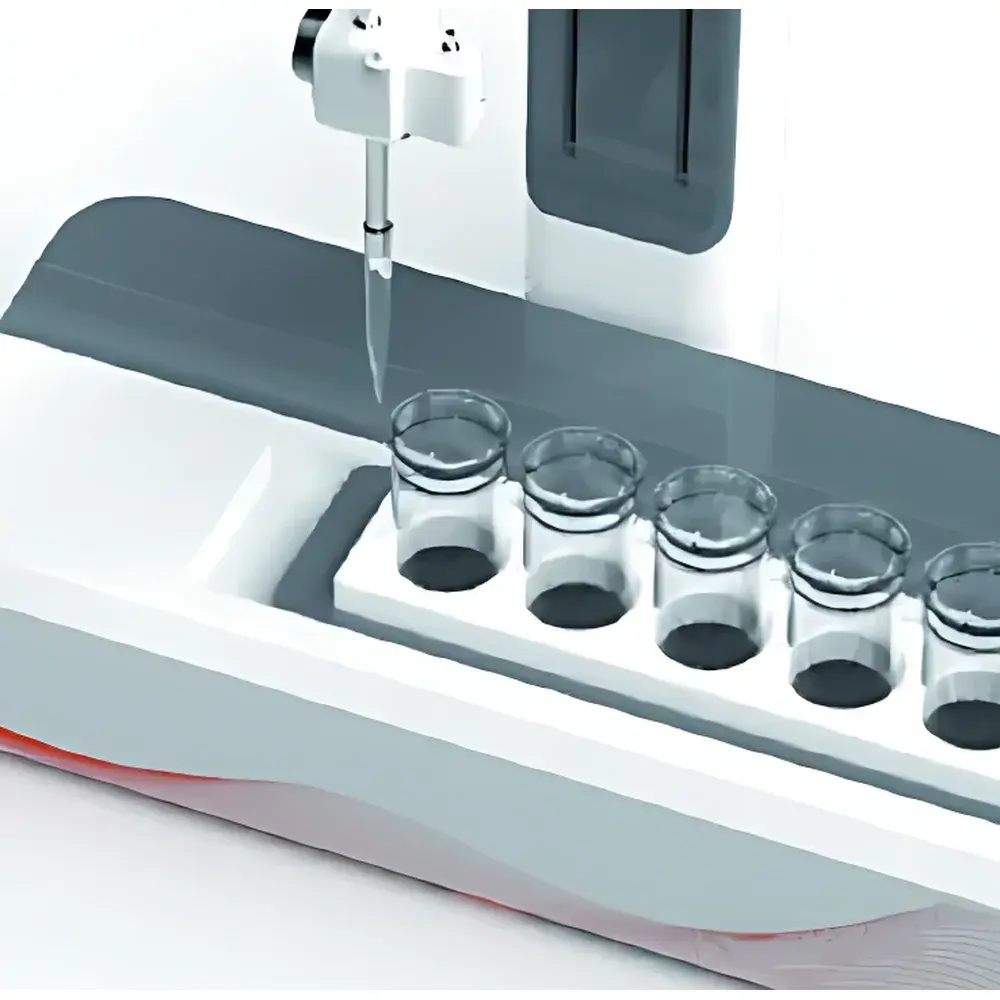
JIQUN MINI-X Automated Liquid Dilution System
| Brand | JIQUN |
|---|---|
| Origin | Jiangsu, China |
| Manufacturer Type | Authorized Distributor |
| Country of Manufacture | China |
| Model | MINI-X |
| Price Range | USD 14,000 – 28,000 |
| Liquid Handling Volume Range | 0.2–100+ mL |
| Sample Carrier Compatibility | Removable sample tray supporting 96-well microplates, vials, and tubes |
| Dispensing Accuracy | Engineered for high reproducibility in routine dilution workflows |
| Mixing Method | Integrated magnetic stirring module |
| Enclosure Material | Chemical-resistant engineering plastics |
| Control Interface | Detachable industrial-grade capacitive touchscreen |
| Installation Flexibility | Benchtop and fume hood-compatible footprint |
| Regulatory Alignment | Designed to support GLP-compliant documentation practices and audit-ready operation logs |
Overview
The JIQUN MINI-X Automated Liquid Dilution System is a compact, benchtop-grade instrument engineered for precise, repeatable liquid handling tasks in regulated and research laboratory environments. It operates on a gravimetric and volumetric hybrid dispensing principle—leveraging high-resolution peristaltic or syringe-based fluid delivery coupled with real-time volume feedback—to execute serial dilutions, reagent addition, standard solution preparation, and multi-step sample conditioning. Unlike manual pipetting or semi-automated systems, the MINI-X integrates dispensing, mixing, and method execution into a single coordinated workflow, minimizing human intervention and inter-operator variability. Its modular architecture supports deployment in QC labs, environmental testing facilities, public health laboratories, and academic core facilities where traceability, throughput, and ergonomic safety are critical operational requirements.
Key Features
- Modular, space-efficient design optimized for installation inside chemical fume hoods or on standard lab benches—footprint under 350 × 300 × 400 mm (W × D × H)
- Detachable capacitive touchscreen interface enabling remote control from outside hazardous containment zones
- Magnetic stirring integration ensures homogeneous mixing post-dispense without cross-contamination risk or mechanical agitation artifacts
- Interchangeable sample carriers accommodate diverse vessel formats: 96-well microplates, 1.5–50 mL centrifuge tubes, glass vials, and custom containers
- Multi-material compatibility: PEEK, quartz, stainless steel, and disposable polypropylene tips—validated for use with organic solvents, aqueous buffers, and corrosive reagents
- Automated calibration routines—including gravimetric verification and tip-volume profiling—support ISO/IEC 17025-aligned validation protocols
- Onboard method storage with timestamped version history; supports recall of up to 100 preconfigured protocols with user-defined naming and parameter locking
- Positioning system based on stepper-motor-driven XYZ gantry with optical end-stop detection—achieving sub-millimeter repeatability across all dispense positions
Sample Compatibility & Compliance
The MINI-X accepts samples across a broad physicochemical spectrum: aqueous solutions, ethanol/water mixtures, low-viscosity organic solvents (e.g., acetonitrile, methanol), and buffered biological matrices. Its fluid path components meet USP Class VI biocompatibility standards for non-leaching surfaces. While not certified as IVD or FDA 510(k)-cleared equipment, the system’s operational logs, method traceability, and electronic signature-ready interface align with principles outlined in FDA 21 CFR Part 11 for electronic records and signatures. Routine maintenance logs, calibration certificates, and performance qualification (PQ) templates are provided to facilitate GLP and ISO 17025 compliance documentation.
Software & Data Management
The embedded control software runs on a Linux-based real-time OS and provides full audit trail functionality—including operator ID, timestamp, method name, executed parameters, and error codes—with immutable logging enabled by default. Export options include CSV and PDF reports compatible with LIMS integration via configurable API endpoints. All methods are stored locally with SHA-256 hash integrity verification; optional network backup to secure NAS or cloud storage (via TLS 1.2+) is supported. Software updates are delivered through signed firmware packages verified at boot time.
Applications
- Preparation of certified reference material (CRM) dilution series for calibration of ICP-MS, HPLC, and UV-Vis spectrophotometers
- Automated dilution of environmental water samples prior to EPA Method 200.8 or ISO 17294-2 analysis
- Reagent addition and internal standard spiking in clinical toxicology workflows compliant with CLIA and CAP requirements
- Serial dilution of bacterial cultures or viral suspensions in biosafety level 2 (BSL-2) containment settings
- High-throughput buffer exchange and assay plate setup for ELISA, qPCR, and cell-based screening assays
FAQ
Does the MINI-X support two-point or multi-point calibration verification?
Yes—the system includes built-in gravimetric calibration routines with user-defined tolerance thresholds and automatic pass/fail reporting.
Can it be integrated into an existing LIMS or ELN environment?
Yes—via RESTful API with OAuth 2.0 authentication and configurable JSON payload schemas for method upload, run initiation, and result ingestion.
Is validation documentation provided for IQ/OQ/PQ execution?
Yes—comprehensive validation protocol templates, test scripts, and acceptance criteria aligned with ASTM E2500 and EU Annex 11 are included with shipment.
What is the maximum number of discrete steps supported within a single method?
Up to 99 sequential operations including dispense, mix, pause, conditional branching, and position move commands.
Are service contracts and remote diagnostics available internationally?
Yes—JIQUN partners with regional service providers across APAC, EMEA, and North America; remote diagnostics require customer-configured firewall exceptions for encrypted SSH tunneling.