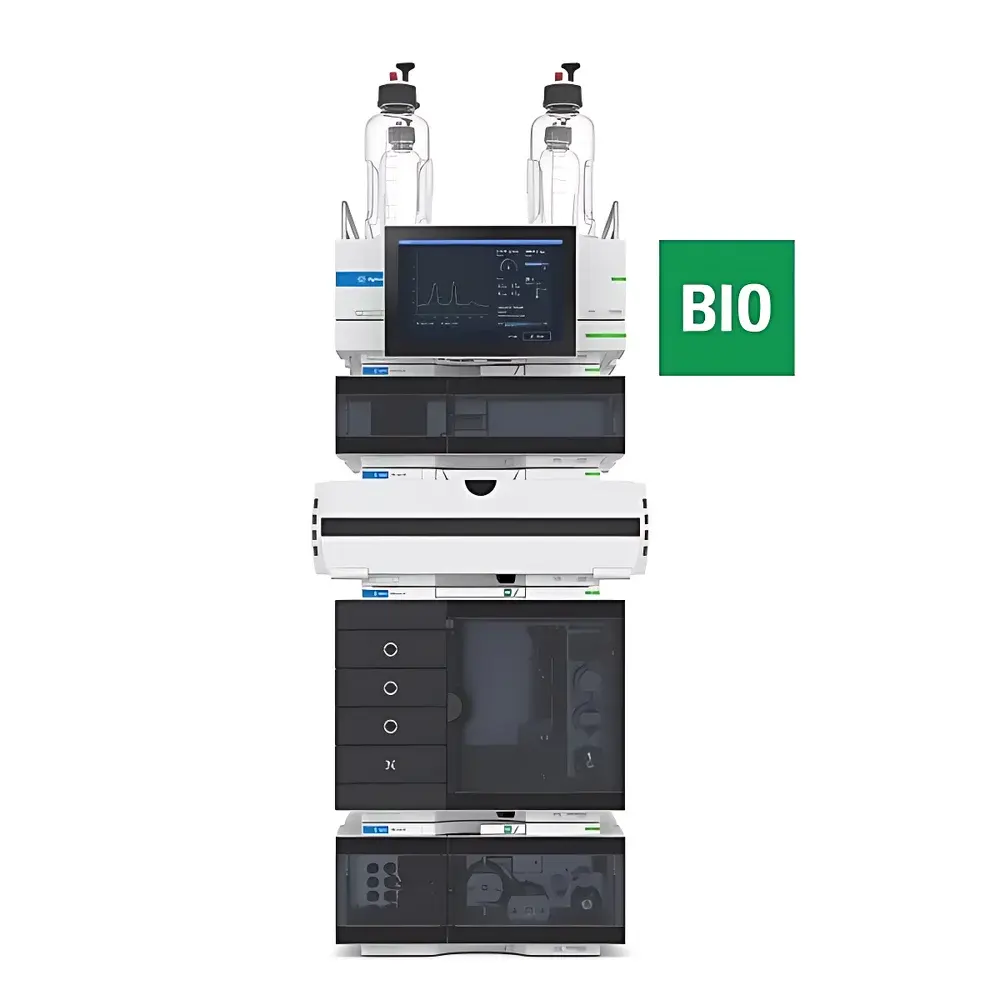
Agilent 1290 Infinity III Bio LC System
| Brand | Agilent Technologies |
|---|---|
| Origin | Germany |
| Manufacturer Type | Original Equipment Manufacturer (OEM) |
| Import Status | Imported |
| Model | 1290 Infinity III Bio LC |
| Instrument Type | Ultra-High-Performance Liquid Chromatograph (UHPLC) |
| Application Scope | Universal for biopharmaceutical and biomolecular analysis |
| Flow Rate Range | 0.001–5 mL/min |
| Maximum Operating Pressure | 1300 bar |
| Sample Capacity | Up to 6144 positions (microplate drawer) |
| Injection Volume Range | 0.001–500 µL (0.1–1500 µL with Multi-Draw Kit) |
Overview
The Agilent 1290 Infinity III Bio LC System is a purpose-engineered ultra-high-performance liquid chromatograph designed specifically for the rigorous demands of biopharmaceutical analysis. Built upon Agilent’s proven 1290 Infinity III platform, this system integrates full bio-inert fluidic pathways—constructed from titanium, PEEK, and specialized ceramic components—to eliminate metal-catalyzed degradation, adsorption, or aggregation of sensitive biomolecules such as monoclonal antibodies (mAbs), antibody-drug conjugates (ADCs), viral vectors, and oligonucleotides. Its operation is grounded in high-pressure liquid chromatography (HPLC) principles, leveraging sub-2-µm particle-packed columns under pressures up to 1300 bar to achieve exceptional resolution, peak capacity, and mass sensitivity—critical for characterizing critical quality attributes (CQAs) including size variants (SEC), charge heterogeneity (IEX), hydrophobicity (HIC), and glycosylation profiles (RP-LC). The system’s minimal dwell volume (<10 µL) and low dispersion design ensure precise gradient delivery and reproducible retention time alignment—essential for method transfer across laboratories and regulatory submissions.
Key Features
- Bio-inert flow path: Titanium pump heads, ceramic check valves, PEEK-silicone tubing, and passivated stainless-steel components minimize surface interactions and preserve biomolecule integrity across pH 1–14 and high-salt mobile phases.
- Ultra-high pressure capability: Stable operation at 1300 bar enables use of sub-2-µm and superficially porous particles (SPP) for maximum efficiency and speed without sacrificing robustness.
- High-throughput autosampling: 6144-position microplate drawer supports unattended operation for large-scale stability studies, lot release testing, and QC batch analysis.
- Flexible injection architecture: Programmable injection volumes from 0.001 µL to 500 µL (expandable to 1500 µL with Multi-Draw Kit) accommodate both nanoscale proteomics and preparative-scale purification workflows.
- Dual-gradient configuration: Optional binary or quaternary solvent manager allows method development versatility—from simple isocratic separations to complex multi-step gradients required for intact mass analysis or 2D-LC coupling.
- Thermal management: Integrated bio-optimized column oven maintains temperature stability ±0.1 °C from 5–110 °C, ensuring retention time reproducibility and column lifetime extension.
Sample Compatibility & Compliance
The 1290 Infinity III Bio LC is validated for compatibility with a broad spectrum of biologics—including mAbs, Fc-fusion proteins, bispecifics, peptides, plasmids, AAVs, and mRNA-LNPs—under physiologically relevant or stress-induced conditions. Its materials-of-construction comply with USP Class VI biocompatibility standards and meet ISO 10993-5 cytotoxicity requirements. All wetted surfaces are certified non-leaching per ASTM F2477 and demonstrate negligible metal ion elution (<0.1 ppb Ni, Cr, Fe) in ICP-MS validation studies. The system supports full audit trail, electronic signatures, and 21 CFR Part 11 compliance when operated with OpenLab CDS or MassHunter software—enabling GxP-aligned workflows in GLP, GMP, and FDA-submission environments.
Software & Data Management
Controlled via Agilent OpenLab CDS or MassHunter Workstation, the system delivers integrated instrument control, sequence management, peak integration, and reporting. Method templates include pre-configured SEC, IEX, HIC, and RP-LC methods compliant with ICH Q5, Q5A(R2), and USP <1058> analytical instrument qualification guidelines. Data integrity features include automated calibration logging, real-time error detection, and immutable audit trails with user-level access controls. Raw data files adhere to vendor-neutral mzML and ANDI-MS formats, facilitating third-party reprocessing and long-term archival in LIMS or ELN systems.
Applications
- Size-exclusion chromatography (SEC) for aggregate/fragment quantification in mAb therapeutics.
- Ion-exchange chromatography (IEX) for charge variant profiling and deamidation assessment.
- Hydrophobic interaction chromatography (HIC) for oxidation and misfolding analysis.
- Reverse-phase LC (RP-LC) of enzymatically digested peptides for peptide mapping and PTM characterization.
- Intact and reduced mass analysis coupled to high-resolution MS detectors.
- Stability-indicating assays under thermal, acidic, or oxidative stress conditions per ICH Q1A–Q5E.
FAQ
Is the 1290 Infinity III Bio LC compatible with standard UHPLC columns from other vendors?
Yes—the system supports all commercially available 2.1 mm and 3.0 mm ID UHPLC columns rated for ≥1300 bar, including those with silica, hybrid, and polymer-based stationary phases.
Can it be used for small-molecule analysis as well as biologics?
Absolutely—its universal gradient architecture, wide flow range (0.001–5 mL/min), and broad detection compatibility make it equally suitable for pharmaceutical small-molecule QC, impurity profiling, and dissolution testing.
Does the system support remote monitoring and predictive maintenance?
Yes—when connected to Agilent’s Intelligent Diagnostics module and Enterprise Informatics suite, it provides real-time status alerts, consumables life tracking, and failure mode prediction based on operational history and pressure/flow deviation trends.
What regulatory documentation is provided with the system?
Agilent supplies a complete IQ/OQ/PQ protocol package, material certification records (MCRs), biocompatibility test reports, and a Declaration of Conformity aligned with EU MDR 2017/745 and FDA 21 CFR Part 820.