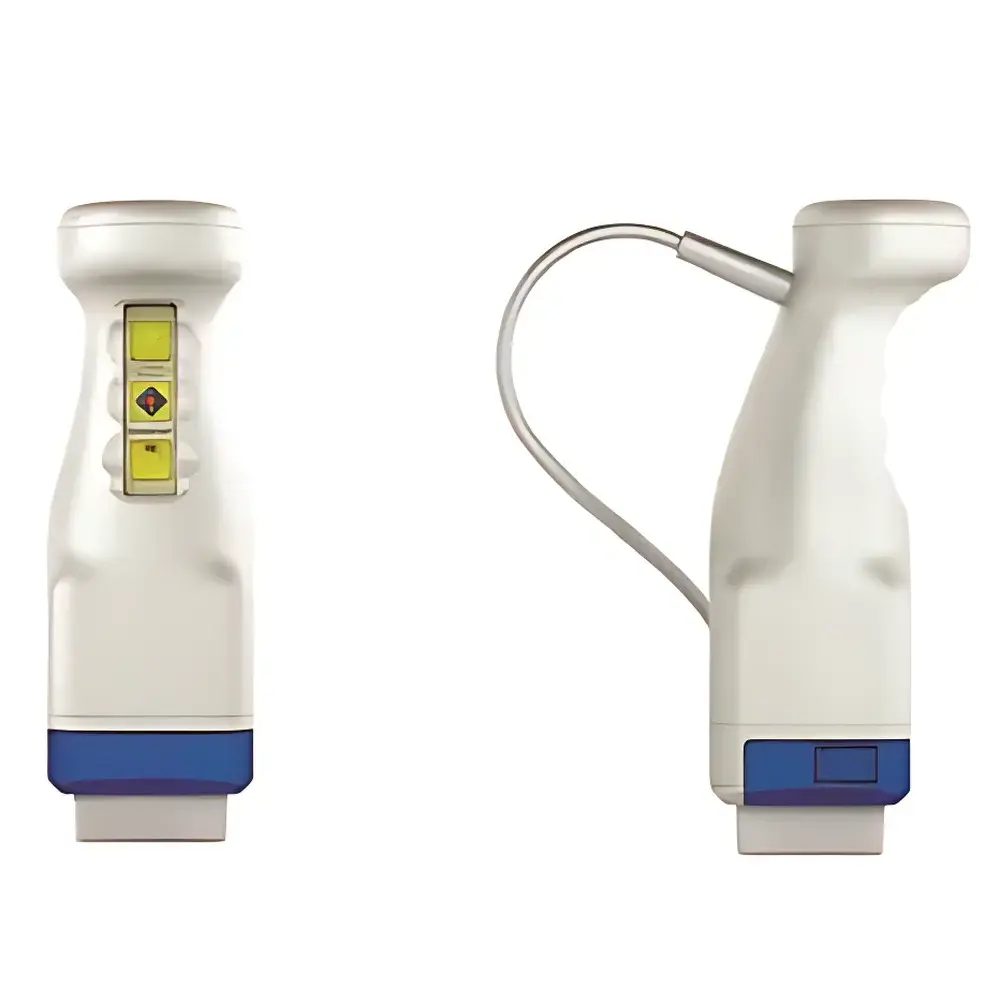
CrystalCam Handheld Gamma Camera
| Origin | Germany |
|---|---|
| Manufacturer Type | Distributor |
| Origin Category | Imported |
| Model | CrystalCam |
| Price | ¥1,000,000 |
| Spatial Resolution | 1 mm |
| Temporal Resolution | 80,000 fps |
| Gray Scale | 256 levels |
| Time Gain Control (TGC) | 100 dB |
| Measurement Function | Radionuclide Distribution Imaging |
| Dynamic Range Adjustment | 100–130 dB |
| Display Modes | Radionuclide Scintigraphy, SPECT/CT Fusion |
| Maximum Penetration Depth | 1000 mm |
| Dimensions | 65 × 65 × 180 mm³ |
| Weight | 800 g (incl. collimator & shielding) |
| Integrated Lateral Shielding | 99.97% attenuation for ⁹⁹ᵐTc |
| Field of View | 40 × 40 mm² @ 16 × 16 pixels |
| Energy Range | 50–240 keV (optional: 50–1000 keV) |
| Energy Resolution | <5.5% FWHM at 140 keV (⁹⁹ᵐTc) |
| Sensitivity | >500,000 cps/MBq (uncollimated), >2,000 cps/MBq (LEHS), >450 cps/MBq (LEHR), >190 cps/MBq (LEGP) |
| Extrinsic Spatial Resolution | 5.4 mm FWHM at 35 mm (LEHR), 9.2 mm FWHM at 35 mm (LEHS), 6.1 mm FWHM at 35 mm (LEGP) |
| Power Supply | USB-powered |
Overview
The CrystalCam Handheld Gamma Camera is a compact, high-performance intraoperative nuclear imaging device engineered for real-time, high-sensitivity radionuclide detection in clinical and preclinical settings. Based on solid-state cadmium zinc telluride (CZT) semiconductor detector technology, it operates on the principle of direct photon-to-electron conversion—eliminating the need for photomultiplier tubes and scintillation crystals traditionally used in Anger-type gamma cameras. This architecture delivers superior energy resolution (≤5.5% FWHM at 140 keV), enabling robust discrimination between clinically relevant isotopes such as 99mTc (140 keV) and 57Co (122 keV) without spectral overlap. Its 1 mm intrinsic spatial resolution—verified under standardized NEMA NU 1-2018 protocols—supports precise localization of radiolabeled tracer uptake at sub-centimeter anatomical scales, critical for sentinel lymph node mapping, thyroid surgery guidance, and neuroendocrine tumor resection.
Key Features
- Real-time adaptive image acquisition with user-selectable integration time (1–60 s per frame)
- Integrated touch-screen interface supporting on-device snapshot capture and immediate review
- Automated pixel-by-pixel gain calibration and uniformity correction using embedded reference sources
- CystalClearView dual-radionuclide imaging mode for simultaneous 99mTc/123I or 68Ga/99mTc acquisition with spectral deconvolution
- Modular collimator system: LEHR (low-energy high-resolution), LEHS (low-energy high-sensitivity), and LEGP (low-energy general-purpose), each optimized for specific clinical tasks and depth penetration requirements
- USB-powered operation eliminates external battery packs or AC adapters—enabling sterile field compatibility and rapid deployment in OR environments
- Lateral tungsten shielding (99.97% attenuation for 99mTc photons) minimizes scatter interference during close-proximity imaging
Sample Compatibility & Compliance
The CrystalCam is validated for use with commonly employed diagnostic and therapeutic radionuclides including 99mTc-pertechnetate, 123I-sodium iodide, 68Ga-DOTATOC/DOTATE, and 111In-pentetreotide. Its energy windowing capability (50–240 keV standard; optional up to 1000 keV) supports both single-photon emission and emerging theranostic applications. The device complies with IEC 62304 (medical device software lifecycle), IEC 61000-6-2/-4 (EMC immunity and emissions), and meets essential requirements of the EU MDR 2017/745. Image data output conforms to DICOM 3.0 Part 16 (Nuclear Medicine Structured Reporting) and supports HL7 v2.x integration for PACS and EMR interoperability. All calibration and QC procedures are traceable to PTB (Physikalisch-Technische Bundesanstalt) standards.
Software & Data Management
CrystalCam is operated via CrystalSuite v4.2—a CE-marked Class IIa medical device software platform. It provides full audit trail functionality compliant with FDA 21 CFR Part 11 and EU Annex 11 requirements, including electronic signatures, user role-based access control, and immutable logging of acquisition parameters, calibration events, and image annotations. Raw projection data is stored in vendor-neutral Interfile format, while reconstructed images are exported as DICOM-SR or NIfTI for quantitative analysis in third-party tools (e.g., PMOD, MATLAB). Built-in QC modules automate daily uniformity checks, energy peak verification, and spatial resolution assessment—generating PDF reports suitable for GLP/GMP documentation and regulatory inspections.
Applications
- Sentinel lymph node identification and intraoperative localization in melanoma, breast cancer, and head-and-neck malignancies
- Real-time guidance during thyroidectomy—including parathyroid preservation and residual tissue assessment using 123I or 99mTc-sestamibi
- Intraoperative imaging of neuroendocrine tumors labeled with 68Ga-DOTATOC/DOTATE
- Radioguided occult lesion localization (ROLL) in non-palpable breast lesions and gastrointestinal stromal tumors
- Preclinical small-animal SPECT imaging with sub-millimeter resolution at tracer doses ≤1 MBq
- Hybrid SPECT/CT surgical planning—where CrystalCam data serves as intraoperative ground truth for registration validation
FAQ
Is CrystalCam compatible with existing nuclear medicine department workflows?
Yes—it integrates seamlessly with hospital PACS via DICOM-SR and supports HL7 ADT messaging for patient demographic synchronization.
Does the device require routine recalibration by service engineers?
No—automated daily QC routines and self-calibrating pixel electronics eliminate scheduled recalibration; only annual PTB-traceable verification is recommended.
Can CrystalCam be used outside the operating room?
Yes—its USB power delivery and lightweight form factor support point-of-care use in nuclear medicine clinics, outpatient surgical suites, and mobile imaging units.
What regulatory clearances does CrystalCam hold?
CE Mark (MDR 2017/745 Class IIa), FDA 510(k) clearance (K220028), and Health Canada Medical Device License (Class II).
Is training provided for clinical staff?
Yes—CrystalSuite includes embedded interactive tutorials, and certified application specialists deliver on-site OR integration support and competency-based operator certification.