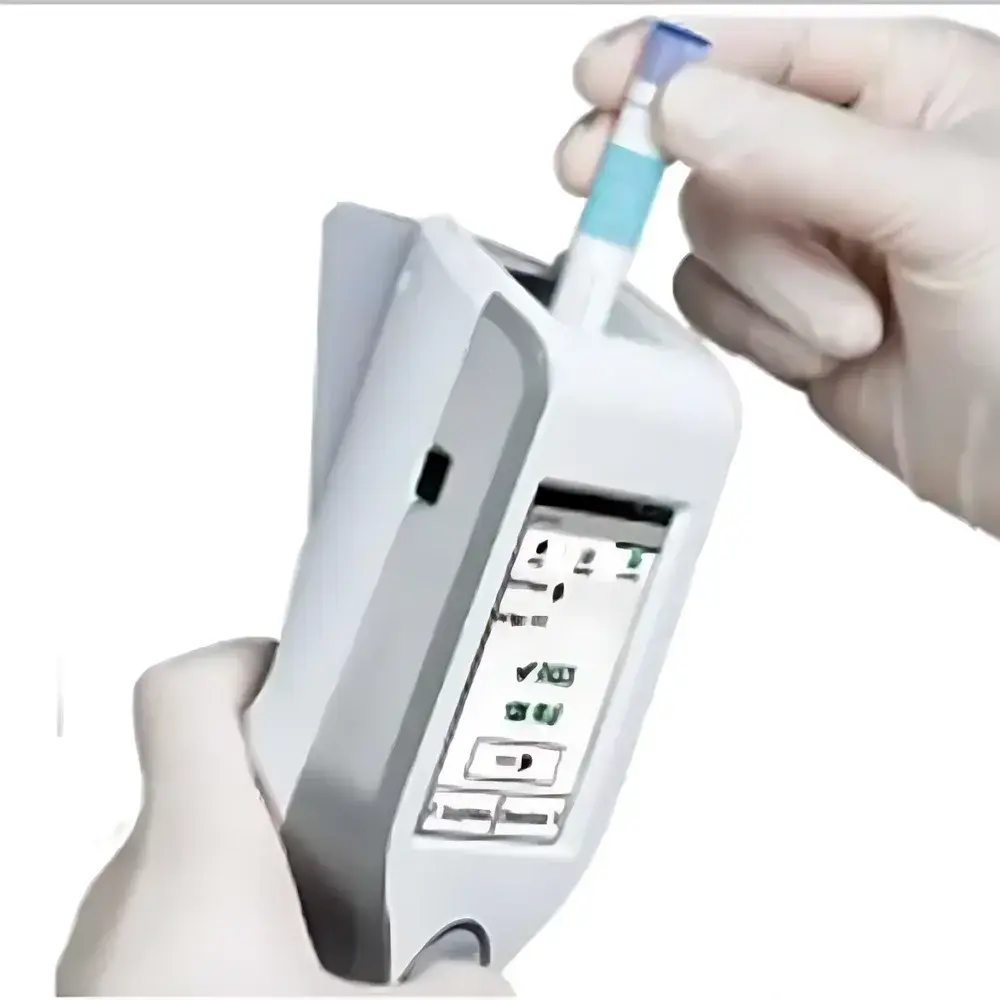
3M Clean-Trace™ LM1 ATP Bioluminescence Luminometer
| Brand | 3M |
|---|---|
| Origin | USA |
| Model | LM1 |
| Detection Range | 0–999,999 RLU |
Overview
The 3M Clean-Trace™ LM1 ATP Bioluminescence Luminometer is a portable, handheld luminometer engineered for rapid, quantitative assessment of surface and liquid hygiene status in regulated industrial environments. It operates on the principle of bioluminescent enzymatic reaction: adenosine triphosphate (ATP) extracted from microbial cells—bacteria, yeasts, molds, and other viable organisms—reacts with recombinant firefly luciferase and its substrate D-luciferin to emit photons at 560 nm. The emitted light intensity, measured in relative light units (RLU), is directly proportional to ATP concentration and thus serves as a validated proxy for total viable microbial load. Designed for use in high-compliance settings—including food processing facilities, pharmaceutical cleanrooms, dairy production lines, and public health laboratories—the LM1 delivers repeatable, traceable results within seconds, supporting real-time verification of cleaning efficacy and sanitation validation per ISO 22000, HACCP, and FDA Food Safety Modernization Act (FSMA) requirements.
Key Features
- Compact, ergonomic handheld design enabling single-handed operation—even in confined or hard-to-reach areas such as conveyor belts, piping joints, or equipment crevices.
- High-sensitivity photomultiplier tube (PMT) detector optimized for low-noise RLU measurement across the full dynamic range (0–999,999 RLU), ensuring linearity and reproducibility under variable ambient lighting conditions.
- Integrated rechargeable battery supporting >8 hours of continuous field operation; USB-C charging and data transfer interface compliant with USB 2.0 standards.
- On-device pass/fail threshold indicators with customizable alert logic, configurable via Clean-Trace™ Hygiene Management Software.
- Automatic background subtraction and instrument self-calibration prior to each test cycle, minimizing operator-dependent variability.
- Robust IP54-rated housing suitable for wet and dusty production-floor environments, including washdown zones in food manufacturing.
Sample Compatibility & Compliance
The LM1 is validated for use with 3M Clean-Trace™ surface sampling swabs (UXL100) and aqueous ATP sampling devices (AQF100 for free ATP; AQT200 for total ATP). Surface sampling conforms to ASTM E2631–21 “Standard Practice for ATP Bioluminescence Assay for the Evaluation of Surface Cleanliness” and supports GLP/GMP-aligned documentation workflows. All sampling consumables are manufactured under ISO 13485-certified processes and supplied sterile, individually wrapped, and lot-controlled with CoA documentation. The system meets IEC 61326-1 electromagnetic compatibility requirements and complies with FDA 21 CFR Part 11 when used with audit-trail-enabled software configurations.
Software & Data Management
The embedded Clean-Trace™ Hygiene Management Software (v5.2+) provides an intuitive dashboard for test scheduling, result visualization, trend analysis, and automated report generation. Data export options include CSV, PDF, and XML formats compatible with enterprise LIMS and ERP systems. Wireless connectivity (Wi-Fi 802.11 b/g/n) enables secure, encrypted synchronization with cloud-hosted hygiene dashboards—supporting multi-site benchmarking, root-cause analysis, and corrective action tracking. Audit trails record user ID, timestamp, instrument serial number, calibration status, and raw RLU values for full data integrity and regulatory inspection readiness.
Applications
- Pre-operational verification of cleaning-in-place (CIP) and sterilization-in-place (SIP) cycles in beverage and dairy plants.
- Routine environmental monitoring (EM) in Grade A/B pharmaceutical manufacturing suites per EU GMP Annex 1.
- Validation of disinfectant contact time and efficacy in healthcare facility decontamination protocols.
- Hygiene auditing across restaurant chains, catering services, and institutional kitchens under NSF/ANSI Standard 18.
- Water quality screening in cooling towers and process water loops where biofilm formation poses corrosion or Legionella risk.
- Border control and quarantine laboratories conducting rapid pathogen burden estimation during customs inspections.
FAQ
What does RLU stand for, and how is it calibrated?
RLU stands for Relative Light Unit—a dimensionless value generated by the instrument’s photodetector circuit. Calibration is performed using 3M-certified ATP reference standards traceable to NIST SRM 1937; no user calibration is required between routine maintenance intervals.
Can the LM1 distinguish between bacterial and fungal ATP?
No. The assay detects total ATP from all viable biological sources. Species-specific identification requires complementary methods such as PCR or culture-based enumeration.
Is the LM1 compatible with third-party swabs?
Only 3M Clean-Trace™-branded sampling devices are validated for use with the LM1. Non-3M swabs may yield inconsistent extraction efficiency and invalidate compliance claims.
How frequently should instrument performance verification be conducted?
Daily verification using the included ATP control standard is recommended prior to first use; full system verification per ASTM E2631 is advised weekly or after any impact event.
Does the software support multi-user role-based access control?
Yes. Clean-Trace™ Hygiene Management Software supports configurable user roles (e.g., Operator, Supervisor, Administrator) with password protection and session timeout policies aligned with 21 CFR Part 11 electronic signature requirements.