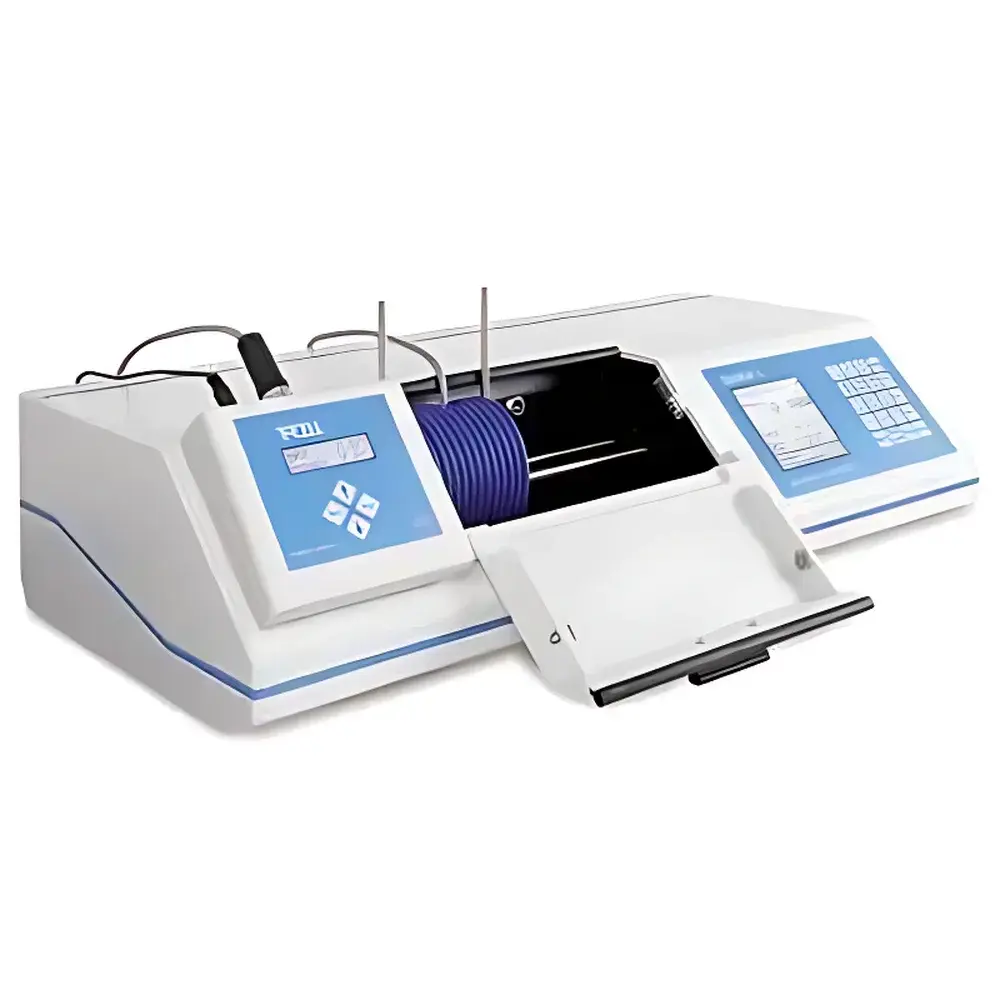
Schmidt+Haensch UniPol L-Series Automatic Polarimeter
| Brand | Schmidt+Haensch (S+H) |
|---|---|
| Country of Origin | Germany |
| Model | UniPol L-Series |
| Product Type | Automatic Polarimeter |
| Temperature Control | Peltier & optional water bath |
| Digital Display | Yes |
| Light Source | LED (589 nm, with 405 nm option on L 2000) |
| Measurement Range | ±360°, ±259°Z |
| Resolution | 0.001° / 0.01°Z (L 2000), 0.01° / 0.02°Z (L, L 1000) |
| Accuracy | ±0.005° / ±0.02°Z (L 2000), ±0.01° / ±0.03°Z (L, L 1000) |
| Temperature Range | 0–99 °C |
| Temperature Resolution | 0.01 °C |
| Temperature Accuracy | ±0.03 °C |
| Compliance | USP <781>, Ph. Eur. 2.2.7, ASTM E1254, GLP/GMP, FDA 21 CFR Part 11 (with Aquisys 2008 software), PTB-certified quartz calibration cells |
Overview
The Schmidt+Haensch UniPol L-Series Automatic Polarimeter is a high-precision optical instrument engineered for the quantitative measurement of optical rotation — the angular deviation of plane-polarized light induced by chiral substances in solution. Based on the fundamental principle of polarimetry, the system employs a stable, narrow-band LED light source at the sodium D-line wavelength (589 nm), optionally extended to 405 nm for specialized applications requiring enhanced spectral discrimination. The instrument utilizes a high-stability Glan-Taylor polarizer and precision half-wave retarder assembly, coupled with a digital photoelectric detection system to resolve minute angular shifts with sub-millidegree resolution. Designed for routine and regulated laboratory environments, the UniPol L-Series delivers traceable, reproducible measurements compliant with pharmacopoeial standards including USP , Ph. Eur. 2.2.7, and ASTM E1254. Its robust mechanical architecture — built upon over 150 years of German optical engineering heritage dating back to 1864 — ensures long-term stability, minimal drift, and metrological integrity across daily operation.
Key Features
- Four independent measurement methods with up to ten user-definable scale units (e.g., °, °Z, % w/w, custom coefficients)
- Configurable display interface: each method supports independent parameter settings including averaging count, delay time, and pass/fail thresholds
- Integrated optical density monitoring to detect sample turbidity or absorption-related artifacts in real time
- Automated statistical evaluation of repeated measurements (mean, SD, CV%, min/max, n-count)
- Programmable kinetic mode for time-resolved tracking of polarization changes during enzymatic reactions, hydrolysis, or crystallization processes
- Peltier-based temperature control (0–99 °C) with embedded Pt100 sensor; automatic temperature correction applied to raw readings per ICH Q5C and Ph. Eur. guidelines
- Direct printer connectivity, Ethernet interface, and remote PC control via RS-232 or TCP/IP
- PTB-certified quartz reference cells supplied as standard, enabling traceable calibration against national metrology institutes
Sample Compatibility & Compliance
The UniPol L-Series accommodates standard 100 mm and 200 mm sample tubes, with optional micro-cell support (available on L 1000 and L 2000 models) for volumes as low as 0.5 mL. Sample compatibility extends across aqueous, alcoholic, and organic solvent systems — provided optical clarity and absence of particulate scatter are maintained. Instrument validation protocols include full IQ/OQ/PQ documentation packages aligned with GMP Annex 15 and ISO/IEC 17025 requirements. When operated with Aquisys 2008 software, the system satisfies FDA 21 CFR Part 11 criteria for electronic records and signatures, including audit trail generation, user access control, and data integrity safeguards. All firmware and calibration routines are version-controlled and validated per GLP principles.
Software & Data Management
Aquisys 2008 is the dedicated PC application for advanced configuration, method development, and regulatory-compliant data handling. It provides full method storage, customizable report templates (PDF/CSV/XLS), electronic signature workflows, and automated backup to network drives or cloud repositories. The software supports multi-instrument fleet management, trend analysis over time, and export of raw detector signals for secondary processing. Audit trails record all user actions — including parameter edits, calibration events, and result approvals — with immutable timestamps and operator IDs. Data files are cryptographically hashed to ensure non-repudiation and tamper evidence, fulfilling ALCOA+ (Attributable, Legible, Contemporaneous, Original, Accurate, Complete, Consistent, Enduring, Available) data governance expectations.
Applications
The UniPol L-Series serves critical quality assurance functions across multiple regulated sectors. In pharmaceutical manufacturing, it verifies enantiomeric purity of active pharmaceutical ingredients (APIs) per USP monographs and supports stability-indicating assays for chiral degradation products. In food and beverage production — particularly sugar refining, dairy processing, and wine analysis — it quantifies sucrose, lactose, and fructose concentrations with direct correlation to °Z (Zuckergrad) scales. Cosmetic manufacturers apply it to assess batch-to-batch consistency of optically active essential oils (e.g., limonene, menthol) and synthetic chiral fragrances. Chemical R&D labs utilize its kinetic mode to monitor asymmetric synthesis progress or polymer helix formation in real time. Academic and governmental research institutions rely on its metrological traceability for structural elucidation studies involving chiral catalysts, biomacromolecules, and liquid crystalline phases.
FAQ
What regulatory standards does the UniPol L-Series meet?
It complies with USP , Ph. Eur. 2.2.7, ASTM E1254, and supports GLP/GMP workflows. With Aquisys 2008, it fulfills FDA 21 CFR Part 11 requirements for electronic records.
Is temperature control mandatory for accurate measurement?
Yes — optical rotation is temperature-dependent. The integrated Peltier system maintains ±0.03 °C stability and applies real-time correction per standardized reference temperatures (e.g., 20 °C or 25 °C).
Can the instrument measure samples with low optical transmission?
The built-in optical density monitoring alerts users when absorbance exceeds safe thresholds, preventing erroneous rotation readings due to signal attenuation.
How is calibration verified and maintained?
Each unit ships with PTB-traceable quartz control plates. Users perform daily verification using these standards; calibration certificates are issued per ISO/IEC 17025 by authorized service centers.
What sample volume is required for micro-cell measurements?
The optional micro-cell accessory (available for L 1000 and L 2000) requires only 0.5 mL for 100 mm pathlength measurements, minimizing reagent consumption and waste.