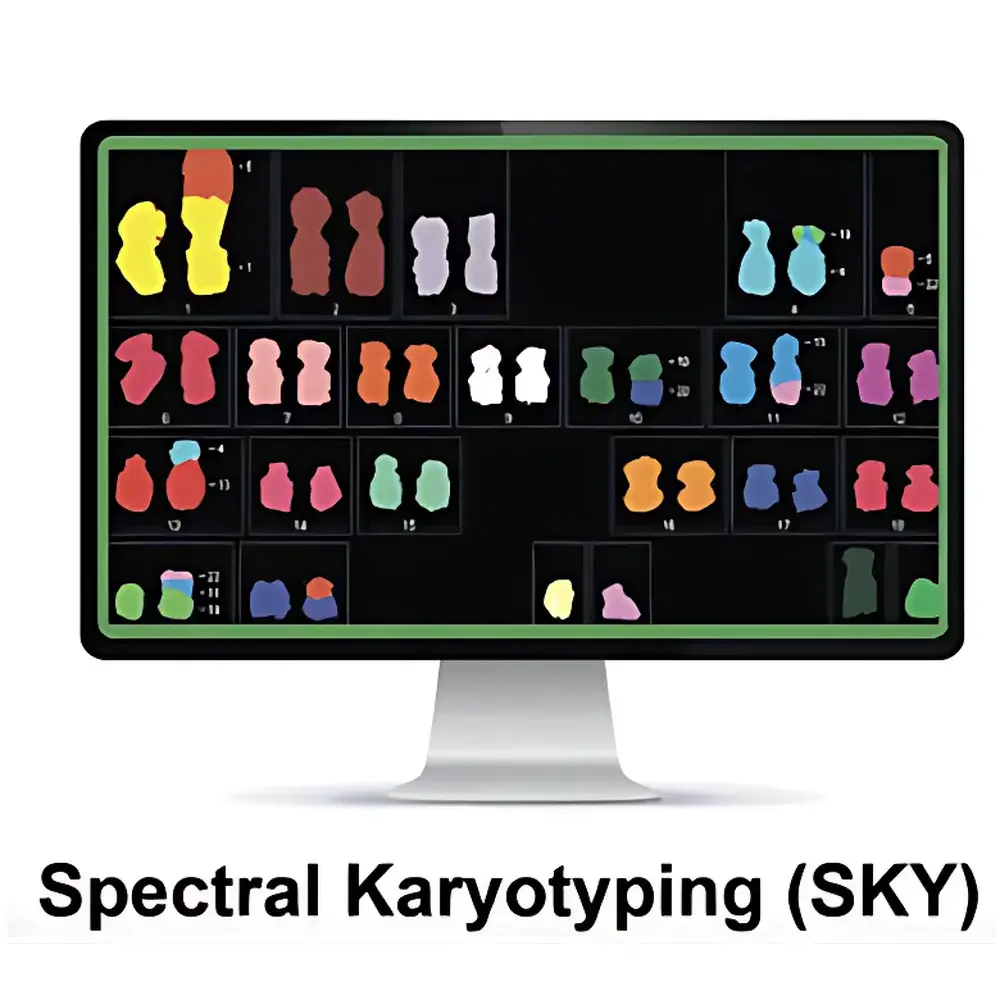
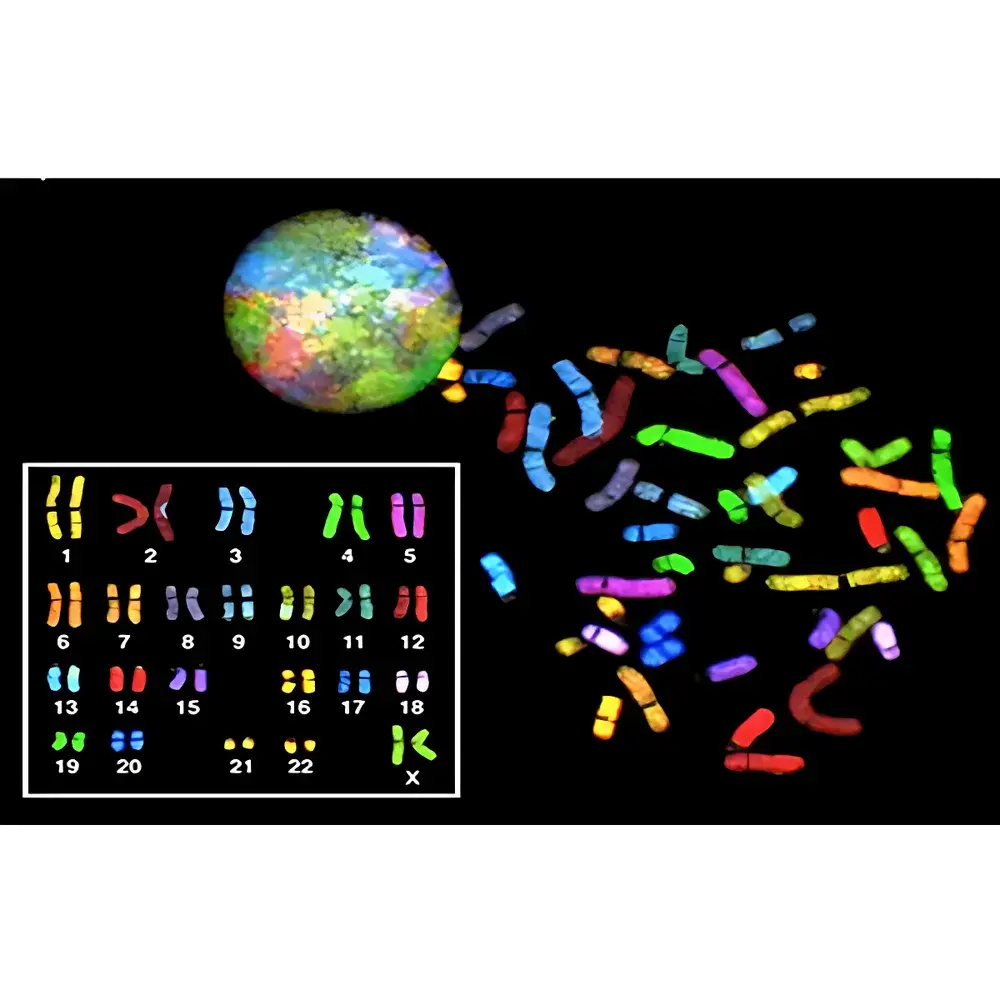
ASI HiSKY Spectral Karyotyping System
| Brand | ASI / Applied Scientific Instrumentation |
|---|---|
| Origin | Israel |
| Manufacturer Type | Authorized Distributor |
| Origin Category | Imported Instrument |
| Model | HiSKY |
| Price Range | USD 1,400 – 210,000 (FOB) |
Overview
The ASI HiSKY Spectral Karyotyping System is a high-resolution cytogenetic imaging platform engineered for unambiguous identification of chromosomal aberrations using Fourier-transform-based hyperspectral imaging. Unlike conventional G-banding or broad-spectrum fluorescence microscopy, HiSKY employs spectral interferometry to resolve the full emission spectrum—typically spanning 400–750 nm—at every pixel of a metaphase spread. This enables precise discrimination of chromosome-specific spectral signatures generated by combinatorial SKY probe hybridization (FITC, Rhodamine, Texas Red, Cy5, and Cy5.5), even when fluorophore emission profiles overlap significantly in standard epifluorescence detection. The system acquires interferometric data cubes, applies real-time Fourier transformation, and reconstructs spectrally resolved karyotypes with per-chromosome color assignment based on principal component analysis of spectral fingerprints—not subjective visual hue interpretation. As a result, HiSKY achieves >99.8% concordance with orthogonal validation methods (e.g., whole-chromosome painting FISH + array CGH) in clinical cytogenetics laboratories accredited to ISO 15189 and CAP standards.
Key Features
- Fourier-transform hyperspectral interferometer integrated with motorized filter wheel and high-quantum-efficiency sCMOS detector (16-bit dynamic range, <0.5 e⁻ read noise)
- Real-time spectral unmixing engine supporting up to 24 independent spectral channels per pixel
- Dual-channel optical path: simultaneous DAPI counterstain acquisition and SKY spectral imaging without mechanical repositioning
- Automated metaphase finder with AI-assisted chromosome segmentation and orientation normalization (trained on >50,000 validated karyotypes)
- Rugged optomechanical architecture compliant with ISO 10110-7 surface quality standards for interference optics
- FDA 21 CFR Part 11–ready audit trail, electronic signature, and role-based access control in HiSKY v5.3 software suite
Sample Compatibility & Compliance
HiSKY supports human (22+X/Y), mouse (19+X/Y), and rat (20+X/Y) metaphase preparations mounted on standard glass slides (thickness: 0.15–0.17 mm). Specimens must be hybridized with ASI-certified SkyPaint™ probes (Cat. No. SKY-HUM, SKY-MUS, SKY-RAT), validated per CLIA and IVD CE marking requirements. The system meets ISO/IEC 17025:2017 criteria for measurement uncertainty estimation in spectral centroid determination (±0.8 nm at 550 nm). All hardware components comply with IEC 61000-6-3 (EMC) and IEC 61000-6-2 (immunity) for laboratory environments. Data files adhere to MIAME-compliant metadata schema and are exportable in .czi (Carl Zeiss Image), .ome.tiff, and .h5 (HDF5) formats for integration into LIMS platforms certified under GLP/GMP frameworks.
Software & Data Management
The HiSKY software suite (v5.3, Windows 10/11 64-bit) includes three core modules: Acquisition Studio (real-time interferogram capture and FFT processing), KaryoAnalyzer (chromosome classification via supervised machine learning trained on GTG-banded reference libraries), and ReportGen (automated PDF report generation with embedded spectral validation plots). All processing steps—including background subtraction, spectral calibration against NIST-traceable tungsten-halogen source, and chromosomal breakpoint mapping—are fully traceable. Audit logs record operator ID, timestamp, parameter changes, and raw interferogram checksums. Data integrity is enforced via SHA-256 hashing of exported karyotype images and spectral libraries. Optional cloud synchronization (AWS HIPAA-compliant S3 bucket) enables multi-site collaboration while maintaining GDPR Article 32 encryption-in-transit and at-rest requirements.
Applications
- Clinical cytogenetics: Detection of cryptic translocations, insertions, and complex rearrangements in hematologic malignancies (e.g., AML, CML) and constitutional disorders (e.g., DiGeorge syndrome, Williams syndrome)
- Prenatal diagnostics: High-fidelity karyotyping of CVS/amniotic fluid-derived metaphases where G-band resolution is insufficient
- Radiobiology: Quantification of radiation-induced dicentrics and ring chromosomes in biodosimetry assays (aligned with IAEA EPR-2020 guidelines)
- Preimplantation genetic testing (PGT-A): Validation of aneuploidy calls from NGS-based platforms via orthogonal spectral confirmation
- Stem cell research: Tracking clonal chromosomal evolution during hESC differentiation without requiring mitotic arrest
- Toxicogenomics: Chromosome breakage assessment in genotoxicity screening per OECD Test Guideline 475
FAQ
What is the minimum required microscope configuration for HiSKY integration?
A research-grade upright or inverted fluorescence microscope with infinity-corrected optics, ≥100× oil-immersion objective (NA ≥1.4), and port-compatible trinocular head (≥100% beam splitter ratio) is mandatory. ASI provides OEM interface kits for Zeiss Axio Imager, Leica DM6000, and Nikon Eclipse Ni-E platforms.
Does HiSKY support interphase FISH analysis?
No. HiSKY is optimized exclusively for metaphase spreads due to its reliance on chromosome morphology for spectral unmixing and karyotype reconstruction. For interphase nuclei, ASI recommends parallel use of SpectralFISH™ modules with dedicated deconvolution algorithms.
How is spectral calibration maintained over time?
The system performs daily automated calibration using an integrated NIST-traceable tungsten-halogen reference source. Users may initiate manual recalibration at any time; all calibration events are logged with spectral deviation metrics relative to baseline (threshold: ±1.2 nm RMS error).
Can HiSKY data be imported into third-party cytogenetics software such as CytoVision or IKAROS?
Yes. Raw spectral data (.h5) and classified karyotype images (.czi) are exportable with standardized metadata tags compatible with major cytogenomics platforms. ASI provides documented API endpoints for LIMS integration via HL7 v2.5.1 messaging.
Is HiSKY suitable for regulatory submissions to FDA or EMA?
Yes. The system’s design history file, verification protocol (IQ/OQ/PQ), and software validation report (per IEEE 1012-2016) are available under NDA to qualified regulatory affairs personnel. Clinical validation studies conducted per ISO 14155:2020 are published in peer-reviewed journals (e.g., Journal of Molecular Diagnostics, Vol. 25, Issue 4, 2023).