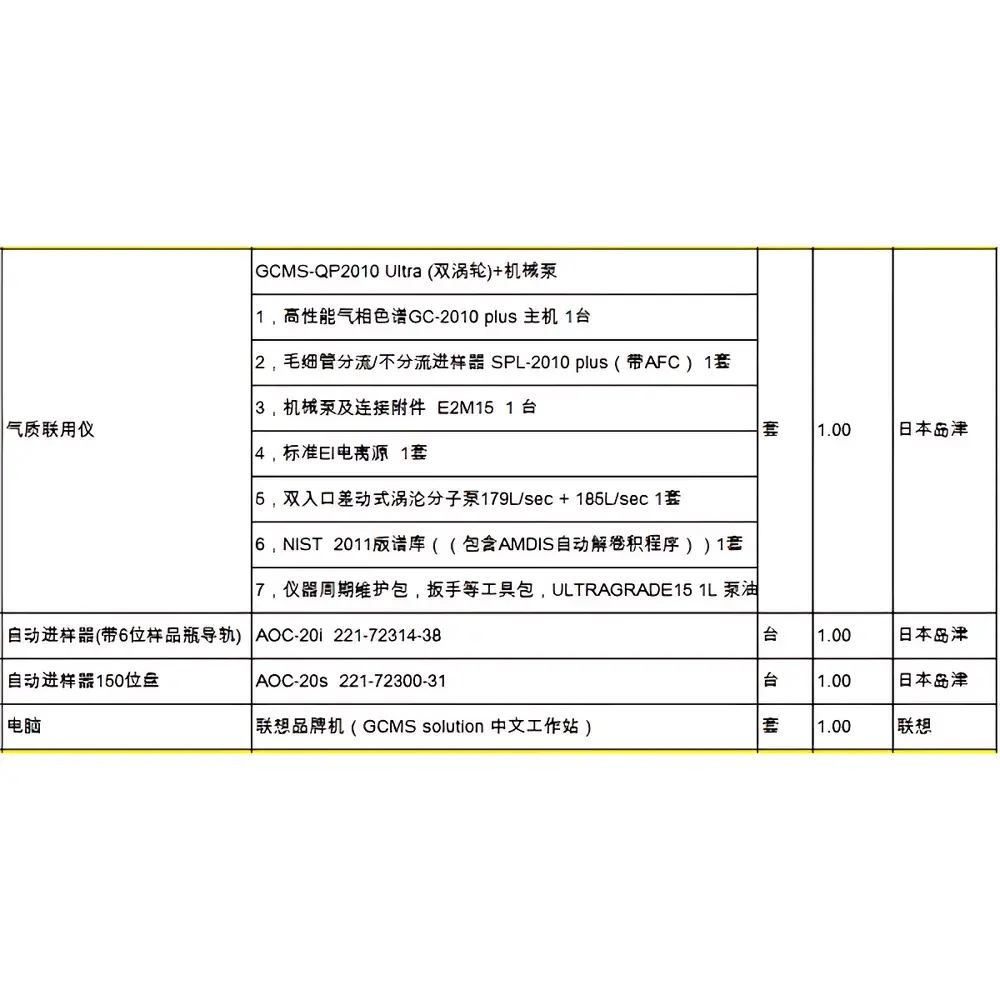
Shimadzu GCMS-QP2010 Ultra Used Gas Chromatography-Mass Spectrometry System
| Brand | Shimadzu |
|---|---|
| Origin | Japan |
| Manufacturer Type | Authorized Distributor |
| Origin Category | Imported Instrument |
| Model | QP2010 Ultra |
| Price | USD 27,500 (approx.) |
Overview
The Shimadzu GCMS-QP2010 Ultra is a high-performance, benchtop gas chromatography-mass spectrometry (GC-MS) system originally engineered for routine and research-grade organic compound identification, quantification, and structural elucidation. Built upon Shimadzu’s proprietary quadrupole mass analyzer architecture and advanced electron ionization (EI) source design, the QP2010 Ultra delivers enhanced sensitivity, improved mass accuracy, and robust spectral reproducibility. Its core measurement principle combines capillary gas chromatographic separation with unit-mass resolution quadrupole mass filtering and detection under controlled vacuum conditions (typically 1×10⁻⁵ Pa). Designed for laboratories requiring regulatory-compliant trace analysis—particularly in environmental monitoring, food safety testing, forensic toxicology, and pharmaceutical impurity profiling—the system supports both full-scan and selected ion monitoring (SIM) acquisition modes with real-time data processing.
Key Features
- Quadrupole mass analyzer with mass range of m/z 1.5–1090, optimized for high transmission efficiency and low chemical noise
- High-efficiency EI ion source operating at 70 eV, with optional CI (chemical ionization) capability via field-replaceable source module
- Advanced AART (Automatic Adjustment of Retention Time) algorithm for reliable peak alignment across multi-batch analyses
- Integrated GC oven with programmable temperature ramping (range: 4–450 °C; ramp rate up to 250 °C/min)
- Auto-tuning function for automatic optimization of detector gain, electron multiplier voltage, and mass calibration
- Robust vacuum system featuring dual-stage turbomolecular pump (67 L/s) coupled with dry scroll backing pump for stable operation and rapid pump-down
- Compliance-ready architecture supporting audit trail, user access control, and electronic signature functions when configured with GCMSsolution Ver. 2.7 or later
Sample Compatibility & Compliance
The GCMS-QP2010 Ultra accepts standard 0.25–0.53 mm ID fused-silica capillary columns (e.g., DB-5ms, HP-5MS, Rtx-5Sil MS) and accommodates liquid, solid (via thermal desorption or derivatization), and gaseous samples following appropriate sample introduction protocols (split/splitless injection, PTV, or headspace). It meets essential performance criteria outlined in ASTM D5845 (determination of volatile organic compounds in water), EPA Method 8270D (semivolatile organics in solid waste), and USP (chromatography). When deployed in GLP- or GMP-regulated environments, the system—paired with validated GCMSsolution software—supports 21 CFR Part 11 compliance through configurable security settings, instrument log archiving, and electronic record integrity safeguards.
Software & Data Management
Controlled exclusively via Shimadzu’s GCMSsolution platform (Windows-based), the QP2010 Ultra provides integrated method development, real-time spectral acquisition, library searching (NIST/EPA/WHO/Wiley libraries included), and quantitative report generation. Data files (.qgd format) are structured for long-term archival and interoperability with third-party chemometric tools. The software implements role-based user management, session logging, and automated backup scheduling. Raw data retention, processing history tracking, and version-controlled method storage align with ALCOA+ (Attributable, Legible, Contemporaneous, Original, Accurate, Complete, Consistent, Enduring, Available) data integrity principles required in regulated QC/QA workflows.
Applications
- Residue analysis of pesticides and veterinary drugs in agricultural commodities per EU SANTE/11813/2021 guidelines
- Identification and quantification of polycyclic aromatic hydrocarbons (PAHs) in soil and sediment extracts
- Screening of illicit drugs and metabolites in biological matrices (urine, blood) using validated SIM methods
- Impurity profiling of active pharmaceutical ingredients (APIs) in accordance with ICH Q3B(R2)
- Volatile organic compound (VOC) emission testing from building materials per ISO 16000-6
- Essential oil composition analysis via retention index matching and spectral deconvolution
FAQ
Is this unit fully refurbished and performance-verified before shipment?
Yes. Each QP2010 Ultra undergoes comprehensive functional validation—including vacuum integrity test, mass calibration verification, sensitivity check using decafluorotriphenylphosphine (DFTPP), and chromatographic resolution assessment—per Shimadzu’s pre-delivery checklist.
What documentation accompanies the instrument?
Customers receive a detailed refurbishment report, calibration certificate traceable to NIST standards, original operation manual (English), GCMSsolution installation media, and a 12-month limited hardware warranty.
Can the system be upgraded to support modern data security requirements?
Yes. Firmware updates to GCMSsolution Ver. 2.8x and later enable enhanced 21 CFR Part 11 compliance features, including biometric-compatible login modules and encrypted audit trail export.
Are consumables and spare parts still available?
All critical consumables—including EI filaments, quadrupole rods, column nuts, septa, and O-rings—are actively stocked by Shimadzu Global Parts Center and authorized regional distributors.
Does the system include a computer and monitor?
A dedicated Windows 10 Pro workstation with dual-monitor support, preloaded with GCMSsolution and licensed antivirus software, is included as standard configuration.