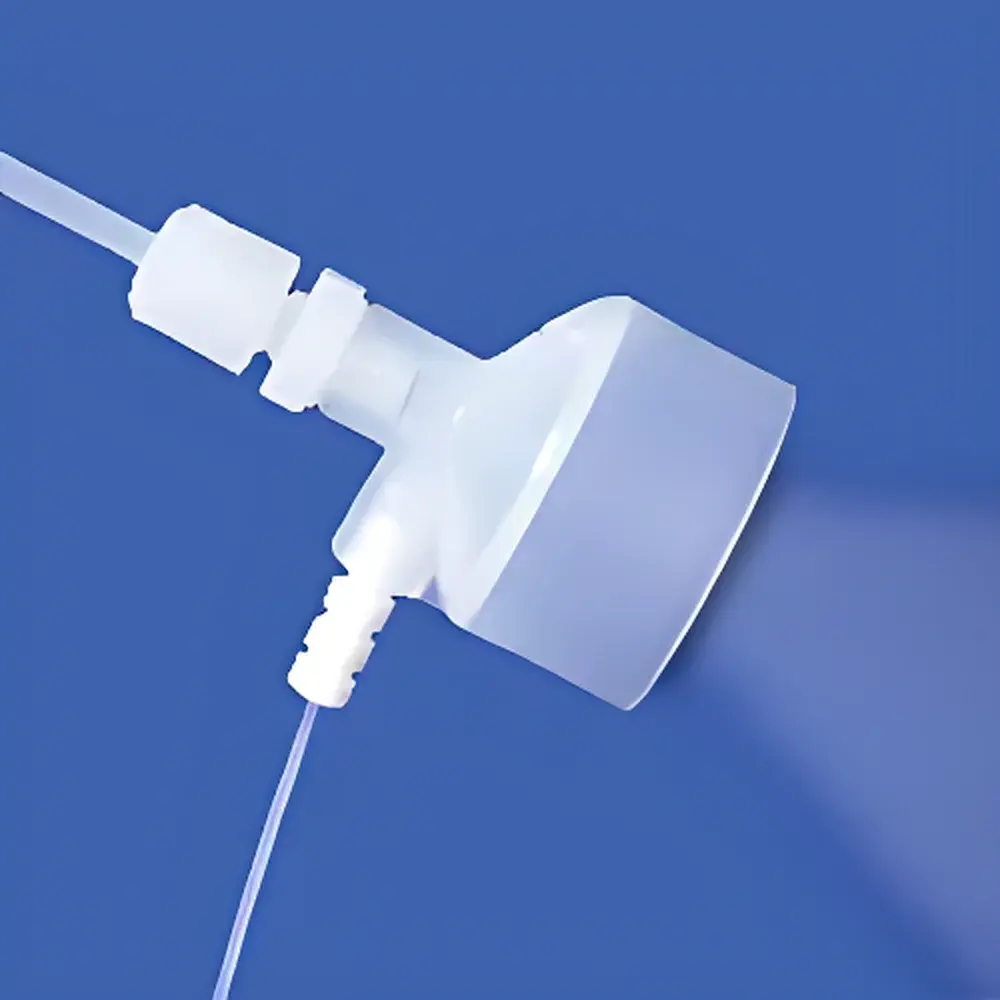
Savillex C-Flow PFA Concentric Nebulizer for ICP-MS
| Brand | Savillex |
|---|---|
| Origin | USA |
| Model | C-Flow |
| Flow Rates | 50 µL/min and 200 µL/min |
| Material | Perfluoroalkoxy (PFA) |
| Compliance | Compatible with Scott-type spray chambers (35 mm), O-ring-free design |
| Certification | Manufactured under ISO 9001-certified processes, compliant with ASTM D5673 and USP <232>/<233> for trace metal analysis |
Overview
The Savillex C-Flow PFA Concentric Nebulizer is a high-performance, chemically inert sample introduction component engineered specifically for ultra-trace elemental analysis by inductively coupled plasma mass spectrometry (ICP-MS) and inductively coupled plasma optical emission spectrometry (ICP-OES). Based on the fundamental principle of pneumatic concentric nebulization—where a pressurized carrier gas shears liquid sample into fine, uniform aerosol droplets—the C-Flow delivers exceptional aerosol generation efficiency across low to moderate uptake rates (50–200 µL/min). Its all-PFA construction eliminates metallic or siliceous contamination pathways, ensuring baseline integrity in sub-ppt-level measurements. Unlike conventional quartz or nickel-alloy nebulizers, the C-Flow’s monolithic PFA body exhibits near-zero memory effects, negligible blank contributions, and resistance to hydrofluoric acid, strong oxidants, and high-salt matrices—critical for environmental, geological, nuclear, and clinical applications where sample purity and instrument uptime are paramount.
Key Features
- Monolithic perfluoroalkoxy (PFA) construction—no metal, ceramic, or adhesive components; fully inert to HF, HNO₃, HCl, aqua regia, and alkaline digestates
- Optimized concentric geometry with precision-machined capillary and gas orifice enabling stable aerosol generation at flow rates as low as 50 µL/min
- Patented straight-through fluid path minimizes clogging risk—even with samples containing up to 0.2% total dissolved solids (TDS)
- O-ring-free interface compatible with standard 35 mm Scott-type spray chambers, eliminating silicone leaching and vacuum seal degradation
- Batch-controlled manufacturing with ≤±2.5% inter-unit aerosol output reproducibility (measured via ¹¹⁵In signal stability over 4-hour runs)
- Thermal stability from –200 °C to +260 °C; pressure-rated to 100 psi; compatible with both peristaltic and syringe pump sample introduction
Sample Compatibility & Compliance
The C-Flow nebulizer supports a broad range of sample types including acid-digested environmental waters, high-purity semiconductor process chemicals, biological digests (e.g., urine, serum), and geochemical reference materials (e.g., NIST SRM 1640a, SLRS-6). Its PFA composition meets stringent requirements for ISO/IEC 17025-accredited laboratories performing trace element analysis per EPA Method 6020B, ASTM D5673 (for drinking water), and USP / (elemental impurities in pharmaceuticals). The absence of extractable organics or metals ensures compliance with GLP/GMP data integrity standards, including full audit trail support when integrated with ICP-MS systems configured for FDA 21 CFR Part 11 compliance.
Software & Data Management
While the C-Flow itself is a passive hardware component, its performance directly impacts key analytical metrics tracked within ICP-MS acquisition software (e.g., Thermo Fisher Qtegra, Agilent MassHunter, PerkinElmer Syngistix). Stable nebulization translates to improved RSDs (24 h without recalibration for multi-element analyses). When paired with Savillex X-Flow lateral-flow nebulizers or dual-channel Scott spray chambers, users can implement automated matrix-matched drift correction protocols embedded in instrument control software—enhancing long-term measurement robustness without manual intervention.
Applications
- Ultra-low detection limit analysis of Li, Be, Mg, and other low-ionization-potential elements in ultrapure water for semiconductor manufacturing
- Routine monitoring of U, Th, and rare earth elements (REEs) in seawater and groundwater per ISO 17294-2
- Quantification of Pt-group elements (PGEs) in geological digests with minimal polyatomic interference
- Isotopic ratio measurements (e.g., ²⁰⁶Pb/²⁰⁷Pb, ²³⁸U/²³⁵U) requiring high signal stability and low oxide formation rates
- High-throughput clinical screening of essential/toxic elements (e.g., Se, As, Cd, Pb) in serum and whole blood using collision/reaction cell ICP-MS
FAQ
What distinguishes the C-Flow from conventional quartz nebulizers?
Quartz nebulizers are susceptible to etching by HF and alkalis, leading to gradual bore enlargement and signal drift. The C-Flow’s PFA body remains dimensionally stable across pH 0–14 and resists thermal shock—ensuring consistent nebulization efficiency over >500 hours of continuous operation.
Can the C-Flow be used with microflow or desolvation sample introduction systems?
Yes—its 50 µL/min variant is validated for use with Aridus III, Apex-Q, and similar desolvating nebulizers when paired with appropriate PFA transfer lines and chilled spray chambers.
Is cleaning required between sample batches?
No routine cleaning is necessary due to PFA’s non-stick surface and absence of crevices. A 2% HNO₃ rinse for 60 seconds suffices for carryover mitigation in most regulatory workflows.
Does Savillex provide certification documentation for GMP audits?
Each C-Flow unit ships with a Certificate of Conformance referencing ISO 9001:2015, material traceability (lot-specific PFA resin batch ID), and functional test data (nebulization efficiency vs. ¹¹⁵In at 100 µL/min).