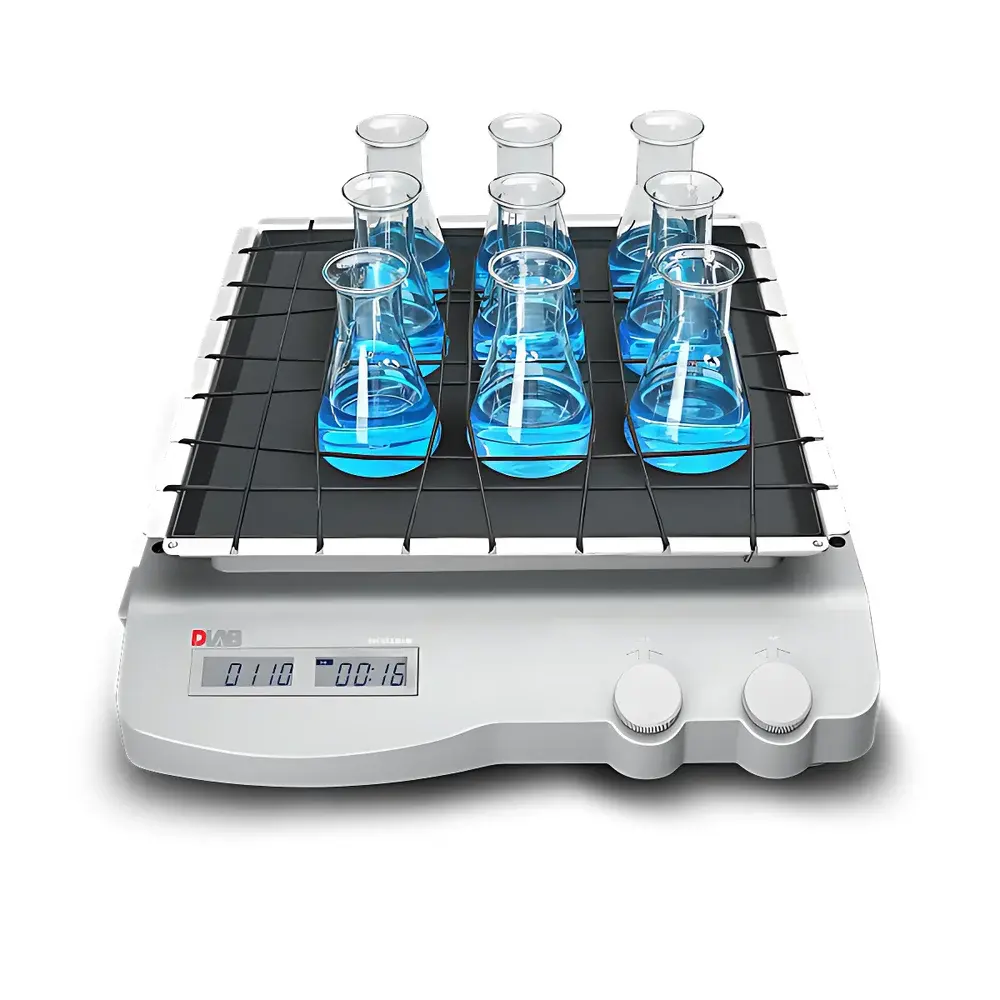
DLAB SK-O330-M Orbital Decolorizing Shaker
| Brand | DLAB |
|---|---|
| Origin | Beijing, China |
| Manufacturer Type | Direct Manufacturer |
| Instrument Category | Orbital Shaker |
| Model | SK-O330-M |
| Oscillation Mode | Orbital |
| Amplitude | 10 mm |
| Speed Range | 70–400 rpm |
| Speed Display | Dual LCD (Time & Speed) |
| Timer Range | 1 min – 99 h 59 min |
| Operating Modes | Timed / Continuous |
| Motor Type | Brushless DC Motor |
| Max Load Capacity (incl. fixtures) | 3 kg |
| Input Voltage | 100–240 V AC, 50/60 Hz |
| Power Consumption | 25 W (Input) / 15 W (Output) |
| Dimensions (L×W×H) | 370 × 420 × 100 mm |
| Tray Size | 335 × 335 mm |
| Net Weight | 9.8 kg |
| Ambient Operating Temperature | 5–40 °C |
| Relative Humidity Limit | ≤80% RH |
| Enclosure Rating | IP21 |
| Safety Features | Overspeed Protection |
| Optional Accessory | Transparent Acrylic Cover with Foam-Insulated Cavity |
Overview
The DLAB SK-O330-M Orbital Decolorizing Shaker is an engineered solution for consistent, low-shear orbital agitation in clinical diagnostics and life science laboratories. Designed specifically to support standardized serological assays—including RPR (Rapid Plasma Reagin) and TRUST (Toluidine Red Unheated Serum Test) for syphilis screening—the instrument employs precise circular motion with a fixed 10 mm amplitude to ensure uniform mixing of antigen-antibody reactions without disrupting flocculent precipitates or particulate aggregates. Its compact footprint (370 × 420 × 100 mm) and lightweight construction (9.8 kg) enable seamless integration into biosafety cabinets, laminar flow hoods, or high-density benchtop workflows. The dual-LCD interface independently displays real-time rotational speed and elapsed or remaining time—critical for protocol adherence in CLIA- and CAP-accredited environments where traceable parameter logging supports GLP/GMP compliance.
Key Features
- Brushless DC motor delivering maintenance-free operation, high torque stability across the full 70–400 rpm range, and extended service life under continuous duty cycles
- Dual independent LCD panels: one dedicated to speed (rpm), the other to timer status—enabling unambiguous verification prior to assay initiation
- Programmable timing from 1 minute to 99 hours 59 minutes, with seamless toggling between timed and continuous modes via front-panel controls
- Overspeed protection circuitry automatically halts rotation if rotational velocity exceeds user-defined or factory-set thresholds—preventing sample splashing or tray dislodgement
- IP21-rated enclosure provides basic protection against vertically falling drips and solid objects ≥12.5 mm, suitable for standard laboratory ambient conditions (5–40 °C, ≤80% RH)
- Standard 335 × 335 mm platform accepts universal clamps, microplate holders, and custom fixtures; optional transparent acrylic cover includes integrated foam-lined cavity for thermal buffering during ambient-temperature incubation
Sample Compatibility & Compliance
The SK-O330-M is validated for use with standard diagnostic formats including glass test tubes (12–16 mm diameter), plastic reaction vials, 96-well microplates, and slide-based agglutination carriers. Its orbital motion profile meets the mechanical agitation requirements specified in CLSI EP12-A2 for qualitative immunoassay reproducibility and ASTM D4084-20 Annex A1 for low-frequency mixing consistency. While not classified as a medical device per FDA 21 CFR Part 820, the shaker supports IVD workflow integrity by enabling strict adherence to manufacturer-recommended incubation protocols for RPR/TRUST reagents (e.g., Bio-Rad, Omega Diagnostics). Its brushless motor design eliminates carbon brush wear debris—reducing contamination risk in sterile or low-bioburden environments.
Software & Data Management
This stand-alone shaker operates without embedded firmware or external software dependencies. All operational parameters are set and monitored locally via tactile membrane buttons and dual LCD readouts. No data export interface (e.g., USB, RS-232, Ethernet) is provided; however, the absence of digital connectivity aligns with ISO/IEC 17025:2017 Clause 7.7.1.2 requirements for instruments used in accredited testing where manual recordkeeping is permitted and auditable. For laboratories implementing electronic lab notebooks (ELN) or LIMS integration, time-stamped operator logs—including start/stop times, speed settings, and duration—can be manually entered with traceability to analyst ID and assay batch number.
Applications
- Syphilis serology: Controlled agitation for RPR and TRUST card tests per CDC-recommended protocols
- Antibody titration workflows requiring gentle, reproducible mixing of serum dilutions
- Cell suspension homogenization prior to centrifugation in hematological or microbiological screening
- Staining and destaining of electrophoretic gels where uniform lateral diffusion is critical
- Low-temperature hybridization support when used with insulated covers in ambient-controlled rooms
FAQ
Is the SK-O330-M compliant with FDA 21 CFR Part 11?
No—this is a Class I benchtop shaker without electronic record storage, audit trail generation, or user authentication capabilities. It supports Part 11 compliance only through manual documentation practices.
Can it accommodate deep-well plates or conical tubes?
Yes—when used with compatible universal clamps or custom-adapted fixtures; maximum load remains 3 kg inclusive of all attachments.
What maintenance is required for the brushless DC motor?
None beyond periodic cleaning of the tray surface and ventilation grilles; no lubrication, brush replacement, or calibration is necessary over its operational lifetime.
Does the transparent cover affect rotational balance or speed accuracy?
No—the cover mounts externally and does not interface with the drive mechanism; speed stability is maintained within ±1 rpm across the full operating range.
Is the unit suitable for use inside a CO₂ incubator?
No—its IP21 rating and 5–40 °C ambient specification preclude operation in humidified, elevated-temperature enclosures. Use only in dry, room-temperature laboratory spaces.