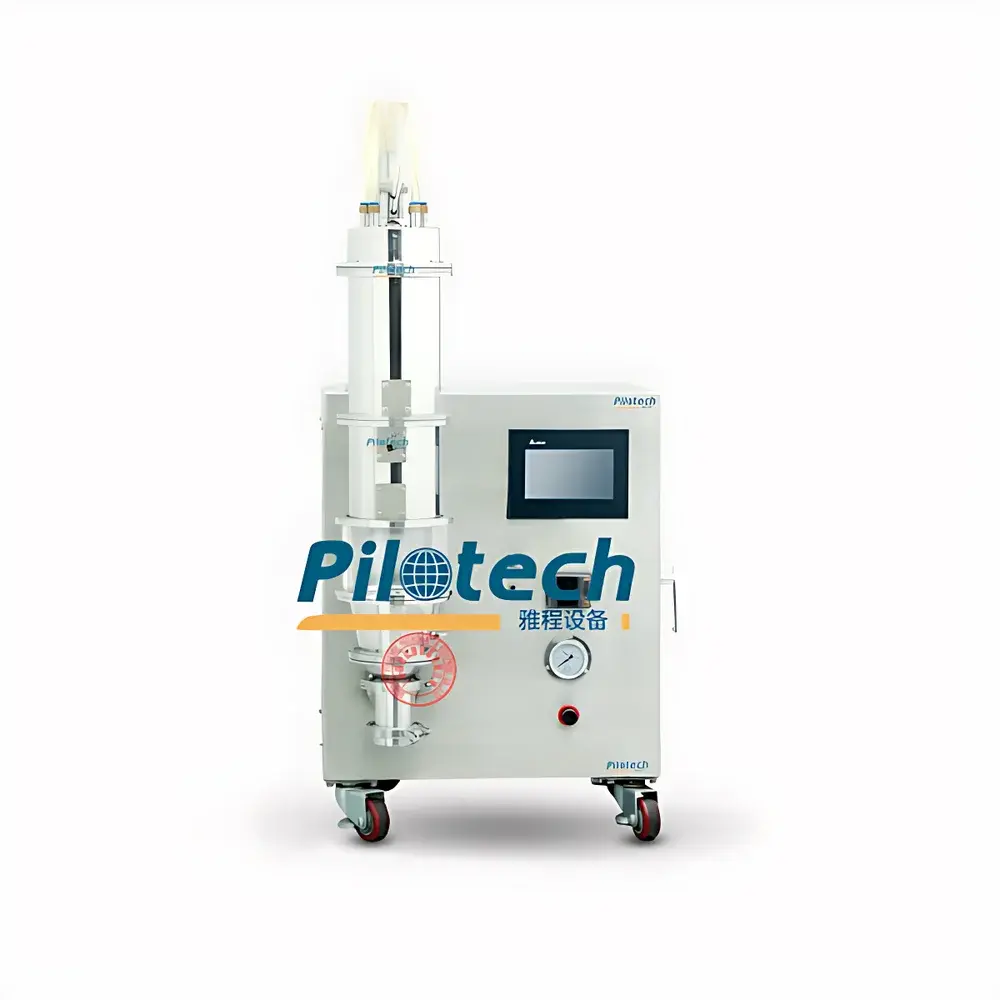
雅程 YC-310 Laboratory-Scale Fluidized Bed Granulator, Coater & Dryer
| Brand | YaCheng / PILOTECH |
|---|---|
| Origin | Shanghai, China |
| Manufacturer Type | OEM Manufacturer |
| Model | YC-310 |
| Pricing | Upon Request |
Overview
The YaCheng YC-310 is a compact, benchtop fluidized bed processor engineered for integrated granulation, coating, and drying of pharmaceutical, nutraceutical, and fine chemical powders in R&D and pilot-scale environments. It operates on the principle of fluidization—where controlled upward airflow suspends solid particles in a dynamic, turbulent state—enabling uniform heat and mass transfer during spray-based unit operations. The system supports three fundamental process configurations: top-spray (for agglomeration granulation and film coating), bottom-spray (for functional enteric or sustained-release coating of pellets), and fluid-bed drying (for gentle, thermodynamically balanced moisture removal). Unlike tray or vacuum drying, fluidized bed processing achieves rapid, homogeneous drying by maximizing interfacial contact between heated air and particle surfaces—reducing typical drying time by up to 20× compared to conventional static methods while preserving thermal sensitivity and particle integrity.
Key Features
- Modular fluidized bed architecture with interchangeable conical product bowls (standard capacity: 0.5–3 L, scalable via optional bowl kits) to accommodate variable batch sizes from 50 g to 500 g of dry powder.
- Dual airflow design: Top-spray configuration features a conical bowl with expansion chamber and multi-port nozzle mounting; bottom-spray mode utilizes an internal draft tube with concentric dual-inlet air distribution for precise core–shell coating of microspheres and mini-tablets.
- PLC-based control system with 7-inch color touchscreen HMI, supporting real-time adjustment of critical parameters—including inlet air temperature (RT–120 °C), blower frequency (0–50 Hz), peristaltic pump speed (0.1–100 rpm), product temperature feedback loop, and programmable pulse reverse-jet cleaning cycles.
- Integrated PID temperature regulation across all thermal zones (inlet heater, product bowl wall, exhaust duct) ensures ±0.5 °C stability over full operating range—critical for thermolabile APIs and polymer film formation.
- Imported components throughout: EC centrifugal blower (Germany), tubular stainless-steel heating elements (USA), industrial-grade PLC controller (Japan), and IP65-rated touch interface for lab environment durability.
- USB data export capability (CSV format) and optional RS485/Modbus RTU interface for integration into centralized lab data management systems compliant with 21 CFR Part 11 audit trail requirements.
Sample Compatibility & Compliance
The YC-310 accommodates a broad spectrum of particulate materials including herbal extracts, amorphous API powders, lactose-based excipients, MCC, HPMC, Eudragit® polymers, and silica-based carriers. It supports both aqueous and organic solvent-based binder solutions (e.g., PVP, HPMC, ethylcellulose) and is validated for use in GLP-compliant formulation development. Process parameters adhere to key industry references: ASTM D5656 (fluidized bed drying kinetics), USP (granulation), and ISO 14644-1 Class 8 cleanroom compatibility when operated within laminar flow hoods. All wetted parts are electropolished 316L stainless steel (Ra ≤ 0.4 µm), meeting ASME BPE-2022 surface finish standards for pharmaceutical equipment.
Software & Data Management
The embedded control firmware records timestamped operational logs—including inlet/outlet temperature differentials, pressure drop across the bed, blower power draw, and pump delivery rate—at user-defined intervals (1–60 seconds). Data files include metadata such as operator ID, batch number, and method version—supporting traceability under GMP Annex 11 and FDA 21 CFR Part 11. Optional software upgrade enables remote monitoring via Ethernet/Wi-Fi, historical trend analysis, and automated report generation (PDF/Excel) with digital signature fields. Audit trails are immutable and stored locally on encrypted SD card with automatic backup to network drive.
Applications
- Development of immediate-release granules from herbal decoctions and botanical extracts.
- Functional coating of 0.3–2.0 mm pellets with pH-dependent (Eudragit L100/S100) or time-controlled (ethylcellulose) polymer films.
- Masking bitter taste of APIs via aqueous hydroxypropyl methylcellulose (HPMC) film coating.
- Production of free-flowing, high-density granules for direct compression tablet manufacturing.
- Drying of spray-congealed waxes, lipid nanoparticles, and moisture-sensitive enzyme powders under low-temperature fluidized conditions.
- Process optimization studies for scale-up to production-scale fluid bed processors (e.g., Glatt GPCG, Freund GRANUPAX).
FAQ
What is the minimum batch size supported by the YC-310?
The system achieves stable fluidization down to 50 g of starting powder using the smallest standard bowl (0.5 L volume), with optimized airflow profiling and reduced nozzle orifice size.
Can the YC-310 perform solvent-based coating safely?
Yes—when equipped with explosion-proof motor options, nitrogen inerting port, and solvent recovery condenser (optional accessory), it complies with ATEX Zone 21 and OSHA 1910.107 requirements for Class IB solvent handling.
Is GMP documentation available for qualification (IQ/OQ/PQ)?
Full validation protocol templates—including sensor calibration certificates, airflow mapping reports, and temperature uniformity studies—are provided upon order confirmation for regulated lab deployment.
How does the reverse-jet cleaning system improve granule yield?
Programmable pulse-jet bursts (0.5–3 bar, 0.1–5 s duration) dislodge fines adhering to filter cartridges without disrupting bed dynamics—increasing overall granule recovery by 8–12% versus continuous filtration.
Does the system support continuous processing mode?
While designed primarily for batch operation, the YC-310’s modular airflow and feed control architecture allows semi-continuous operation via synchronized powder feeder and peristaltic liquid dosing—validated for proof-of-concept continuous granulation studies per ICH Q5C guidelines.