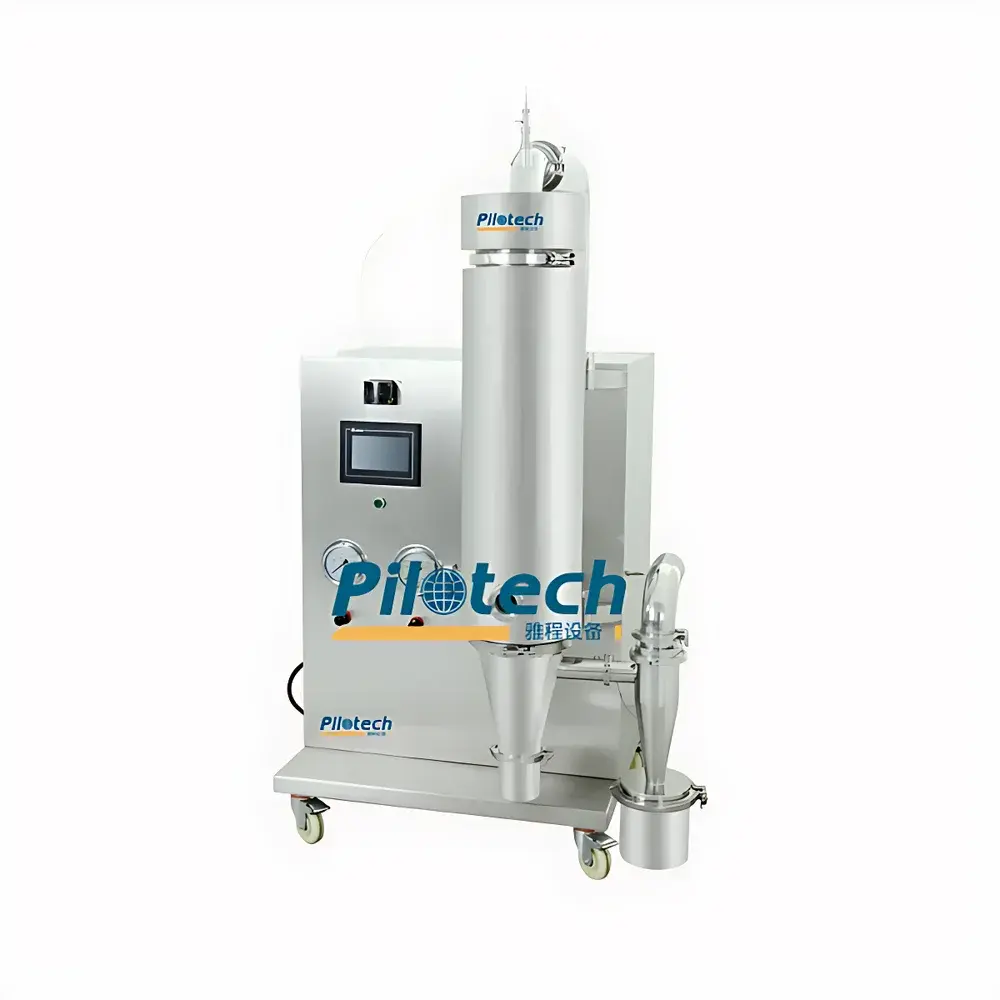
YaCheng YC-018 Benchtop Spray Dryer
| Brand | YaCheng / PILOTECH |
|---|---|
| Origin | Shanghai, China |
| Manufacturer Type | Direct Manufacturer |
| Model | YC-018 |
| Sample Type | Aqueous solutions only |
| Max Feed Rate | 3000 mL/h |
| Inlet Air Temperature Range | 105–280 °C |
| Particle Size Output | 1–80 µm |
Overview
The YaCheng YC-018 Benchtop Spray Dryer is an engineered solution for laboratory-scale spray drying and microencapsulation of heat-sensitive aqueous feedstocks. Based on the principle of single-stage concurrent-flow spray drying, it atomizes liquid feed into fine droplets via a pneumatic nozzle, exposing them to controlled hot air for rapid solvent evaporation within 1.0–1.5 seconds. This short residence time preserves thermolabile compounds—including proteins, enzymes, probiotics, botanical extracts, and pharmaceutical actives—while enabling reproducible particle formation across a broad size spectrum (1–80 µm). Designed for seamless transition from feasibility studies to pilot-scale process development, the YC-018 bridges the gap between traditional benchtop dryers (typically limited to ≤25 µm output) and industrial units, supporting both formulation screening and early-phase GMP-relevant material generation.
Key Features
- Optimized thermal architecture with dual-range inlet air temperature control (105–280 °C), enabling gentle drying of sugar-rich herbal extracts at low setpoints and high-temperature ceramic precursor drying at upper limits.
- High-efficiency cyclonic recovery system achieving up to 92% solid yield—significantly exceeding standard single-cyclone configurations—minimizing sample loss during R&D workflows.
- Integrated cleanable nozzle (1.5 mm standard; optional 0.5/0.8/1.0/2.0 mm) with programmable needle-clearing function to prevent clogging during viscous or particulate-laden feed processing.
- Full-color LCD touchscreen interface with real-time dynamic visualization of critical parameters: inlet/outlet temperature, blower frequency, peristaltic pump speed, and nozzle clearance cycle timing.
- PID-controlled heating system with ±1 °C thermal stability, ensuring precise thermal management essential for method transfer and regulatory documentation.
- Compact footprint (<1 m² floor space) with ultra-low noise operation (<55 dB(A)) due to integrated oil-free air compressor and acoustic-dampened enclosure—compliant with ISO 14040 and national laboratory ambient noise standards.
- Dual-mode operation: fully automated sequence control for repeatable batch execution, plus manual override for exploratory parameter tuning.
Sample Compatibility & Compliance
The YC-018 accepts aqueous-based feeds only—including solutions, colloidal suspensions, and emulsions—with solids content ranging from 1% to 30% w/w. It has been validated for use with biologics (e.g., lysozyme, insulin analogs), natural product extracts (e.g., ginsenosides, curcuminoids), food-grade polymers (e.g., maltodextrin, whey protein isolate), and inorganic precursors (e.g., alumina sols, zirconia nanoparticles). All wet-contact surfaces—including drying chamber, cyclone, collection vessel, and tubing—are constructed from electropolished 304 stainless steel (ASTM A276), ensuring corrosion resistance and ease of cleaning. The system supports GLP-compliant documentation through timestamped parameter logging and user-accessible data export (CSV format), and its closed-loop design aligns with ISO 22000 and USP <1117> guidance for non-sterile pharmaceutical drying processes.
Software & Data Management
The embedded control firmware records all operational parameters—including temperature ramps, pump flow profiles, and nozzle activation events—with millisecond-level time stamps. Data are stored locally on internal flash memory and exportable via USB port for integration into LIMS or electronic lab notebook (ELN) platforms. While not FDA 21 CFR Part 11-certified out-of-the-box, the system’s audit trail functionality (user login tracking, parameter change logs, session start/stop timestamps) provides foundational traceability required for GxP-aligned method development. Optional RS-485 or Ethernet communication modules enable remote monitoring and SCADA integration in centralized lab infrastructure.
Applications
- Pharmaceutical: Production of inhalable dry powders, sustained-release microparticles, and excipient-coated APIs under controlled thermal history.
- Food & Nutrition: Encapsulation of probiotics, omega-3 oils, and vitamins; production of instant beverage powders with improved solubility and shelf stability.
- Materials Science: Synthesis of catalyst supports (e.g., Pt/CeO₂ aggregates in 60–80 µm range), battery cathode precursors, and functional ceramic granules.
- Natural Products: Stabilization of polyphenols, flavonoids, and polysaccharides without Maillard degradation—validated by HPLC and DSC post-drying analysis.
- Academic Research: Teaching unit for mass/heat transfer fundamentals; platform for DOE-driven optimization of drying kinetics and morphology control.
FAQ
What types of solvents can be processed in the YC-018?
Only aqueous solutions are supported. Organic solvents, including ethanol, acetone, or chloroform, are strictly prohibited due to explosion risk and material compatibility constraints.
Is the system suitable for Good Manufacturing Practice (GMP) environments?
It is designed for R&D and preclinical manufacturing. While compliant with ISO 9001 and basic GMP hygiene principles (e.g., cleanable surfaces, traceable operation), formal qualification (IQ/OQ/PQ) must be performed per site-specific protocols.
Can particle size distribution be controlled beyond the stated 1–80 µm range?
Yes—size modulation is achieved via coordinated adjustment of feed concentration, inlet temperature, atomization pressure, and pump rate. Narrower distributions (±15% span) are attainable using optimized nozzle selection and feed rheology control.
Does the system include validation documentation?
Factory calibration certificates for temperature sensors and flow meters are provided. Full validation packages (including installation, operational, and performance qualification templates) are available upon request for regulated users.
What maintenance is required for long-term reliability?
Daily: Visual inspection of nozzle tip and cyclone seals; weekly: lubrication-free compressor filter replacement; quarterly: PID controller recalibration and blower bearing assessment per manufacturer service schedule.