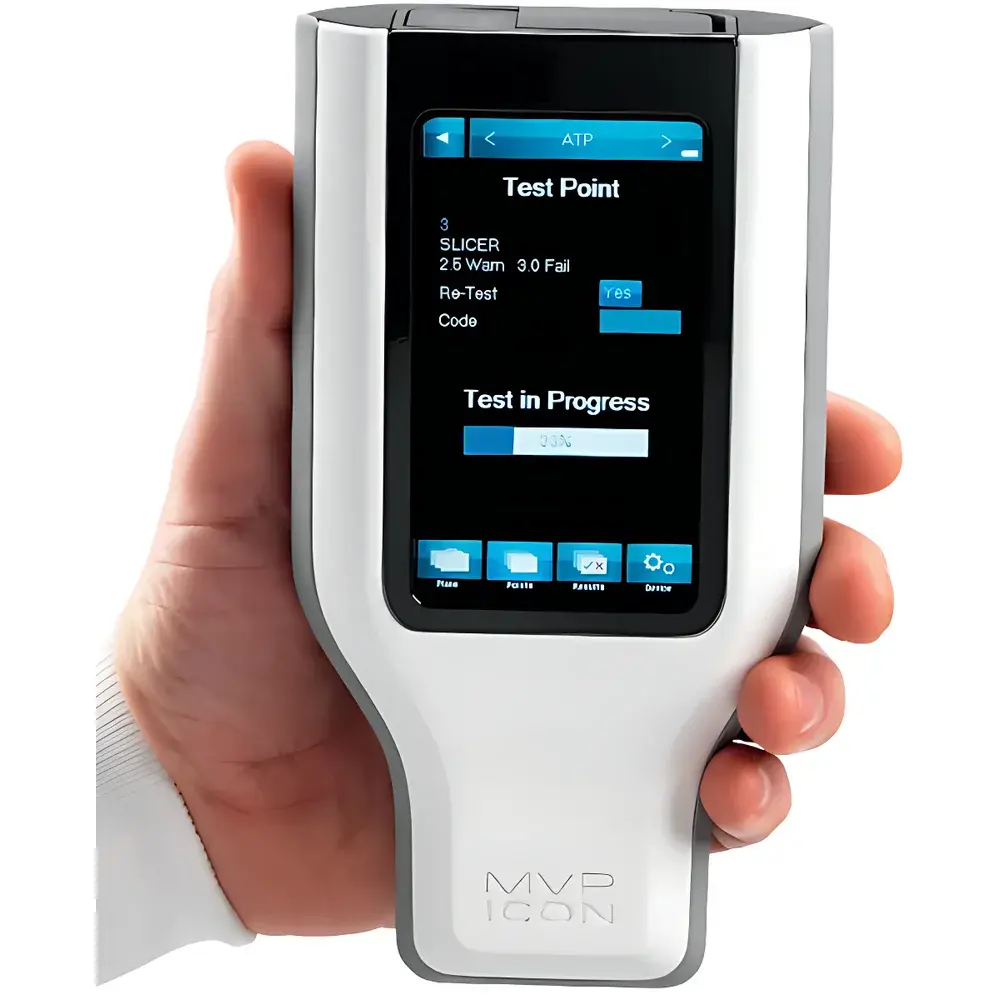
Millipore® MVP ICON™ Environmental Monitoring System
| Brand | Millipore |
|---|---|
| Origin | USA |
| Manufacturer Type | Authorized Distributor |
| Origin Category | Imported |
| Model | MVP ICON™ |
| Pricing | Available Upon Request |
Overview
The Millipore® MVP ICON™ Environmental Monitoring System is a purpose-built, integrated platform engineered for real-time, multi-parameter verification of critical control points in hygienic processing environments. Designed explicitly for HACCP (Hazard Analysis and Critical Control Points) and food/pharmaceutical facility validation workflows, the system employs modular sensor interfaces to concurrently monitor disinfectant concentration (via conductivity), pH, temperature, and bioluminescent ATP (adenosine triphosphate) levels — all recognized as key indicators of microbial load and cleaning efficacy. Unlike standalone meters or manual logbooks, the MVP ICON™ implements a closed-loop monitoring architecture: sensors feed time-stamped, calibrated data directly into an embedded industrial-grade controller, which applies preconfigured logic rules to trigger alerts, initiate corrective actions, and generate audit-ready records. Its measurement principles align with established methodologies — conductivity-based concentration estimation per ASTM D1125 and ISO 7888; pH per ISO 3696 and USP ; ATP quantification using luciferin-luciferase bioluminescence calibrated against ISO 22000 Annex B and AOAC Official Method 2014.01.
Key Features
- Modular, hot-swappable sensor architecture supporting up to six simultaneous input channels — including dual conductivity/pH/temperature combo probes and dedicated ATP luminometers.
- Embedded microprocessor with real-time data acquisition at configurable intervals (1 sec to 24 hr), ensuring temporal resolution appropriate for both rapid sanitization cycle verification and long-term trend analysis.
- On-device graphical interface with color-coded status indicators, intuitive navigation, and contextual help — operable without external PCs or tablets.
- Automated pass/fail evaluation against user-defined limits for each parameter, with configurable alarm escalation (visual, audible, relay output, and optional email/SMS via Ethernet/Wi-Fi gateway).
- Ruggedized IP65-rated enclosure rated for washdown environments, constructed from 316 stainless steel and chemical-resistant polymers compliant with FDA 21 CFR Part 117 and EU GMP Annex 1 requirements.
- Integrated non-volatile memory storing ≥12 months of raw data (timestamped, sensor ID-tagged, and checksum-verified) with automatic rollover and tamper-evident logging.
Sample Compatibility & Compliance
The MVP ICON™ accommodates standard industry sampling protocols across liquid-phase hygiene monitoring applications: CIP rinse water, sanitizer tank solutions (e.g., peracetic acid, sodium hypochlorite, quaternary ammonium), clean-in-place (CIP) return lines, and environmental swab eluates for ATP testing. Sensor modules are validated for use with aqueous matrices in pH 2–12, conductivity 0–200 mS/cm, temperature −10 °C to +85 °C, and ATP concentrations spanning 0.01–100 RLU (Relative Light Units). The system supports compliance with multiple regulatory frameworks: HACCP Plan documentation per Codex Alimentarius CAC/RCP 1-1969; environmental monitoring programs under FDA 21 CFR Part 111 (Dietary Supplements) and Part 211 (Pharmaceuticals); and data integrity requirements aligned with ALCOA+ principles (Attributable, Legible, Contemporaneous, Original, Accurate, Complete, Consistent, Enduring, Available).
Software & Data Management
Data export and review are facilitated through the MVP ICON™’s native USB port (for encrypted CSV/Excel exports) and optional Ethernet connectivity for integration into facility SCADA or LIMS platforms. The embedded firmware includes full audit trail functionality: every configuration change, calibration event, user login/logout, and alarm activation is recorded with operator ID, timestamp, and pre-/post-value context. All stored data are digitally signed and cryptographically hashed to ensure immutability. For regulated environments, the system supports 21 CFR Part 11-compliant electronic signatures when paired with optional identity verification hardware (e.g., RFID badge reader or biometric module). Historical reports — including shift summaries, deviation logs, and trend charts — can be generated on-device or via the optional MVP ICON™ Data Manager desktop application (Windows 10/11, TLS 1.2 secured).
Applications
- Real-time verification of sanitizing solution concentration during CIP cycles in beverage, dairy, and pharmaceutical manufacturing lines.
- Continuous monitoring of pH and conductivity in purified water distribution loops to detect membrane fouling or resin exhaustion.
- ATP-based surface hygiene verification following equipment disassembly and manual cleaning — integrated with digital SOP execution prompts.
- Environmental monitoring program (EMP) support in Grade A/B cleanrooms per ISO 14644-1 and EU GMP Annex 1, correlating ATP spikes with particle counts and microbiological air sampling.
- Validation and requalification of cleaning processes per ICH Q5A(R2) and PDA Technical Report No. 29.
FAQ
Does the MVP ICON™ require annual calibration certification?
Yes — while factory-calibrated sensors include NIST-traceable certificates, routine user verification per ASTM E2911 is recommended prior to each shift or batch. Calibration logs are automatically archived with metadata.
Can the system integrate with existing MES or ERP platforms?
Yes — via Modbus TCP or OPC UA protocol over Ethernet, enabling direct data ingestion into Siemens Opcenter, Rockwell FactoryTalk, or SAP Manufacturing Integration and Intelligence (MII).
Is ATP measurement performed in situ or after sample transfer?
ATP assays require sample collection and immediate analysis in the field-deployable luminometer module; no incubation or lab-based processing is needed.
What cybersecurity standards does the MVP ICON™ meet?
Firmware complies with IEC 62443-4-2 SL2 requirements, including secure boot, encrypted firmware updates, role-based access control, and TLS 1.2 for remote communications.
How is data backup and disaster recovery handled?
Local storage is mirrored to removable encrypted USB drives; optional cloud sync (AWS IoT Core) provides geo-redundant backup with AES-256 encryption and retention policies configurable per GDPR or 21 CFR Part 11.