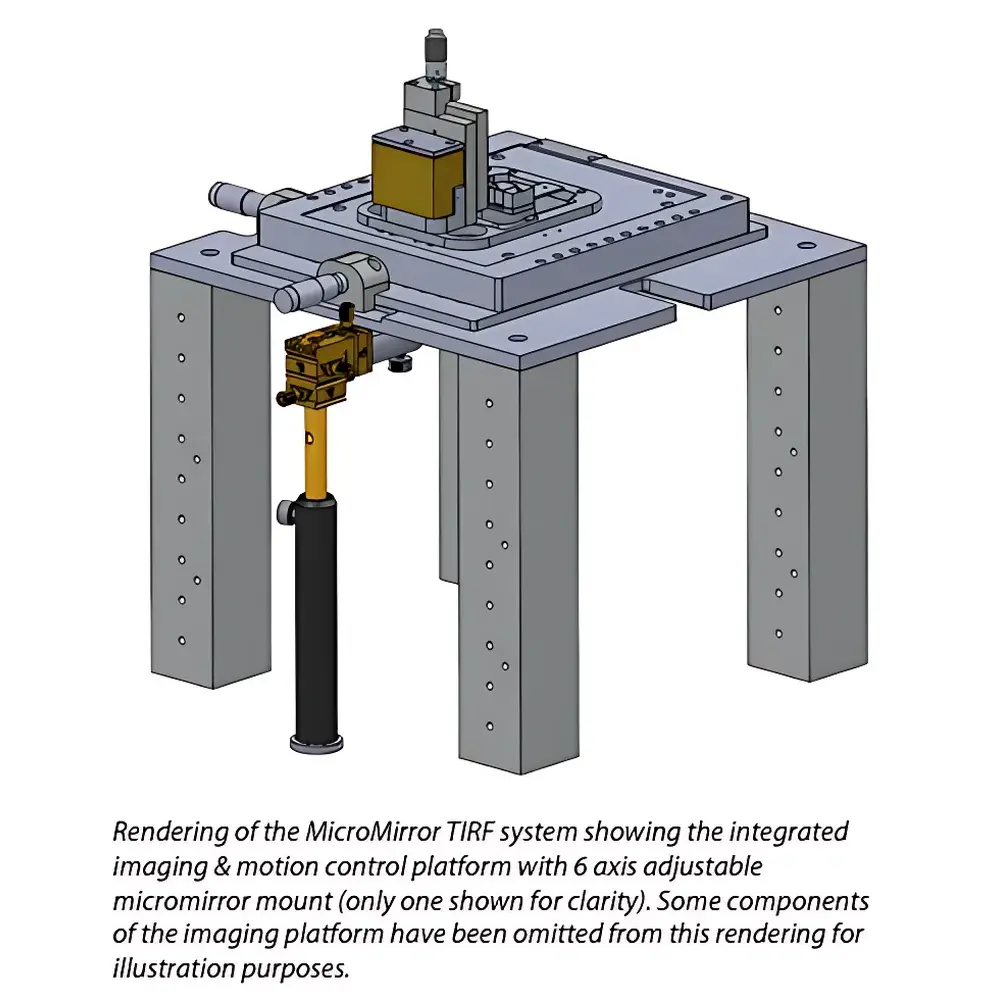
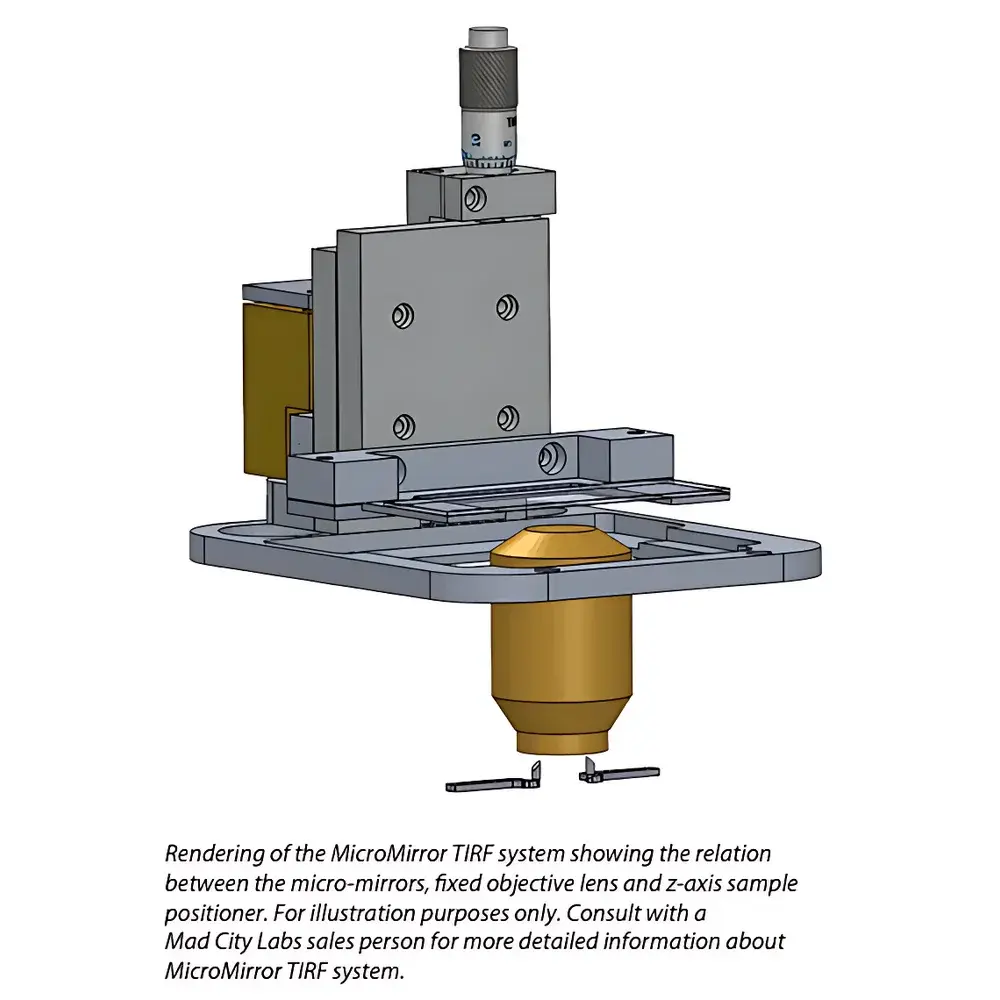
Mad City Labs MicroMirror TIRF System for Single-Molecule Imaging
| Brand | Mad City Labs (MCL Think Nano) |
|---|---|
| Origin | USA |
| Manufacturer Type | Authorized Distributor |
| Product Category | Imported Instrument |
| Model | MicroMirror TIRF System |
| Instrument Type | Inverted Fluorescence Microscope |
| Excitation Source | Metal Halide Lamp |
| Eyepiece Compatibility | Universal Eyepiece Mount |
| Objective Compatibility | Universal Objective Mount |
| Fluorescence Configuration | Integrated Through-Objective Total Internal Reflection Fluorescence (TIRF) |
| Illumination Flexibility | Multi-Source Compatible |
| Observation Head | Monocular, Full-Internal-Reflection Optical Path |
Overview
The Mad City Labs MicroMirror TIRF System is a purpose-built, inverted fluorescence microscope platform engineered for high-fidelity single-molecule imaging using total internal reflection fluorescence (TIRF) microscopy. Unlike conventional dichroic-based TIRF systems, this instrument implements a through-the-objective excitation architecture that replaces the spectral beam splitter with two broadband micro-mirrors positioned at the objective’s back focal plane. This optical redesign eliminates wavelength-dependent transmission losses and polarization artifacts inherent to dielectric dichroics, enabling simultaneous, stable illumination across multiple laser lines—critical for multi-color co-localization, FRET, and sequential activation studies of biomolecular complexes. The system operates on the physical principle of evanescent wave excitation: when collimated light undergoes total internal reflection at the glass–water interface beneath the specimen, an exponentially decaying evanescent field (typically < 150 nm penetration depth) selectively excites fluorophores proximal to the coverslip surface. This provides exceptional axial sectioning capability and signal-to-noise ratio (SNR), making it the method of choice for observing dynamic assembly, conformational transitions, and real-time kinetics of membrane-associated or surface-tethered macromolecular machines.
Key Features
- Micro-Mirror TIRF Optics: Dual 3 mm aluminum-mounted broadband micro-mirrors replace dichroic filters, supporting simultaneous multi-laser excitation (e.g., 405/488/561/640 nm) without alignment compromise or throughput loss.
- Open-Path Optical Architecture: Fully accessible excitation and emission pathways facilitate integration of custom lasers, AOTFs, acousto-optic deflectors, or additional detection channels—including EMCCD, sCMOS, or SPAD arrays.
- Integrated Nanopositioning Platform: XYZ closed-loop piezoelectric stage with proprietary PicoQ® capacitive sensors delivers sub-nanometer resolution (0.4 nm), 200 µm travel range, and thermal drift compensation—essential for long-duration drift-corrected time-lapse imaging.
- TIRF Lock™ Feedback System: Real-time stabilization of the evanescent field intensity via quadrant photodiode (QPD)-based sensing and software-controlled Z-axis correction, maintaining consistent excitation angle and penetration depth over hours.
- Modular Mechanical Design: Anodized aluminum frame ensures mechanical rigidity and thermal stability; integrated slide holder accommodates standard 25 × 75 mm or custom microfluidic chambers.
- Adjustable Micromirror Mounts: Five-axis precision kinematic mounts allow iterative fine-tuning of mirror tilt and lateral position—enabling optimal critical angle calibration for diverse refractive index interfaces (e.g., aqueous buffer, glycerol, oil).
Sample Compatibility & Compliance
The MicroMirror TIRF System supports a broad range of biological specimens, including live cells cultured on #1.5 coverslips, reconstituted lipid bilayers, DNA origami substrates, immobilized protein complexes, and microfluidically confined organelles. Its universal objective and eyepiece compatibility permits use with high-NA oil-, water-, or glycerol-immersion objectives (up to 100×, NA ≥ 1.49). The open optical layout allows direct integration with environmental control chambers (temperature, CO₂, humidity) and electrophysiology rigs. While not a regulated medical device, the system’s design adheres to principles aligned with GLP-compliant workflows: full audit trail logging in TIRF Lock™ and GLIMPSE software, timestamped metadata embedding per acquisition, and deterministic motion control traceability—supporting reproducibility requirements under ISO/IEC 17025 and NIH rigorous methodology guidelines.
Software & Data Management
Control and data acquisition are coordinated through three interoperable software modules: Nano-Drive® (nanopositioner hardware abstraction layer), Nano-Route®3D (multi-dimensional trajectory planning and synchronization), and GLIMPSE (dedicated TIRF imaging suite with real-time feedback, multi-channel registration, and drift correction). All software communicates via USB 2.0 and supports TTL synchronization outputs for external trigger coordination (e.g., laser pulsing, camera exposure gating). Data export conforms to OME-TIFF standards, ensuring compatibility with Fiji/ImageJ, Python (scikit-image, napari), and commercial analysis platforms such as Imaris or Huygens. Audit logs include full parameter histories, sensor readouts, and motion command timestamps—facilitating compliance with FDA 21 CFR Part 11 where electronic records are validated within institutional SOPs.
Applications
- Real-time visualization of kinesin/dynein motility on microtubule networks
- Single-particle tracking of GPCR oligomerization and trafficking at plasma membranes
- Stepwise assembly kinetics of nuclear pore complex subunits
- Conformational dynamics of CRISPR-Cas9 target search and cleavage
- Multi-color colocalization of synaptic vesicle proteins during exocytosis
- Quantitative binding affinity measurements via surface-immobilized titration assays
FAQ
What distinguishes MicroMirror TIRF from conventional dichroic TIRF systems?
It eliminates the dichroic mirror entirely, replacing it with broadband micro-mirrors—thereby removing wavelength-specific insertion loss, polarization sensitivity, and alignment fragility associated with multi-laser experiments.
Can I integrate pulsed lasers or supercontinuum sources?
Yes—the open excitation path and modular mirror mounts support fiber-coupled or free-space coupling of ultrafast or tunable sources, provided beam diameter and divergence fall within the objective back-aperture constraints.
Is the system compatible with STORM/PALM super-resolution modalities?
Absolutely—the high SNR, precise Z-stabilization, and multi-color flexibility make it an ideal platform for single-molecule localization microscopy requiring >10,000 frames and sub-20 nm localization precision.
What level of technical support is provided for optical alignment?
Mad City Labs supplies comprehensive alignment protocols, including QPD-based critical angle optimization routines and video-guided mirror positioning workflows—supported by application engineers trained in biophysical microscopy.
Does the system meet vibration isolation requirements for single-molecule work?
The anodized aluminum chassis exhibits low resonant frequency and high damping; optimal performance is achieved when mounted on active or passive optical tables meeting ISO 23820 Class A specifications (vibration transmissibility < 0.1% at 10 Hz).