SUNS SUNS Negative-Pressure Air-Tightness Tester
| Brand | SUNS |
|---|---|
| Origin | Shenzhen, China |
| Manufacturer Type | Direct Manufacturer |
| Country of Origin | China |
| Model | Negative-Pressure Air-Tightness Tester |
| Quotation | Available Upon Request |
| Test Pressure Range | –70 kPa |
Overview
The SUNS Negative-Pressure Air-Tightness Tester is an engineered solution for quantitative leakage detection and integrity verification of sealed medical devices and packaging systems under controlled sub-atmospheric conditions. Designed in accordance with the fundamental principles of differential pressure decay testing, this instrument applies a precisely regulated negative pressure (vacuum) to the test specimen—typically a sealed container, syringe, IV bag, catheter housing, or implantable device enclosure—and monitors pressure stabilization and decay rate over time. The system operates on the physical premise that any micro-leak will permit ambient air ingress, resulting in measurable pressure recovery toward atmospheric equilibrium. This method delivers high reproducibility for detecting leaks down to 1 × 10–5 mbar·L/s (equivalent to ~0.1 mL/min at –70 kPa), making it suitable for critical quality control in Class II and Class III medical device manufacturing where package integrity directly impacts sterility assurance.
Key Features
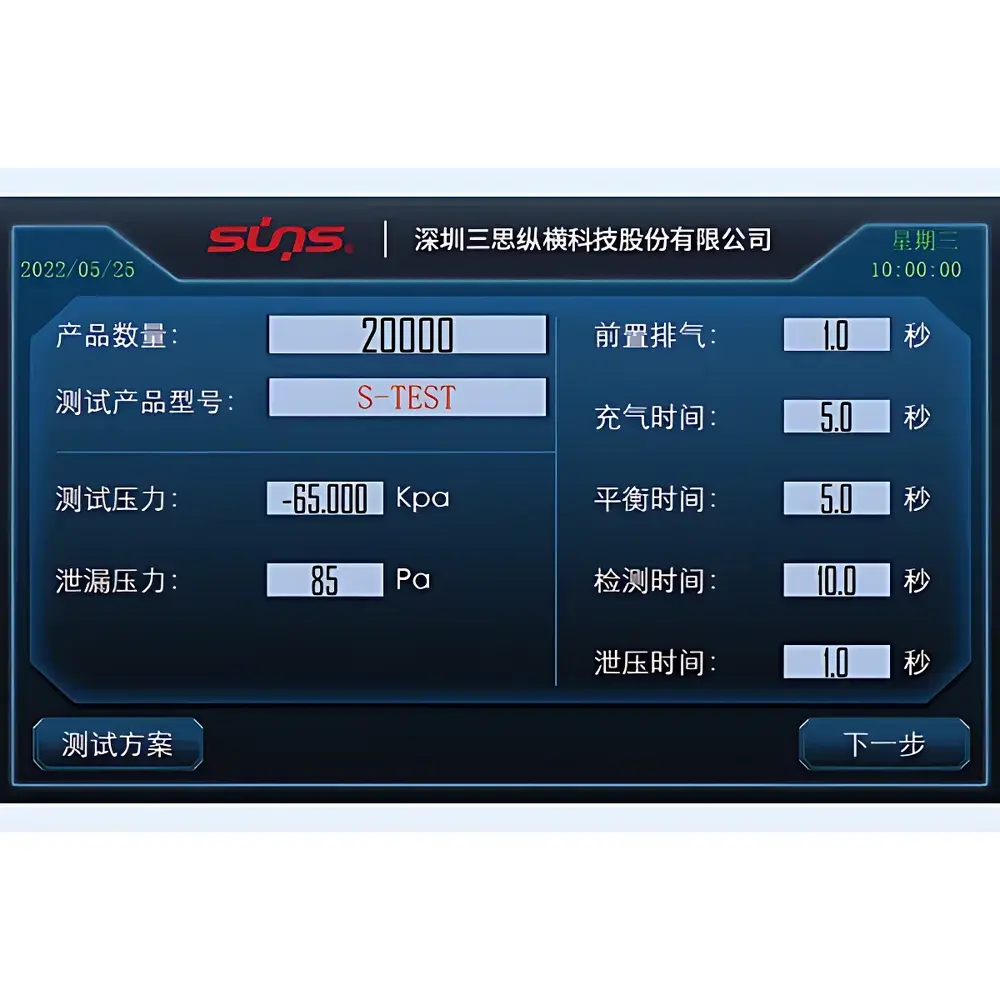
- Stable, digitally controlled vacuum generation capable of achieving and maintaining up to –70 kPa (absolute pressure ≈ 30 kPa) with ±0.3 kPa regulation accuracy
- Integrated high-resolution pressure transducer with temperature compensation and long-term zero drift stability (<0.05 kPa/month)
- Dual-chamber configuration option available for simultaneous reference-compensated testing, minimizing environmental influence on measurement fidelity
- Modular pneumatic architecture featuring stainless steel manifolds, fluoropolymer-sealed solenoid valves, and ISO 8573-1 Class 2 compressed air compatibility
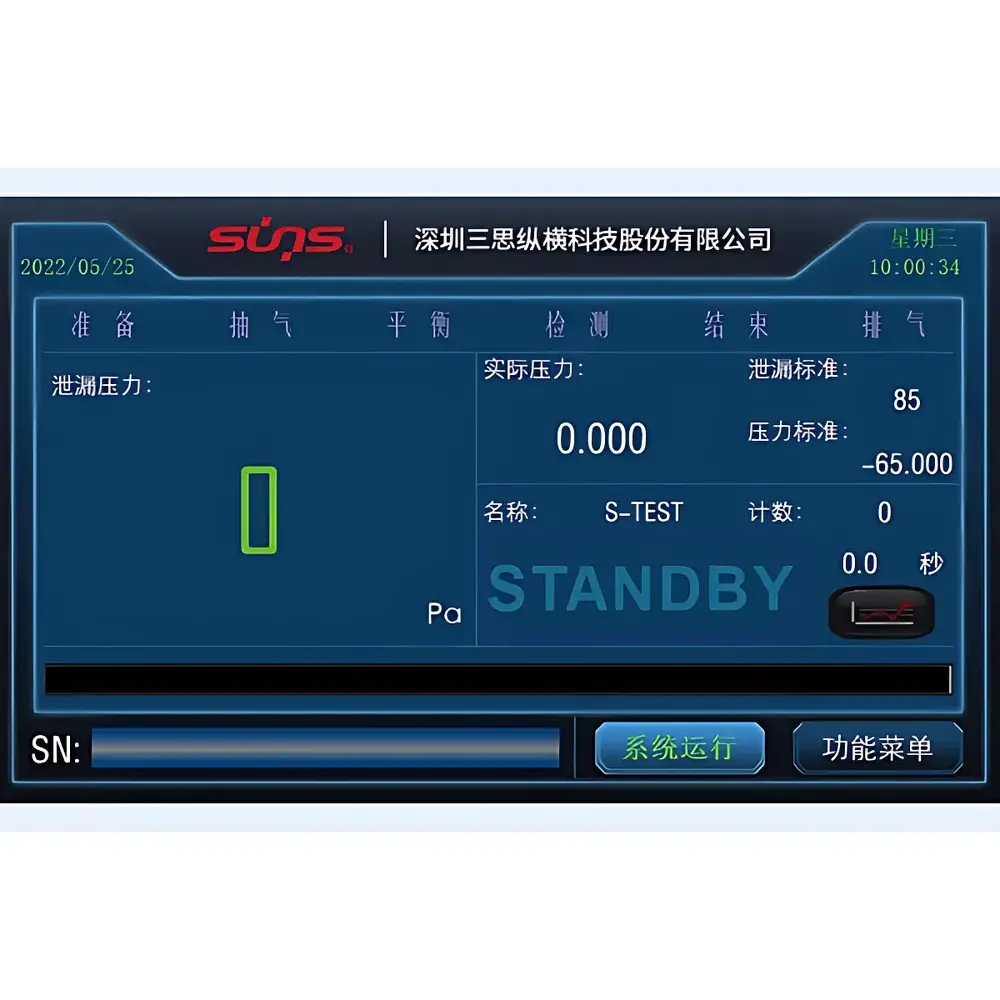
- Front-panel touchscreen interface with intuitive test sequence programming, real-time pressure curve visualization, and pass/fail threshold assignment per ASTM F2096 and ISO 11607-2
- Rugged industrial-grade enclosure rated IP54, designed for continuous operation in ISO 7 cleanroom-adjacent QC laboratories
Sample Compatibility & Compliance
The tester accommodates a broad range of medical device formats including pre-filled syringes, blister packs, pouches, vials with rubber stoppers, surgical instrument trays, and silicone-based implant housings. Fixture adaptability supports custom tooling for non-standard geometries via standardized NPT 1/4″ and G1/8″ ports. All test protocols align with internationally recognized standards for package integrity validation: ASTM F2096 (bubble emission test correlation), ISO 11607-2 (validation of forming, sealing, and assembly processes), and EU MDR Annex I General Safety and Performance Requirements (GSPR 10.2). Data output meets ALCOA+ criteria for audit readiness, supporting GLP and GMP environments requiring full traceability per FDA 21 CFR Part 11 when paired with optional software modules.
Software & Data Management
The embedded firmware enables local storage of ≥10,000 test records with timestamp, operator ID, batch number, test parameters, raw pressure vs. time data, and pass/fail status. Optional PC-based SUNS IntegritySuite™ software provides advanced statistical process control (SPC), automated report generation (PDF/CSV), multi-site data aggregation, and electronic signature workflows compliant with 21 CFR Part 11 Annex 11. Audit trails record all user actions—including parameter edits, result overrides, and calibration events—with immutable timestamps and user attribution. Exported datasets include metadata required for regulatory submissions to notified bodies and health authorities.
Applications
- Final packaging integrity verification for sterile barrier systems prior to sterilization cycle release
- In-process leak testing during thermoforming and heat-sealing operations for flexible packaging lines
- Design verification of closure systems for prefilled drug delivery devices (e.g., autoinjectors, pen injectors)
- Accelerated aging study support—monitoring seal degradation after accelerated shelf-life simulation
- Root cause analysis of field failures linked to packaging breach or material permeation
- Supplier qualification testing for contract manufacturers producing Class IIa–III medical devices
FAQ
What is the maximum achievable test pressure?
The system is calibrated and validated to deliver stable negative pressure up to –70 kPa (gauge), corresponding to approximately 30 kPa absolute pressure at sea level.
Can this instrument be used for positive-pressure leak testing?
No—the SUNS Negative-Pressure Air-Tightness Tester is specifically configured for vacuum-decay methodology. Positive-pressure applications require a separate pressurized decay or mass flow configuration.
Is calibration traceable to national standards?
Yes—factory calibration uses pressure standards certified to NIST-traceable references, with documentation provided per ISO/IEC 17025 requirements.
Does the system support integration with MES or LIMS platforms?
Yes—via TCP/IP or RS-232 serial protocol using Modbus RTU or custom ASCII command sets; API documentation available upon request.
What maintenance intervals are recommended?
Routine verification every 3 months; full recalibration annually or after any impact event affecting sensor integrity or pneumatic seals.