Harvard Apparatus PUMP33 DDS Dual-Channel Syringe Pump
| Brand | Harvard Apparatus |
|---|---|
| Origin | USA |
| Model | PUMP33 DDS |
| Instrument Type | Microfluidic Syringe Pump |
| Flow Rate Range | 1.02 pL/min to 106 mL/min |
| Accuracy | ±0.25% |
| Repeatability | ±0.05% |
| Display | 7″ Color Touchscreen LCD |
| Input Voltage | 100–240 VAC, 50/60 Hz |
| Dimensions (H×W×D) | 28 × 39 × 21 cm |
| Weight | 9.9 kg |
| Certifications | CE, ETL (UL & CSA), CB Scheme, RoHS, WEEE |
Overview
The Harvard Apparatus PUMP33 DDS is a microprocessor-controlled dual-channel syringe pump engineered for high-precision fluid delivery in research laboratories. It operates on the principle of linear motor-driven plunger displacement, enabling accurate volumetric dispensing and aspiration across an exceptionally broad flow range—from picoliter-per-minute ultra-low-flow applications (e.g., single-cell microinjection or electrophysiology) to milliliter-per-minute macro-scale perfusion tasks. Unlike single-channel systems, the PUMP33 DDS integrates two independently programmable pumping channels within a single chassis, each capable of autonomous operation or coordinated control—making it suitable for complex experimental paradigms requiring synchronized or counter-directional fluid handling. Its architecture adheres to IEC 61010-1 safety standards for laboratory equipment and supports GLP-compliant workflows through audit-ready parameter logging and deterministic execution.
Key Features
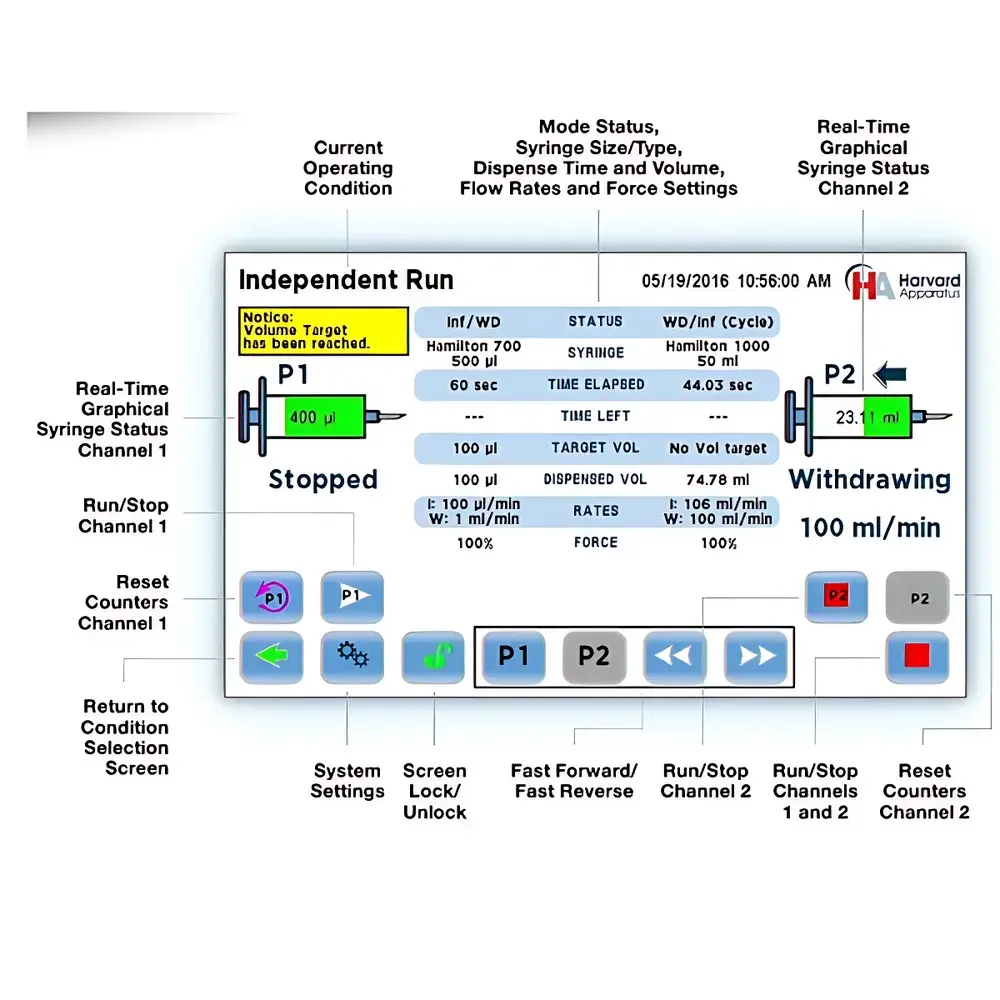
- Dual independent channels (labeled P1 and P2) with fully decoupled parameter sets—including syringe type, diameter, target volume/time, flow rate, and force limit.
- 7-inch color touchscreen interface with intuitive graphical navigation, real-time status visualization, and on-device program editing without external software.
- Universal syringe compatibility: accommodates glass, plastic, and stainless-steel syringes from 0.5 µL (ID ≥ 0.013 mm) to 60 mL (ID ≤ 32.573 mm), including non-standard and custom-bore formats upon verification.
- Three operational modes: Independent (two discrete pumps), Reciprocal (anti-phase bidirectional flow ideal for valve-based continuous infusion), and Synchronized (coordinated delivery with merged volumetric throughput).
- Programmable mechanical force up to 70 lbf (31.75 kgf) at full stroke speed; adjustable torque limits prevent syringe barrel deformation or plunger slippage during high-viscosity or high-backpressure applications.
- Comprehensive I/O infrastructure: dual 15-pin D-sub TTL I/O ports (one per channel), footswitch inputs, audio alarm outputs, and screen lock functionality for unattended operation in shared lab environments.
Sample Compatibility & Compliance
The PUMP33 DDS is validated for use with standard laboratory-grade syringes meeting ISO 7886-1 specifications. It does not support clinical or in vivo human use and is explicitly designated for non-diagnostic, non-therapeutic research applications only. All firmware and hardware design comply with EU Directive 2014/30/EU (EMC), 2014/35/EU (LVD), and RoHS 2011/65/EU. The device carries CE marking under Class I pollution degree and Installation Category II per IEC 60664-1. For regulated environments, optional timestamped event logs (including start/stop triggers, error codes, and parameter changes) can be exported via USB or RS-232 for traceability in GLP or preclinical study documentation.
Software & Data Management
The pump operates autonomously via its embedded controller but supports ASCII command protocol over RS-232 (9-pin D-sub, multiplexed dual bidirectional) and USB Type-B interfaces for integration into automated test benches or custom LabVIEW/Python control suites. RS-485 daisy-chaining enables multi-pump synchronization across distributed fluidic networks. Firmware updates are performed via USB mass storage mode. No proprietary runtime or license is required—command syntax follows Harvard Apparatus’ documented SCPI-like structure, ensuring long-term maintainability and interoperability with third-party data acquisition platforms. Audit trails record all user-initiated actions with timestamps, user ID fields (if configured externally), and checksum-verified parameter snapshots.
Applications
- Neuroscience: Controlled intracerebral or intraventricular microinfusion in rodent models, paired with electrophysiological recording systems.
- Microfluidics: Precise reagent metering in droplet generation, organ-on-chip perfusion, and gradient formation assays.
- Materials science: Sol-gel precursor delivery, nanoparticle suspension dosing, and controlled polymer extrusion in additive manufacturing R&D.
- Pharmacology: Multi-step compound dilution series generation, timed co-infusion of agonist/antagonist pairs, and PK/PD model validation infusions.
- Chemistry: Automated titration, catalyst injection in flow reactors, and slow-addition synthesis protocols requiring sub-microliter precision.
FAQ
Is the PUMP33 DDS suitable for Good Laboratory Practice (GLP) environments?
Yes—when used with appropriate procedural controls, its deterministic execution, parameter logging capability, and timestamped event history support GLP audit requirements. Full 21 CFR Part 11 compliance requires integration with validated third-party electronic lab notebook (ELN) or LIMS systems.
Can I use non-Harvard syringes with this pump?
Yes—the platform accepts any syringe meeting dimensional tolerances specified in the manual (0.5 µL to 60 mL capacity, ID 0.013–32.573 mm). Calibration is performed per syringe using built-in diameter input; no vendor-specific cartridges are required.
What is the difference between Reciprocal and Synchronized mode?
Reciprocal mode drives both channels in opposite directions at identical speeds—ideal for closed-loop circulation with external valves. Synchronized mode aligns all parameters (flow rate, volume, timing) to deliver combined throughput, effectively doubling maximum flow when using matched syringes.
Does the pump support remote triggering via TTL signals?
Yes—each channel has dedicated TTL-compatible digital I/O lines for external start/stop/pause commands, pulse-width modulation input for dynamic rate adjustment, and status output signaling completion or fault conditions.
How is accuracy maintained across such a wide flow range?
Accuracy is preserved through real-time closed-loop position feedback via high-resolution optical encoders, adaptive current regulation to compensate for load variation, and factory calibration against NIST-traceable flow standards across nine decades of flow rate (pL/min to mL/min).