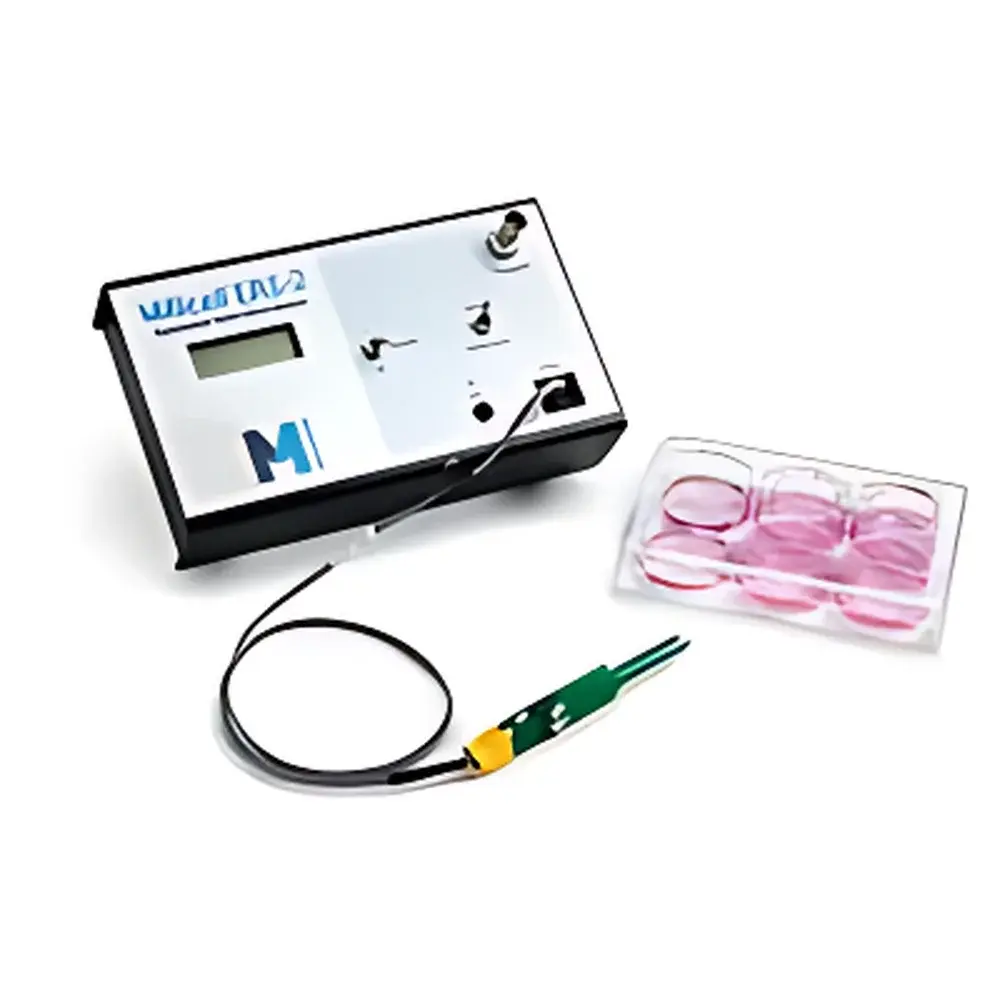
Millipore Millicell-ERS Voltohmmeter MERS00002
| Origin | USA |
|---|---|
| Manufacturer Type | Authorized Distributor |
| Origin Category | Imported |
| Model | MERS00002 |
| Price | USD 3,950 (excl. VAT & shipping) |
| Voltage Range | ±999.0 mV |
| Voltage Resolution | 0.1 mV |
| Resistance Range | 0–10,000 Ω |
| Resistance Resolution | 1 Ω |
| Excitation Current | ±10 µA nominal at 12.5 Hz AC square wave |
| Power Supply | Rechargeable 6 V NiMH 2200 mAh battery |
| Battery Life | 8–10 hrs per full charge |
| Analog Output | 1–10 V (1 mV/Ω scaling) |
| Operating Temperature | 10–38 °C |
| Relative Humidity | 0–90% non-condensing |
| Dimensions | 19 × 11 × 6 cm |
| Weight | 1.4 kg |
Overview
The Millipore Millicell-ERS Voltohmmeter (Model MERS00002) is a precision, benchtop instrument engineered for quantitative electrophysiological assessment of epithelial and endothelial monolayers cultured in Millicell® insert systems. It operates on the principle of low-frequency alternating current (AC) impedance measurement—specifically, a 12.5 Hz square-wave current excitation—to determine transepithelial electrical resistance (TEER) and membrane potential (Vte) without inducing electrochemical artifacts. Unlike DC-based methods, this AC technique eliminates electrode polarization, prevents metal ion deposition on Ag/AgCl electrodes, and ensures zero net charge transfer across the cell layer—critical for maintaining monolayer integrity during longitudinal assays. The system delivers TEER values with high reproducibility and minimal drift, supporting rigorous evaluation of barrier function in applications ranging from drug permeability screening to tight junction dynamics under inflammatory or hypoxic conditions.
Key Features
- AC-based measurement architecture (12.5 Hz square wave, ±10 µA nominal) ensures artifact-free TEER quantification and preserves cellular viability over repeated measurements.
- Dual-parameter readout: simultaneous display of transepithelial voltage (±999.0 mV, 0.1 mV resolution) and resistance (0–10,000 Ω, 1 Ω resolution).
- Integrated analog output (1–10 V, scaled at 1 mV/Ω) enables real-time data logging via external DAQ systems or chart recorders for kinetic studies.
- Rechargeable 6 V NiMH 2200 mAh battery supports up to 10 hours of continuous operation; 12 VDC external charger included for lab-integrated power management.
- Calibration-ready design with STX04 calibration resistor kit (included), traceable to NIST-certified standards, ensuring measurement consistency across instruments and laboratories.
- Compact, lightweight housing (1.4 kg, 19 × 11 × 6 cm) optimized for biosafety cabinet integration and shared-core facility deployment.
Sample Compatibility & Compliance
The Millicell-ERS MERS00002 is validated exclusively for use with MilliporeSigma’s Millicell®-CM, -PC, and -PET insert systems (0.4 µm, 1.0 µm, and 3.0 µm pore sizes). Its STX01 dual-electrode probe set is sterilizable by ethanol immersion and compatible with standard tissue culture protocols. The instrument meets IEC 61010-1:2010 safety requirements for laboratory electrical equipment and complies with electromagnetic compatibility (EMC) standards per EN 61326-1. While not FDA-cleared as a diagnostic device, its measurement methodology aligns with widely adopted pharmacological and toxicological assay guidelines—including OECD TG 428 (skin absorption), ASTM E2289 (in vitro blood-brain barrier models), and USP (permeability testing)—supporting GLP-compliant study execution when paired with documented SOPs and audit-trail-enabled data handling practices.
Software & Data Management
The MERS00002 operates as a standalone hardware platform with no proprietary software dependency. All measurements are displayed directly on its high-contrast LCD screen and exported via analog output for integration into validated third-party acquisition environments (e.g., LabChart, MATLAB, or custom Python-based acquisition scripts using NI DAQmx). For regulated environments, users may implement 21 CFR Part 11-compliant electronic records by coupling the analog output with timestamped, password-protected acquisition software featuring audit trail, user access control, and electronic signature functionality. Raw voltage/resistance values are recorded without internal averaging or filtering—preserving original signal fidelity for post-hoc statistical analysis or outlier detection per ISO/IEC 17025 requirements.
Applications
- Quantitative assessment of tight junction integrity in Caco-2, MDCK, HT-29/B6, and primary human bronchial epithelial (HBEC) monolayers.
- Real-time monitoring of barrier disruption induced by cytokines (e.g., TNF-α, IFN-γ), pathogens, or nanomaterial exposure.
- Permeability coefficient (Papp) calculation in absorption-enhancement or efflux-inhibition studies (e.g., P-gp modulation).
- Validation of organ-on-chip microfluidic barrier models prior to compound dosing.
- Quality control of transwell-based assay platforms during method transfer between R&D and QC laboratories.
FAQ
Can the MERS00002 be used with non-Millicell inserts?
No. The STX01 electrode geometry and spacing are specifically designed for Millicell® insert wells. Use with alternative inserts risks inaccurate current field distribution and invalid TEER values.
Is the analog output calibrated and linear across the full resistance range?
Yes. The 1–10 V output is factory-calibrated to deliver 1 mV per ohm (e.g., 1,000 Ω = 1.000 V), verified during final QA against traceable reference resistors.
How often does the system require recalibration?
Annual calibration is recommended for GLP/GMP workflows; however, daily verification using the included STX04 calibration resistor (1,000 Ω ±0.1%) is advised before critical experiments.
Does the device support temperature compensation?
No. TEER values must be corrected manually for temperature using established conversion factors (e.g., 2.5% per °C deviation from 37 °C) per literature protocols.
Can measurements be performed inside a CO2 incubator?
No. The unit is rated for ambient lab conditions only (10–38 °C, non-condensing humidity). Measurements must be conducted outside the incubator; brief removal (<5 min) of inserts has minimal impact on monolayer physiology if pre-equilibrated.