KANOMAX EP-CRMS Cleanroom Real-Time Particle Monitoring System
| Brand | KANOMAX |
|---|---|
| Origin | Liaoning, China |
| Manufacturer Type | Authorized Distributor |
| Product Category | Domestic |
| Model | EP-CRMS |
| Price Range | USD 14,000 – 28,000 (FOB) |
| Instrument Type | Online Continuous Monitoring System |
| Flow Rate | High-Flow (28.3 L/min standard configuration) |
| Flow Accuracy | < ±1 particle per 5 min |
| Timing Accuracy | ±1 s over 6-min sampling interval |
| Repeatability | ≤ ±10% FS |
| Particle Size Distribution Error | ≤ ±30% |
| Indication Error | < ±30% FS |
Overview
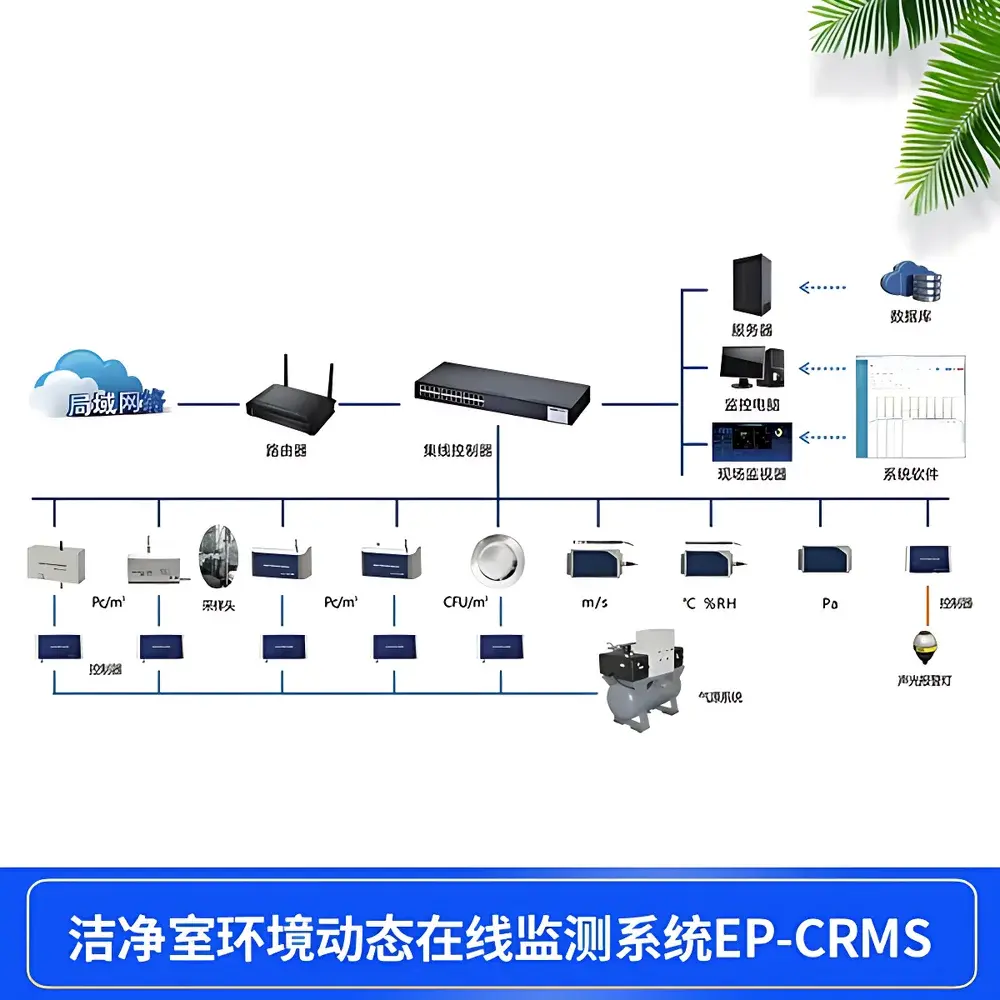
The KANOMAX EP-CRMS Cleanroom Real-Time Particle Monitoring System is an integrated, multi-point online environmental monitoring platform engineered for continuous, compliant surveillance of airborne particulate contamination in regulated pharmaceutical, biotechnology, and microelectronics manufacturing environments. It operates on the principle of light scattering detection—utilizing long-life laser diode sources calibrated per ISO 21501-4 and JIS B 9921—to quantify suspended particles across user-selectable size channels (e.g., 0.3/0.5 µm, 0.5/5.0 µm, or up to six channels including 0.3, 0.5, 1.0, 3.0, 5.0, and 10.0 µm). Designed specifically for Grade A/B cleanroom zones as defined in EU GMP Annex 1 and FDA cGMP guidance, the system enables simultaneous, synchronized acquisition of particle counts, viable microbial data (via optional impactor-based air samplers), and critical environmental parameters—including differential pressure, temperature, relative humidity, airflow velocity, noise, and illuminance. Its architecture supports dynamic process validation by capturing time-stamped, location-tagged datasets that directly feed into statistical process control (SPC), trend analysis, and regulatory reporting workflows.
Key Features
- Modular sensor architecture supporting scalable deployment—from single-point qualification to enterprise-wide distributed monitoring networks.
- Dual-redundant dry-running oil-free vacuum system with automatic failover between two 28.3 L/min pumps, ensuring uninterrupted sampling continuity during maintenance or pump failure.
- Real-time volumetric flow verification and closed-loop control via dedicated particle and microbial sampling controllers—guaranteeing precise 1 m³ cumulative sample volume per test cycle.
- ISO 14698-1–compliant active air sampling for viable microorganisms using stainless-steel (SUS316) impactors with ≥98% collection efficiency at ≥12 m/s impaction velocity and 0.45 m/s isokinetic inlet velocity.
- Comprehensive alarm management: configurable multi-level thresholds (alert, action, alarm), root-cause tagging, audible/visual notification, and automated equipment interlock (e.g., halting fill lines upon deviation).
- Full audit trail compliance: immutable electronic records of user logins, parameter changes, data modifications, alarm events, and electronic signatures—all traceable to individual operators and timestamps.
- Intelligent predictive maintenance: embedded calibration scheduling logic based on sensor runtime, exposure history, and usage metrics—reducing unplanned downtime and manual tracking overhead.
Sample Compatibility & Compliance
The EP-CRMS meets stringent international regulatory requirements for continuous environmental monitoring in GxP environments. Its design, installation, and operational protocols align with EU GMP Annex 1 (2022 revision), FDA 21 CFR Part 11 (electronic records/signatures), USP and , ISO 14644-1/2/3, ISO 21501-4 (particle counter performance), ISO 14698-1/-2 (biocontamination control), and Chinese GMP 2010 with GB/T 16292–2010 (cleanroom testing methods) and GB 50591–2010 (cleanroom construction standards). All wetted parts—including isokinetic sampling probes, internalized PTFE-lined tubing, and 316L stainless steel manifolds—are chemically resistant to vaporized hydrogen peroxide (VHP), ethanol, and other common sterilants used in aseptic processing cycles.
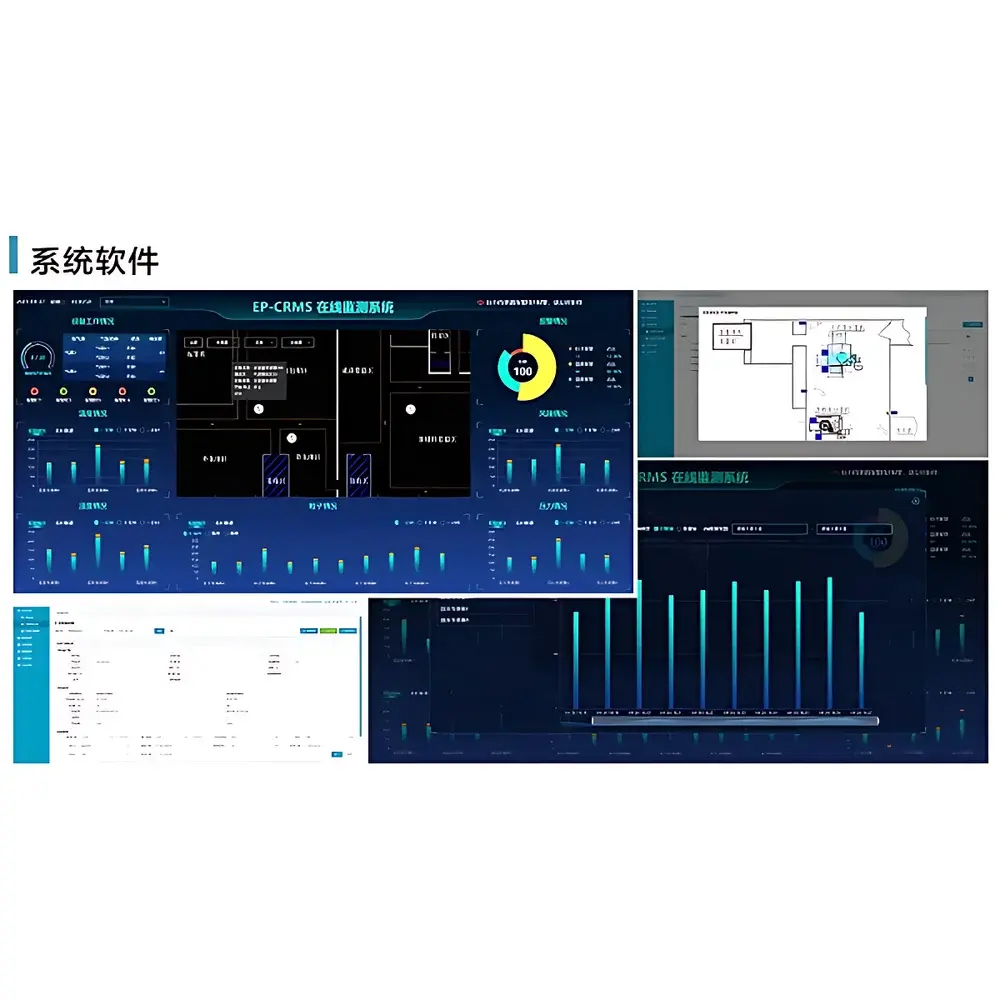
Software & Data Management
The system employs a centralized server-based architecture running validated database software compliant with ALCOA+ principles (Attributable, Legible, Contemporaneous, Original, Accurate, Complete, Consistent, Enduring, Available). Data is stored in relational SQL format with automatic daily backups, encrypted archival, and role-based access controls. Reporting modules generate customizable PDF/Excel exports—including UCL (Upper Confidence Limit) calculations per ISO 14644-1, real-time dashboards, historical trend plots, and automated daily/weekly/monthly summary reports. The software supports full 4Q validation documentation (DQ/IQ/OQ/PQ), FAT/SAT protocols, risk assessments per ICH Q9, and electronic signature workflows satisfying 21 CFR Part 11 Subpart B requirements—including audit trail review, record retention policies, and operator re-authentication for critical actions.
Applications
The EP-CRMS is deployed across high-risk aseptic processing zones including isolators, RABS, lyophilizers, tunnel sterilizers, vial filling lines, and aseptic compounding suites. It supports routine environmental monitoring (EM) programs for both static and dynamic conditions; facilitates media fill investigations; validates cleaning and disinfection cycles; monitors HVAC performance and filter integrity; and provides objective evidence for regulatory inspections (FDA, EMA, NMPA, PMDA). In semiconductor and flat-panel display manufacturing, it serves as a critical tool for ISO Class 3–5 cleanroom certification and contamination source identification during yield loss analysis.
FAQ
Does the EP-CRMS support integration with existing MES or SCADA systems?
Yes—via Modbus RTU/TCP, Ethernet/IP, or OPC UA protocols; custom API endpoints are available under NDA for enterprise-level interoperability.
What is the maximum number of monitoring points supported in a single network?
The system scales to >200 distributed sensors per server node, with hierarchical clustering options for multi-facility deployments.
How frequently must particle sensors be recalibrated?
Per ISO 21501-4, annual calibration is recommended; however, the system’s built-in zero-count verification and flow stability monitoring enable extended calibration intervals under documented risk assessment.
Can the system meet FDA 21 CFR Part 11 requirements out-of-the-box?
Yes—the software includes preconfigured electronic signature workflows, audit trail review tools, and password complexity enforcement aligned with Part 11 Subpart B specifications.
Is remote access to live data and alarms permitted?
Secure remote access is enabled via TLS 1.2–encrypted web interface or dedicated client application, with MFA and session timeout policies enforced per organizational IT security policy.